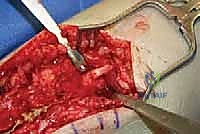
Comprehensive Introduction and Patho-Epidemiology
The management of the recalcitrant, painful neuroma of the foot and ankle represents one of the most formidable challenges in modern orthopedic surgery. This operative masterclass is designed to transcend the rudimentary concepts of peripheral nerve transection, elevating the discourse to the meticulous identification, precise resection, and strategic proximal burial of disorganized neural tissue. Our definitive objective is to provide enduring relief from the debilitating, often life-altering neuropathic pain that characterizes these lesions. To achieve this, the operating surgeon must possess not only an encyclopedic understanding of lower extremity neuroanatomy but also a refined, almost microscopic surgical technique. The procedure, while mechanically straightforward in its description, demands an intimate comprehension of the peripheral nervous system's capricious anatomical variability and its profound capacity for maladaptive regeneration.
To effectively combat this pathology, we must first appreciate the complex genesis of these painful lesions at a cellular and macroscopic level. Fundamentally, a neuroma is a disorganized, chaotic proliferation of neural tissue, Schwann cells, and fibroblasts that forms at the site of a severe nerve injury. When a peripheral nerve is transected, crushed, or stretched beyond its physiological limits, the proximal nerve stump initiates a robust regenerative response, sending out a multitude of axonal sprouts. Under normal circumstances, these sprouts seek the distal endoneurial tubes to guide their reinnervation. However, when this distal pathway is obliterated, obstructed by scar tissue, or surgically absent, these regenerating axons become trapped. They coil upon themselves, forming a tangled, hypersensitive bulbous mass—the terminal or stump neuroma. This mass acts as an ectopic pacemaker, generating spontaneous, aberrant action potentials that the central nervous system interprets as severe, burning, or electrical pain.
The etiology of nerve damage in the foot and ankle is multifactorial, often culminating in this maladaptive neuroma formation. Direct contusion from high-energy blunt trauma or crush injuries can directly obliterate the fascicular architecture of the nerve. Stretch injuries represent a particularly insidious mechanism in the lower extremity. Consider the biomechanics of a severe ankle inversion sprain or a complex rotational ankle fracture; the extreme, sudden motion can stretch peripheral nerves well beyond their elastic limit, inducing immediate Wallerian degeneration. The superficial peroneal nerve, for instance, is highly susceptible to profound stretch injury during severe ankle inversion, as is the sural nerve. The structural integrity of the nerve is compromised, leading to intraneural fibrosis and subsequent neuroma-in-continuity formation.
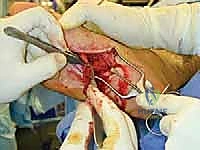
Unfortunately, iatrogenic insult remains the most frequent catalyst for neuroma formation in the foot and ankle surgeon's practice. Surgical incisions, aggressive retraction, errant suture placement, or misplaced arthroscopic portals can inadvertently lacerate or entrap regional nerves. A Lisfranc fracture open reduction and internal fixation (ORIF) or a complex midfoot arthrodesis inherently endangers the superficial and deep peroneal nerves. Hallux valgus corrections, particularly those utilizing dorsomedial incisions, notoriously threaten the dorsomedial cutaneous nerve. Calcaneal and fifth metatarsal fracture fixations place the sural nerve at significant, unavoidable risk. Even routine Achilles tendon repairs or Haglund resections can compromise the sural nerve, specifically its posterior arborizations. Neuromas present along a vast clinical spectrum, ranging from small, asymptomatic bulbous swellings to massive accumulations of hypersensitive nerve endings that generate crippling allodynia. Our operative focus is directed solely at those unpredictable, intensely painful neuromas that have definitively failed exhaustive conservative management.
Detailed Surgical Anatomy and Biomechanics
Before a scalpel ever touches the skin, the orthopedic surgeon must possess a three-dimensional, encyclopedic knowledge of the regional neurovascular anatomy. The foot and ankle are innervated by five primary sensory nerves, but the surgeon must operate under the assumption that anatomical variability is the absolute rule, not the exception. Relying solely on textbook illustrations will inevitably lead to surgical misadventure.
The Superficial Peroneal Nerve
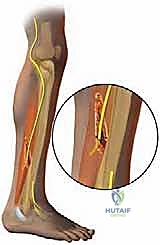
Emerging from the common peroneal nerve, the superficial peroneal nerve (SPN) descends through the lateral compartment of the leg, initially coursing deep to the peroneus longus and brevis muscles. Approximately two-thirds of the way down the leg—a highly variable transition point—it pierces the crural fascia to become a superficial structure. It then courses anterior to the fibula, dividing into its terminal branches: the medial dorsal cutaneous nerve and the intermediate dorsal cutaneous nerve, which provide sensory innervation to the majority of the dorsal foot.
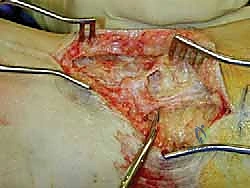
The SPN is exquisitely vulnerable during lateral approach ankle fracture ORIF, the establishment of the anterolateral arthroscopic portal, and virtually any lateral midfoot reconstructive procedure. Its terminal medial branch, the dorsomedial cutaneous nerve, is particularly at risk during medial eminence resection in bunion surgery. Clinically, following a severe stretch injury or prior surgical trauma, the SPN frequently becomes densely adherent to the underlying muscle and investing fascia. This dense cicatricial adherence obliterates normal tissue planes, making surgical dissection highly treacherous and significantly increasing the risk of secondary iatrogenic injury if not approached with meticulous, loupe-assisted magnification.
The Deep Peroneal Nerve
Originating alongside the SPN from the common peroneal nerve, the deep peroneal nerve (DPN) takes a deeper, more protected course. It runs intimately with the anterior tibial artery and accompanying veins, forming a neurovascular bundle that descends along the interosseous membrane in the anterior compartment. It passes beneath the superior and inferior extensor retinacula at the level of the ankle joint.
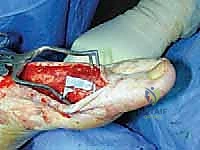
While primarily a motor nerve to the anterior compartment musculature (tibialis anterior, extensor hallucis longus, extensor digitorum longus, and peroneus tertius), it provides crucial sensory innervation to the first dorsal web space. The DPN is at risk during severe dorsal crush injuries, Lisfranc fracture-dislocations, and anterior approaches to the ankle or midfoot. Although major motor nerves are generally spared from resection due to the resulting catastrophic functional deficit, the terminal sensory branches of the DPN in the midfoot can be resected and buried with minimal functional consequence, provided the primary motor branches to the leg have already branched off proximally.
The Sural Nerve
The sural nerve is formed by the variable confluence of the medial sural cutaneous nerve (a branch of the tibial nerve) and the lateral sural cutaneous nerve (a branch of the common peroneal nerve). It courses superficially over the gastrocnemius muscle bellies, descending between the peroneal tendons and the lateral border of the Achilles tendon. It wraps around the posterolateral aspect of the ankle, providing sensory innervation to the lateral hindfoot, the lateral malleolus, and the lateral border of the foot down to the fifth digit.
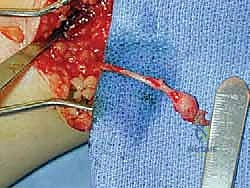
Due to its superficial and relatively exposed posterolateral course, the sural nerve is highly susceptible to iatrogenic injury during extensile lateral approaches for calcaneal ORIF, fifth metatarsal base fixations, Achilles tendon ruptures, and lateral ankle ligamentous reconstructions (e.g., Broström-Gould procedures). While anatomically one of the easiest nerves to identify surgically, it has a notorious clinical reputation for aggressive postoperative regeneration. Sural neuromas frequently require exceptionally proximal dissection and deep intramuscular burial into the soleus or gastrocnemius muscle bellies to prevent recurrent symptomatic stump neuroma formation.
The Saphenous and Tibial Nerves
The saphenous nerve, the longest terminal branch of the femoral nerve, descends along the medial aspect of the leg in close, often intertwined association with the great saphenous vein. It passes anterior to the medial malleolus, providing sensation to the medial leg, ankle, and midfoot. Its intimate vascular association makes careful, blunt dissection crucial to avoid catastrophic venous hemorrhage or secondary nerve ischemia. It is frequently injured during medial malleolar fracture fixation or vein harvesting procedures.
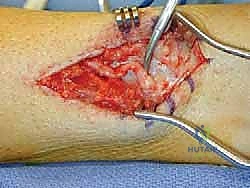
The tibial nerve descends posterior to the medial malleolus through the fibro-osseous tarsal tunnel before arborizing into the medial and lateral plantar nerves, and the medial calcaneal branches. As a critical mixed motor and sensory nerve, the main trunk of the tibial nerve is almost never a primary target for neuroma resection, as transection would result in devastating intrinsic muscle paralysis and plantar anesthesia, leading to Charcot arthropathy or neurotrophic ulceration. Resection is strictly reserved for radical salvage scenarios or pre-amputation pain control. However, understanding its precise anatomy is vital for differentiating a true neuroma from compressive tarsal tunnel syndrome.
Exhaustive Indications and Contraindications
The decision to proceed with surgical transection and proximal burial of a neuroma is a definitive, irreversible intervention. It is a "one-way street" that permanently sacrifices the sensory territory of the target nerve. Therefore, patient selection must be ruthlessly stringent, relying on a combination of rigorous clinical examination, exhaustive non-operative trials, and highly specific diagnostic modalities.
| Category | Specific Criteria | Clinical Rationale |
|---|---|---|
| Primary Indications | Refractory Neuropathic Pain | Failure of >6 months of comprehensive conservative management (gabapentinoids, SNRIs, orthoses, desensitization). |
| Positive Diagnostic Nerve Block | Complete or near-complete (>80%) temporary relief of primary pain following targeted local anesthetic injection proximal to the neuroma. | |
| Identifiable Neuroma-in-Continuity | Clear clinical (Tinel's sign) or radiographic (Ultrasound/MRI) evidence of a discrete neuroma that is mechanically irritated. | |
| Failed Prior Decompression | Persistent pain following a previous attempt at external neurolysis or nerve decompression where the nerve is now encased in unyielding scar tissue. | |
| Absolute Contraindications | Active Complex Regional Pain Syndrome (CRPS) | Operating during the acute, sympathetically mediated phase of CRPS will exponentially worsen the condition and trigger a catastrophic pain crisis. |
| Reversible Entrapment Neuropathy | If the nerve is structurally intact but compressed (e.g., primary tarsal tunnel syndrome), decompression is indicated, not destructive transection. | |
| Active Local Infection | Cellulitis or deep space infection at the planned surgical site absolutely precludes elective nerve surgery. | |
| Unrealistic Patient Expectations | Patients who do not comprehend that the surgery will result in permanent numbness in the nerve's distribution are not surgical candidates. |
The most critical preoperative indicator of surgical success is the diagnostic nerve block. This must be performed meticulously by the operating surgeon. A small volume (1-2 cc) of short-acting local anesthetic (e.g., 1% Lidocaine) is injected precisely around the nerve trunk proximal to the suspected neuroma site. The patient is then immediately re-examined. If the primary burning or electrical pain is entirely eradicated while the block is active, the surgeon can be highly confident that the targeted nerve is the primary pain generator. Failure to achieve relief suggests either an incorrect anatomical diagnosis, central pain sensitization, or an alternative pathology such as a radiculopathy.
Contraindications must be actively ruled out. The presence of generalized, non-dermatomal hyperalgesia, profound trophic skin changes, and vasomotor instability strongly suggests Complex Regional Pain Syndrome (CRPS). While a localized neuroma can act as the nidus for CRPS, operating on a limb with active, spreading sympathetic dysregulation is fraught with peril. In such cases, aggressive sympathetic blocks, rigorous physical therapy, and neuromodulatory pharmacotherapy must precede any consideration of surgical intervention.
Pre-Operative Planning, Templating, and Patient Positioning
Thorough preoperative planning is the bedrock of a successful neuroma resection. The surgeon must synthesize historical data, physical examination findings, and advanced imaging to formulate a precise surgical blueprint. The physical examination is paramount; the surgeon must map the exact point of maximal tenderness and trace the proximal course of the nerve. A highly localized, strongly positive Tinel's sign—where gentle percussion reproduces the patient's exact electrical, shooting pain—is the hallmark of the neuroma. This precise location should be marked on the patient's skin in the preoperative holding area while the patient is awake and able to provide feedback.
Advanced imaging, while secondary to the clinical exam, provides invaluable anatomical context. High-resolution ultrasonography has emerged as the gold standard for visualizing peripheral neuromas in the lower extremity. It allows for dynamic assessment, precise measurement of the neuroma bulb, and evaluation of the surrounding soft tissue envelope. Magnetic Resonance Imaging (MRI), particularly utilizing high-resolution neurography protocols (e.g., 3D STIR sequences), can beautifully delineate the disorganized neural tissue, surrounding perineural edema, and any space-occupying lesions (ganglion cysts, retained hardware) contributing to nerve irritation. Electrodiagnostic studies (EMG/NCS) are generally unhelpful for distal sensory neuromas but remain vital for ruling out proximal radiculopathy or generalized peripheral neuropathy.
Patient positioning is dictated entirely by the anatomical target. For the superficial and deep peroneal nerves, the patient is positioned supine with a bump under the ipsilateral hip to internally rotate the leg, bringing the anterolateral structures into optimal view. For the sural nerve, the patient is best positioned prone or in the lateral decubitus position, allowing unhindered access to the posterolateral calf and hindfoot. A thigh tourniquet is universally applied to ensure a bloodless surgical field, which is an absolute necessity for microsurgical nerve dissection. However, prior to exsanguination, it is often helpful to mark the course of superficial veins (like the lesser saphenous vein) to avoid inadvertent damage during the approach.
Anesthetic considerations require careful dialogue with the anesthesia team. While general anesthesia is standard, regional anesthesia (popliteal or sciatic nerve blocks) can provide excellent postoperative pain control. However, in patients with a history of profound neuropathic pain or suspected early CRPS, the placement of an epidural catheter should be strongly considered. Epidural anesthesia blunts the massive afferent pain barrage that occurs during nerve transection, theoretically reducing the risk of central sensitization and postoperative sympathetic flare-ups.
Step-by-Step Surgical Approach and Fixation Technique
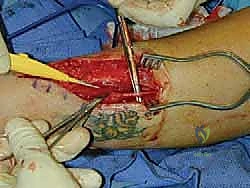
The surgical execution of neuroma resection and proximal burial is an exercise in meticulous tissue handling and microsurgical precision. The procedure is performed under continuous loupe magnification (minimum 2.5x to 3.5x) utilizing specialized micro-instruments. The overarching philosophy is to identify the nerve in pristine, unscarred tissue proximal to the zone of injury, trace it distally to the neuroma, resect the pathological tissue entirely, and secure the proximal stump in a mechanically shielded, well-vascularized environment.
Incision and Proximal Dissection
The surgical incision is planned longitudinally, parallel to the course of the nerve, but ideally avoiding placement directly over the anticipated burial site to prevent future incisional irritation. If previous surgical scars exist, they may be utilized, but the surgeon must be prepared for dense subcutaneous fibrosis. The initial dissection proceeds through the dermis and subcutaneous fat using sharp dissection or needle-tip electrocautery on a very low setting. Blunt dissection with tenotomy scissors is utilized to spread the tissues longitudinally, parallel to the neurovascular structures.
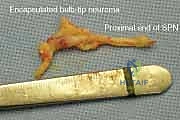
The critical first step is identifying the normal, healthy nerve trunk proximal to the neuroma. Attempting to find the nerve directly within the scarred neuroma bed is a recipe for disorientation and inadvertent partial transection. Once the healthy, glistening white epineurium of the proximal nerve is identified, it is gently encircled with a vessel loop. The nerve is then carefully neurolysed and traced distally toward the zone of pathology.
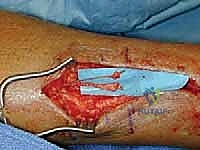
Neuroma Resection
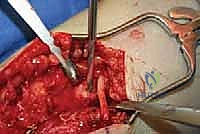
As the dissection progresses distally, the nerve will typically lose its normal fascicular architecture, expanding into a fibrotic, bulbous mass—the neuroma. This mass is often densely adherent to surrounding fascia, tendons, or even bone. The neuroma is meticulously dissected free from its attachments. Once fully mobilized, the nerve must be transected. This is not a casual cut; it must be a single, decisive, and perfectly orthogonal transection using a fresh, ultra-sharp microsurgical blade (e.g., a #15 or #11 blade) or specialized nerve scissors. Crushing the nerve with dull scissors will incite a more aggressive regenerative response.

The transection should occur far enough proximally to ensure that only healthy, unscarred fascicles remain in the proximal stump. The resected neuroma is invariably sent for formal histopathological examination to confirm the diagnosis and rule out rare peripheral nerve sheath tumors (e.g., schwannoma, neurofibroma).
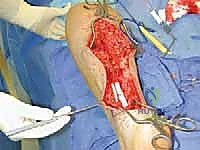
Proximal Burial Techniques
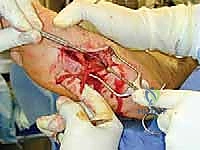
The fate of the proximal nerve stump dictates the ultimate success or failure of the operation. If left free in the subcutaneous tissue, a recurrent, painful stump neuroma is a virtual certainty. The goal of proximal burial is to place the regenerating nerve endings into an environment free from mechanical traction, external pressure, and scar adherence. Two primary techniques are employed: intramuscular burial and intraosseous burial.
Intramuscular Burial: This is the most common technique for nerves like the sural or superficial peroneal. A healthy, robust muscle belly proximal to the resection site is identified (e.g., the soleus or the peroneus brevis). A small, deep pocket is created within the muscle fibers using a hemostat. The nerve stump is gently transposed into this muscular crypt. Crucially, the nerve must lie completely tension-free; any longitudinal traction will cause ischemic pain and early failure. To prevent the nerve from retracting or pulling out of the muscle, the epineurium of the nerve stump is meticulously sutured to the surrounding epimysium using 6-0 or 7-0 non-absorbable monofilament sutures (e.g., Prolene or Nylon).
Intraosseous Burial: For certain nerves, particularly in the distal foot where muscle bellies are sparse, intraosseous burial into the metatarsals, cuboid, or calcaneus can be highly effective. A small drill hole (typically 3.0mm to 4.0mm) is created in the cortical bone. The nerve stump is carefully inserted into the intramedullary canal. It is secured either by suturing the epineurium to the adjacent periosteum or by utilizing a specialized nerve anchor or fibrin glue. This technique provides absolute mechanical shielding but requires careful attention to avoid sharp cortical edges that could sever the nerve stump.
Following secure burial, the tourniquet is deflated, and meticulous hemostasis is achieved. Hematoma formation in the burial bed can lead to intense fibrosis and failure. The wound is closed in layered fashion, ensuring the deep dermal layers are approximated without placing any tension on the underlying buried nerve.
Complications, Incidence Rates, and Salvage Management
Despite flawless surgical technique, neuroma surgery carries a notoriously high complication profile. The peripheral nervous system's biological imperative to regenerate makes long-term success challenging. The surgeon must be intimately familiar with potential modes of failure and possess a robust armamentarium of salvage techniques.
| Complication | Estimated Incidence | Pathophysiology & Clinical Presentation | Salvage Management Strategy |
|---|---|---|---|
| Recurrent Stump Neuroma | 15% - 30% | The buried nerve stump escapes its muscular/osseous crypt or the burial site itself becomes subject to mechanical compression. Presents as recurrent, localized Tinel's sign and burning pain at the burial site. | Revision surgery with more proximal resection and deeper burial. Consideration of Targeted Muscle Reinnervation (TMR) or Regenerative Peripheral Nerve Interfaces (RPNI). |
| Complex Regional Pain Syndrome (CRPS) | 5% - 10% | Sympathetic nervous system over-activation triggered by surgical trauma. Presents as severe, non-dermatomal pain, allodynia, skin color/temperature changes, and severe edema. | Immediate aggressive intervention. High-dose oral corticosteroids, gabapentinoids, sympathetic nerve blocks (lumbar sympathetic block), and intense desensitization physical therapy. |
| Wound Dehiscence / Infection | 2% - 5% | Standard surgical risk, exacerbated by poor vascularity or underlying medical comorbidities (diabetes, smoking). | Local wound care, appropriate culture-directed oral or intravenous antibiotics. Rare need for formal operative debridement unless deep space involvement occurs. |
| Phantom Limb/Nerve Pain | Variable | Central nervous system maladaptation to the loss of peripheral sensory input. The patient feels pain in the numb, resected territory. | Primarily pharmacological management (SNRIs, Tricyclic Antidepressants, Gabapentinoids). Mirror box therapy and cognitive behavioral therapy. |
When a standard proximal burial fails, the orthopedic surgeon must escalate to advanced salvage techniques. Historically, wrapping the nerve stump in synthetic conduits or vein grafts was attempted, but results were inconsistent. Modern salvage heavily favors physiological management of the nerve stump.
Targeted Muscle Reinnervation (TMR) and Regenerative Peripheral Nerve Interfaces (RPNI) represent the vanguard of neuroma salvage. In TMR, the painful neuroma is resected, and the proximal nerve stump is coapted (sutured) to a nearby, expendable motor nerve branch supplying a local muscle. This gives the regenerating sensory axons a "target" to innervate, physiologically downregulating the chaotic regenerative response and preventing neuroma formation. RPNI involves wrapping the transected nerve stump in a free autologous muscle graft, providing a highly vascularized, target-rich environment for the axons to harmlessly arborize within the graft rather than forming a painful mass.
Phased Post-Operative Rehabilitation Protocols
The surgical procedure is only the first phase of treatment; the postoperative rehabilitation protocol is equally critical to ensuring a successful, pain-free outcome. The protocol is designed to meticulously balance the need for early tissue healing with the absolute necessity of preventing nerve adherence and central pain sensitization.
Phase I: Maximum Protection and Healing (Weeks 0-3)
Immediately postoperatively, the primary objective is to protect the fragile burial site and ensure primary wound healing. The patient is typically placed in a bulky Jones dressing and a well-padded short leg splint or controlled ankle motion (CAM) boot. Weight-bearing status is strictly dependent on the burial site. If the nerve was buried intramuscularly in the proximal leg, weight-bearing as tolerated in the boot may be permissible. However, if an intraosseous burial was performed in the foot, strict non-weight-bearing is mandatory to prevent micromotion and subsequent pull-out of the nerve stump. The limb must remain elevated to minimize edema, which can increase tissue tension and compress the newly buried nerve.
Phase II: Controlled Mobilization and Desensitization (Weeks 3-6)
Once the surgical incision has completely healed and sutures are removed, the focus shifts to controlled mobilization and the initiation of desensitization. The patient is transitioned out of the splint, and active and passive range of motion exercises for the ankle and subtalar joints are commenced. The goal is to induce gentle, physiological gliding of
Clinical & Radiographic Imaging Archive