SUBPECTORAL BICEPS TENODESIS WITH AN INTERFERENCE SCREW
The management of proximal biceps tendon pathology has evolved significantly, with subpectoral biceps tenodesis emerging as the gold standard for active patients. The mini-open subpectoral approach, popularized by Mazzocca et al., is performed through a small cosmetic incision over the bicipital groove via the anterior aspect of the deltoid or a subdeltoid incision.
By securing the tendon distal to the bicipital groove, this technique eliminates the groove as a source of persistent anterior shoulder pain—a common complication in high-groove or arthroscopic tenodesis techniques.
Indications and Patient Selection
The indications for the subpectoral interference screw technique mirror those of arthroscopic biceps tenodesis. Surgical intervention is highly recommended in the presence of:
* Severe Tenopathy: Greater than 50% macroscopic tenopathy with persistent, refractory anterior shoulder pain.
* Partial Tears: Tears involving >50% of the long head of the biceps (LHB) tendon footprint or substance.
* Biceps Instability: Subluxation or dislocation of the LHB out of the bicipital groove, often associated with subscapularis tears or pulley lesions.
* SLAP Lesions in Older Patients: Type II SLAP tears in patients over 40 years of age, where tenodesis has shown superior outcomes compared to primary repair.
💡 Clinical Pearl: The Length-Tension Relationship
The primary biomechanical advantage of the subpectoral approach is the reliable restoration of the anatomical length-tension relationship of the biceps muscle. Failure to properly tension the tendon leads to early fatigue, cramping, and the cosmetic "Popeye" deformity.
Preoperative Preparation and Positioning
- Anesthesia: General anesthesia is typically combined with an interscalene regional nerve block for optimal perioperative analgesia.
- Positioning: The patient is placed in either the beach-chair or lateral decubitus position, depending on the surgeon's preference for the concomitant arthroscopic portion of the procedure.
- Diagnostic Arthroscopy: Make a standard posterior portal. Perform a comprehensive diagnostic sweep of the glenohumeral joint.
- Arthroscopic Release: Place a traction suture through the proximal biceps tendon. Using an arthroscopic electrocautery device or biting forceps, release the LHB tendon directly at its origin on the superior labrum/supraglenoid tubercle.
Surgical Technique: Step-by-Step
1. Surgical Exposure
With the arm abducted and internally rotated, palpate the inferior border of the pectoralis major tendon. Make a 1-cm longitudinal incision on the medial aspect of the arm, starting just superior to the inferior border of the pectoralis tendon and continuing to 3 cm below it.
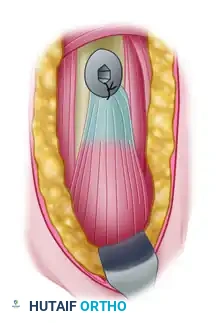
Inject the incision site with a local anesthetic containing epinephrine to ensure subcutaneous hemostasis and provide preemptive analgesia.
⚠️ Surgical Warning: Incision Placement
For optimal cosmesis, the entire incision may be placed within the axillary fold if the surgeon is intimately familiar with the local anatomy. However, straying too far medially endangers the neurovascular bundle.
Cut down through the subcuticular tissue, utilizing electrocautery to meticulously control bleeding. A Gelpi or Weitlaner self-retaining retractor is highly effective for maintaining exposure.
2. Deep Dissection and Tendon Identification
Clear the overlying fatty tissue until the fascial layer overlying the pectoralis major, coracobrachialis, and short head of the biceps is identified.
Pitfall Avoidance: If these anatomical landmarks are not readily visible, the dissection plane is likely too lateral. Conversely, if the cephalic vein is encountered within the deltopectoral groove, the dissection is too proximal and lateral.
After identifying the inferior border of the pectoralis major, incise the fascia overlying the coracobrachialis and biceps in a proximal-to-distal direction. It is critical to visualize the horizontal fibers of the pectoralis muscle and dissect strictly below this level.
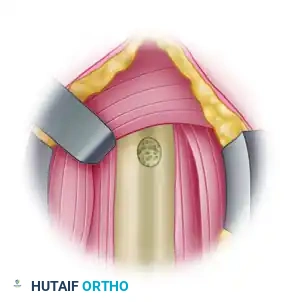
Using blunt finger dissection under the inferior edge of the pectoralis muscle, palpate up the anteromedial humerus to identify the longitudinal, fusiform structure of the LHB tendon.
3. Retraction and Tendon Delivery
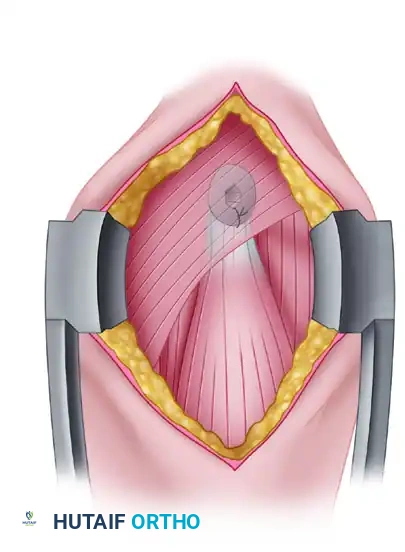
Place a pointed Hohmann retractor into the pectoralis major tendon, resting the tip on the proximal humerus to retract the muscle belly proximally and laterally. Position a blunt Chandler retractor on the medial aspect of the humerus to gently retract the coracobrachialis and the short head of the biceps.
⚠️ Surgical Warning: Musculocutaneous Nerve
Vigorous medial retraction with the Chandler retractor must be strictly avoided. The musculocutaneous nerve typically penetrates the coracobrachialis 5 to 8 cm distal to the coracoid process and is highly susceptible to traction neuropraxia.
Once the biceps tendon is identified, pass a blunt probe or right-angle clamp deep to it. Pull the tendon to deliver it fully into the surgical wound.
4. Tendon Preparation
Reflect the local periosteum to create a rectangular footprint roughly 2 × 1 cm, located approximately 1 cm proximal to the inferior border of the pectoralis major tendon.
To ensure appropriate tensioning, resect the diseased proximal portion of the tendon. You must leave exactly 20 to 25 mm of tendon proximal to the musculotendinous junction of the biceps.
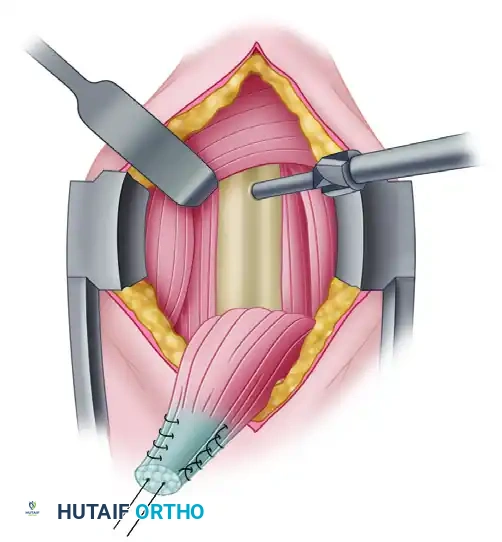
Using a Krackow stitch or a locking whipstitch, weave a No. 2 FiberWire (or equivalent high-strength nonabsorbable suture) into the proximal 15 mm of the tendon.
Secure enough of the tendon to guarantee adequate interference fixation within the bony tunnel. The goal is to position the musculotendinous junction exactly beneath the inferior border of the pectoralis major tendon. This step is the absolute crux of the procedure for restoring the muscle-tendon unit's proper tension and preventing cosmetic deformity.
5. Bone Tunnel Preparation
Using a guidewire, drill perpendicular to the humeral shaft. Over-ream with an 8-mm reamer to create a unicortical bone tunnel exactly 15 mm deep. Thoroughly irrigate the tunnel to remove all bone debris, which can impede screw insertion.

6. Interference Screw Fixation
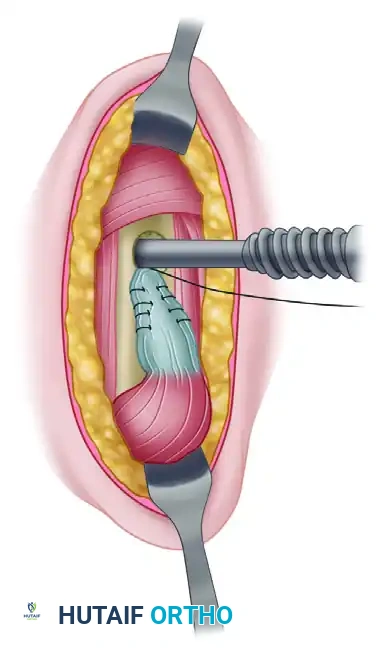
Thread one limb of the suture through the cannulated Arthrex biotenodesis screwdriver and the corresponding bioabsorbable or PEEK interference screw (typically 8 × 12 mm). Place a hemostat on the distal end of the suture to maintain tension.
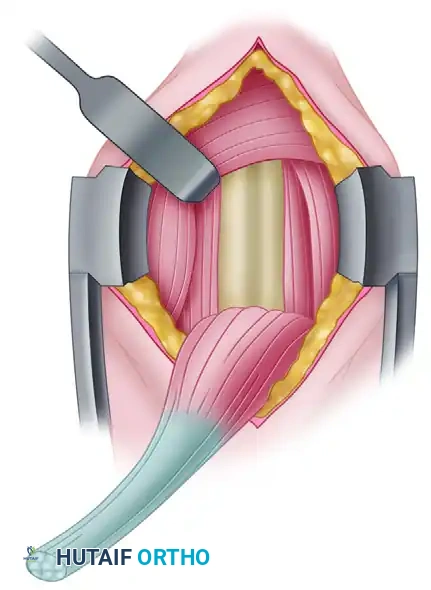
Place the tip of the tenodesis screwdriver into the bone tunnel, pushing the tendon into the socket. Advance the screw over the tendon, maintaining axial pressure.
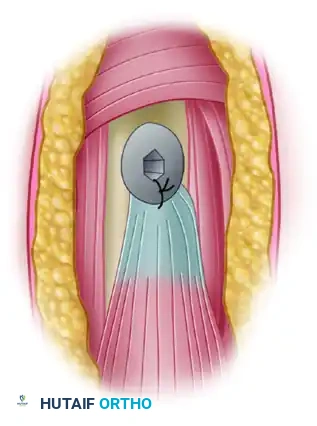
When the screw head sits flush with the anterior humeral cortex, carefully remove the screwdriver.
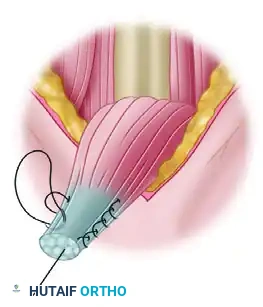
Tie the free limbs of the suture over the top of the screw and tendon. This creates a hybrid fixation construct, combining the aperture compression of the interference screw with the suspensory stability of a suture anchor.
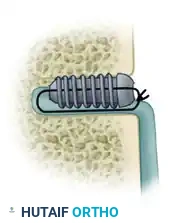
Upon completion, the musculotendinous junction must rest in its exact anatomical location underneath the inferior border of the pectoralis major. Complete the procedure with standard layered wound closure.
ANTERIOR SHOULDER INSTABILITY: EVIDENCE-BASED MANAGEMENT
Biceps pathology frequently coexists with anterior shoulder instability, particularly in overhead athletes and trauma patients. Since Detrisac and Johnson first introduced the staple capsulorrhaphy in the 1970s, arthroscopic shoulder stabilization has undergone a massive technological evolution. The development of modern suture anchors, capsular plication techniques, and rotator interval closures has reduced the recurrence rate of arthroscopic stabilization to levels comparable to open techniques—provided patient selection is rigorous.
Evolution of Indications and Contraindications
As arthroscopic techniques evolved, the absolute and relative contraindications became clearer. The literature demonstrates that arthroscopic Bankart repair is highly successful in the absence of significant bone loss, but fails predictably when critical structural deficits are ignored.
- The Burkhart and DeBeer Paradigm: In a landmark study of 190 patients, Burkhart and DeBeer noted a catastrophic increase in recurrence rates (from 6.5% to 89%) in contact athletes when a >25% glenoid bone defect or an "engaging" Hill-Sachs lesion was present.
- The Voos et al. Study: Voos reported an overall 18% recurrence rate, which spiked to 37.5% in patients with specific risk factors: age younger than 20 years, generalized hyperlaxity, and a Hill-Sachs lesion larger than 25 mm³.
- The Boileau Instability Severity Index Score (ISIS): Boileau et al. observed 91 patients over 36 months, noting a 15.3% overall recurrence rate. To better predict failure, they developed the ISIS score. A score over 6 points strongly suggests that an arthroscopic Bankart repair will fail, and an open Latarjet procedure should be considered.
Instability Severity Index Score (ISIS)
| Prognostic Factors | Points |
|---|---|
| Age at surgery (yr) | |
| ≤ 20 | 2 |
| > 20 | 0 |
| Degree of sport participation | |
| Competitive | 2 |
| Recreational or none | 0 |
| Type of sport | |
| Contact or forced overhead | 1 |
| Other | 0 |
| Shoulder hyperlaxity | |
| Anterior or inferior hyperlaxity | 1 |
| Normal laxity | 0 |
| Hill-Sachs on AP radiograph | |
| Visible in external rotation | 2 |
| Not visible in external rotation | 0 |
| Glenoid loss of contour on AP radiograph | |
| Loss of contour | 2 |
| No lesion | 0 |
| Total Maximum Points | 10 |
(Adapted from Balg F, Boileau P: The instability severity index score, J Bone Joint Surg 89B:1470, 2007.)
Advanced Imaging: When to Obtain a CT Scan
While MRI or MR Arthrogram (MRA) is the gold standard for evaluating soft tissue (labrum, capsule, rotator cuff), a 3D Computed Tomography (CT) scan is critical for quantifying bone loss. Saito et al. utilized 3D CT to evaluate glenoid rim defects, finding that in 123 recurrent dislocators, the defect orientation consistently pointed to the 3-o’clock position.
💡 Clinical Pearl: Indications for CT in Instability (Box 52-2)
- Radiographic or MRI evidence of bone loss
- High-energy trauma mechanisms
- Multiple instability episodes (chronic dislocators)
- Failed prior stabilization procedures
- Instability occurring in the mid-range of motion
- Inferior instability patterns
- Instability occurring during sleep
- History of difficult closed reductions
Pathoanatomy: ALPSA and HAGL Lesions
Anterior Labroligamentous Periosteal Sleeve Avulsion (ALPSA)
Described by Neviaser, the ALPSA lesion involves the anterior labrum stripping off the glenoid and displacing medially along the scapular neck, taking the intact periosteum with it. Ozbaydar et al. found ALPSA lesions in 28% of 99 instability patients. These patients were typically younger (<21 years) and had a higher number of preoperative dislocations (12.3 vs. 4.9) compared to standard Bankart patients. The failure rate for arthroscopic repair of ALPSA lesions was notably higher (19.2%) than for standard Bankart lesions (7.4%), emphasizing the need to aggressively mobilize the labrum back to the glenoid rim before fixation.
Humeral Avulsion of the Glenohumeral Ligament (HAGL)
Originally described by Nicola in 1942 and later expanded upon by Bach, Wolf, and Baker, the HAGL lesion represents an avulsion of the inferior glenohumeral ligament from its humeral insertion. Wolf identified HAGL lesions in 9.3% of instability patients.
HAGL lesions can occur anteriorly or posteriorly, with or without a bony avulsion (B-HAGL), resulting in a "floating" inferior glenohumeral ligament. Preoperative MRI in the acute setting, or MRA in the subacute setting, is vital for diagnosis. While arthroscopic repairs are described, most authors agree that a mini-open procedure remains the most reproducible method for addressing HAGL lesions.
Surgical Decision Making and Modifiers
Modern shoulder stabilization requires a tailored approach. Barber, Ide, and Mazzocca reported arthroscopic recurrence rates of 7% to 11% in contact athletes, which compares favorably to historical open Bankart procedures (Uhorchak 23%, Pagnani 3%, Magnusson 11%).
However, specific modifiers dictate a shift in surgical strategy:
💡 Clinical Pearl: Indications for Shoulder Stabilization Modifiers (Box 52-3)
- Bone loss > 25%: Mandates a Glenoid Latarjet procedure or bone grafting.
- Humeral head defect > 6 mm deep: Consider a Remplissage procedure (infraspinatus tenodesis into the defect) for collision athletes.
- Soft tissue multidirectional instability: Requires an arthroscopic capsular shift and plication.
- ALPSA Lesion: Must restore anatomy anteriorly; consider aggressive capsular plication.
- Anterior HAGL: Mini-open or advanced arthroscopic repair.
- Posterior HAGL: Arthroscopic repair.
- Concomitant SLAP or Cuff Lesion: Requires simultaneous repair.
Postoperative Rehabilitation Protocol
Whether performing a subpectoral biceps tenodesis, an anterior stabilization, or both, the postoperative protocol must protect the surgical construct while preventing stiffness.
- Phase I (0-4 Weeks): Sling immobilization. Passive range of motion (PROM) for the shoulder. For tenodesis, avoid active elbow flexion and active forearm supination to protect the interference screw construct.
- Phase II (4-8 Weeks): Discontinue sling. Begin active-assisted range of motion (AAROM) progressing to active range of motion (AROM). Initiate gentle isometric strengthening. For instability repairs, limit external rotation to 30-40 degrees to protect the anterior capsule.
- Phase III (8-12 Weeks): Advance strengthening. Begin light isotonic bicep curls and rotator cuff strengthening.
- Phase IV (3-6 Months): Return to sport-specific activities. Overhead athletes and contact sport participants must demonstrate symmetric strength and dynamic stability before full clearance.
At this time, arthroscopic procedures with capsular plication, interval closure, and labral repair produce results comparable to open procedures in appropriately selected patients. Surgeons must critically evaluate their technical expertise, the patient's pathoanatomy, and prognostic factors to choose the optimal intervention.
📚 Medical References
- Biceps tenodesis: indications and techniques, Oper Tech Sports Med 10:99, 2002.
- Edwards TB, Walch G: Repair of tears of the subscapularis tendon, Oper Tech Sports Med 10:86, 2002.
- Ellman H: Diagnosis and treatment of incomplete rotator cuff tears, Clin Orthop Relat Res 254:64, 1990.
- Ellman H, Hanker G, Bayer M: Repair of the rotator cuff: endresult study of factors infl uencing reconstruction, J Bone Joint Surg 68A:1136, 1986.
- Esch JC, Ozerkis LR, Helgager JA, et al: Arthroscopic subacromial decompression: results according to the degree of rotator cuff tear, Arthroscopy 4:241, 1988.
- Fealy S, April EW, Khazzam M, et al: The coracoacromial ligament: morphology and study of acromial enthesopathy, J Shoulder Elbow Surg 14:542, 2005.
- Ferrari DA: Anterior superior anatomy of the shoulder, Orthop Trans 9:42, 1985.
- Fisher L, Kurtz A, Shipley M: Association between cheiroarthropathy and frozen shoulder in patients with insulin dependent diabetes mellitus, Br J Rheumatol 25:141, 1986.
- Flatow E, Connor P, Levine W, et al: Coracoacromial arch reconstruction for anterosuperior subluxation after failed rotator cuff surgery, J Shoulder Elbow Surg 6:228, 1997.
- Flugstad D, Matsen FA, Larry I, et al: Failed acromioplasty and the treatment of the impingement syndrome. Paper presented at the Second Open Meeting of the American Shoulder and Elbow Surgeons, New Orleans, 1986.
- Fuchs B, Weishaupt D, Zanetti M, et al: Fatty degeneration of the muscles of the rotator cuff: assessment by computed tomography versus magnetic resonance imaging, J Shoulder Elbow Surg 8:599, 1999.
- Fukuda H: Shoulder impingement and rotator cuff disease, Curr Orthop 4:225, 1990.
- Fukuda H, Craig EV, Yamanaka K: Surgical treatment of incomplete thickness tears of rotator cuff: long-term follow-up, Orthop Trans 11:237, 1987.
- Fukuda H, Hamada K, Yamanaka K: Pathology and pathogenesis of bursal side rotator cuff tears: views from en bloc histologic sections, Clin Orthop Relat Res 254:75, 1990.
- Fukuda H, Mikasa M, Yamanaka K: Incomplete thickness rotator cuff tears diagnosed by subacromial bursography, Clin Orthop Relat Res 223:51, 1987.
- Galatz LM, Griggs S, Cameron BD, et al: Prospective longitudinal analysis of postoperative shoulder function: a ten-year study of full-thickness rotator cuff tears, J Bone Joint Surg 83A:1052, 2001.
- Gartsman GM: Arthroscopic assessment of rotator cuff tear reparability, Arthroscopy 12:546, 1996.
- Gartsman GM: Massive, irreparable tears of the rotator cuff: results of operative debridement and subacromial decompression, J Bone Joint Surg 79A:715, 1997.
- Gartsman GM: Arthroscopic management of rotator cuff disease, J Am Acad Orthop Surg 6:259, 1998.
- Gartsman GM, Brinker MR, Khan M, et al: Self-assessment of general health status in patients with fi ve common shoulder conditions, J Shoulder Elbow Surg 7:228, 1998.
- Gartsman GM, Hasan SS: Specialty update: what’s new in shoulder and elbow surgery, J Bone Joint Surg 87A:226, 2005.
- Gartsman GM, Khan M, Hammerman SM: Arthroscopic repair of full-thickness tears of the rotator cuff, J Bone Joint Surg 80:832, 1998.
- Gartsman GM, Taverna E: The incidence of glenohumeral joint abnormalities associated with full-thickness, reparable rotator cuff tears, Arthroscopy 13:450, 1997.
- Gartsman GM, Taverna E, Hammerman SM: Arthroscopic treatment of acute traumatic anterior glenohumeral dislocation and greater tuberosity fracture, Arthroscopy 15:648, 1999.
- Gerber C, Fuchs B, Hodler J: The results of repair of massive tears of the rotator cuff, J Bone Joint Surg 82A:505, 2000.
- Gerber C, Krushell RJ: Isolated rupture of the tendon of the subscapularis muscle: clinical features in 16 cases, J Bone Joint Surg 73B:389, 1991.
- Gerber C, Maquieira G, Espinosa N:
