STIMULATION OF FRACTURE HEALING
The successful management of complex fractures, nonunions, and critical-sized bone defects relies heavily on the surgeon’s ability to manipulate the biological and mechanical environment. The stimulation of fracture healing is fundamentally rooted in the "Diamond Concept," which emphasizes the synergistic interaction of osteogenic cells, an osteoconductive scaffold, osteoinductive growth factors, and a stable mechanical environment. When the natural physiological response is insufficient due to trauma, infection, or host factors, exogenous stimulation via bone grafting or biological substitutes becomes mandatory.
BONE GRAFTING: THE BIOLOGICAL TRIAD
Autologous Bone Grafts
Autologous bone grafting (autograft) remains the undisputed gold standard in orthopedic reconstructive surgery. Its superiority stems from its unique ability to provide the complete biological triad required for bone regeneration:
- Osteoconduction: The graft provides a three-dimensional structural scaffold (trabecular network) that facilitates the ingrowth of host capillaries, perivascular tissue, and osteoprogenitor cells—a process known as creeping substitution.
- Osteoinduction: The graft contains matrix-bound bone morphogenetic proteins (BMPs) and other growth factors that actively recruit host mesenchymal stem cells (MSCs) and induce their differentiation into bone-forming osteoblasts.
- Cellular Osteogenesis: Autografts transfer viable, primitive osteocytes and osteoprogenitor cells directly to the recipient site, which can immediately begin synthesizing new osteoid.
Sources of Autologous Bone
Autologous grafts can be harvested from multiple anatomical sites, dictated by the volume required and the structural needs of the recipient bed.
Local Bone Harvest
Local bone removed during the approach or at the time of arthrodesis is an excellent, zero-morbidity source of graft material. To optimize its biological potential, all adherent soft tissue must be meticulously debrided. The bone is then morcelized into smaller fragments using a rongeur or a specialized bone mill. Finely milling the bone exponentially increases the surface area, thereby maximizing the release of osteoinductive proteins and the availability of live cells.
Iliac Crest Bone Graft (ICBG)
The iliac crest is the second most common and historically the most reliable area for autograft harvest.
* Posterior Iliac Crest: Offers a significantly larger volume of cancellous bone (up to 50–80 cc) compared to the anterior crest. It can be harvested as morcelized cancellous bone or as a structural tricortical graft.
* Anterior Iliac Crest: Typically yields smaller volumes (20–30 cc) and is often utilized for smaller defects or upper extremity surgery.
Surgical Pitfall: Bone harvest from the iliac crest is notoriously prone to donor-site complications. Morbidity includes chronic donor-site pain (reported in up to 30% of patients), injury to the lateral femoral cutaneous nerve (anterior) or cluneal nerves (posterior), hematoma, cosmetic deformity, pelvic fractures, and gait disturbances.
THE REAMER-IRRIGATOR-ASPIRATOR (RIA) SYSTEM
To mitigate the morbidity of iliac crest harvest while obtaining massive volumes of highly osteogenic material, the Reamer-Irrigator-Aspirator (RIA) system was developed. Originally designed to reduce intramedullary pressure and thermal necrosis during femoral nailing, the RIA has evolved into a premier technique for harvesting autologous bone graft.
The RIA system simultaneously reams the intramedullary canal, irrigates the site to reduce thermal injury, and aspirates the marrow and bone debris into a specialized filter. The resulting effluent is a rich amalgamation of morcelized autologous bone, mesenchymal stem cells, and potent bone growth factors.
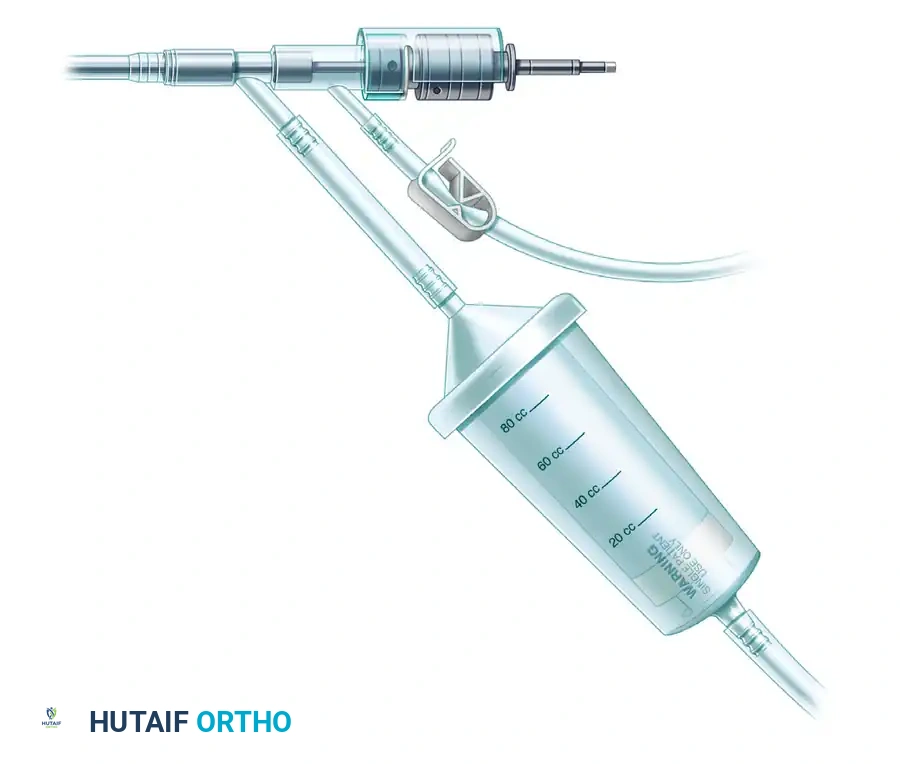
FIGURE 53-16: Reamer, irrigator, aspirator (RIA) for obtaining marrow during femoral reaming; the aspirated marrow can be used for bone grafting. Note the drive shaft, aspiration tube connected to a suction canister, and the 100 cc capacity graft filter mesh sized to capture morcelized bone and marrow.
RIA Surgical Technique and Considerations
When utilizing the RIA for spinal fusions or distant nonunions, the surgeon must carefully plan patient positioning. Harvesting from the femur may require obtaining the graft prior to the primary procedure, necessitating a separate setup, positioning (often supine for the femur, then prone for the spine), and draping sequence.
Complications and Mitigation Strategies for RIA
While the RIA provides excellent graft volume (often exceeding 60–80 cc), the technique is not without significant risks. Iatrogenic fractures of the donor femur have been reported, sometimes necessitating immediate intramedullary fixation. Furthermore, eccentric reaming can lead to cortical perforation. Significant, sometimes life-threatening, blood loss from continuous marrow aspiration is a major concern.
To avoid or minimize these complications, the following strict, evidence-based protocols must be adhered to:
- Preoperative Templating: Preoperative orthogonal radiographs of the donor bone must be meticulously evaluated for pre-existing deformity. The isthmus must be measured to determine the safe limits of reamer diameter.
- Blood Management: Cross-matched blood must be available in the operating room to replace aspirated blood and marrow elements.
- Suction Control: The aspirator must be strictly turned off when active reaming is not being performed to avoid unnecessary and rapid blood loss.
- Intraoperative Assessment: The donor bone should be carefully evaluated fluoroscopically after reaming to check for eccentric thinning or perforations. A prophylactic intramedullary nail should be immediately available in the room if a perforation or impending fracture is detected.
- Postoperative Rehabilitation: Postoperative ambulation must be protected (e.g., restricted weight-bearing with crutches or a walker) to prevent delayed donor bone fracture.
- Hemodynamic Monitoring: The patient’s hematocrit and hemoglobin levels must be checked at the conclusion of the procedure and monitored serially over the next 24 hours to detect delayed or significant blood loss.
- Patient Selection: Patients with known metabolic bone diseases, such as severe osteoporosis or even localized osteopenia of the donor bone, are poor candidates for RIA harvest due to the unacceptably high risk of iatrogenic fracture.
HOST CLASSIFICATION FOR FRACTURE HEALING
The success of any bone grafting procedure is intimately tied to the physiological status of the host. A compromised host will struggle to incorporate even the most robust autograft. The following classification system is critical for determining the risk of complications in open fractures and nonunion surgery.
Box 53-5: Host Classification for Determining Risk of Complications
Type A Host (Normal Physiology)
* No systemic illness (e.g., no insulin-dependent diabetes mellitus, rheumatoid arthritis, or active infection).
* No immunosuppression (neither pharmacological nor disease-mediated).
* No substance abuse (tobacco, alcohol, illicit drugs).
* Stable socioeconomic status (adequate nutrition, housing, and postoperative assistance).
* Normal neuropsychiatric history.
Type B Host (Compromised Physiology)
* One systemic illness that is medically controlled (e.g., controlled diabetes mellitus, long-term anticoagulant or antiplatelet therapy).
* No immunosuppression.
* Isolated tobacco or recreational drug use.
* Impaired socioeconomic status (lack of adequate nutrition, unstable housing, or lack of assistance).
* History of neuropsychiatric impairment that is currently treated and stable.
Type C Host (Severely Compromised Physiology)
* Uncontrolled or multiple systemic illnesses.
* Active immunosuppression (pharmacological or disease-mediated).
* Polysubstance abuse.
* Unstable economic status (malnourished, homeless, lacking any social assistance).
* History of neuropsychiatric impairment that remains untreated.
* Note: A combination of multiple Type B factors often functionally downgrades a patient to a Type C host.
Clinical Pearl: In Type C hosts, the biological environment is so hostile that standard autografting may fail. These patients often require aggressive optimization of their medical status prior to surgery, and may benefit from the addition of potent osteoinductive factors (like recombinant human BMP-2) to overcome their intrinsic biological deficits.
BONE GRAFT SUBSTITUTES
Although autogenous material remains the gold standard for filling bone defects caused by high-energy trauma, chronic osteomyelitis, tumor resection, or complex revision surgery, its limitations are well-documented. Autograft harvest increases the morbidity of the surgical procedure, prolongs anesthesia time, increases blood loss, and frequently causes significant postoperative donor-site complications (e.g., chronic pain, cosmetic defects, fatigue fractures, and heterotopic ossification). Furthermore, the absolute volume of autogenous bone available is finite.
Because of these inherent limitations, a vast array of bone graft substitutes has been developed to act as bone void fillers or autograft extenders.
The Laurencin Classification System
A comprehensive bone graft substitute classification system proposed by Laurencin et al. divides these materials into five major categories based on their primary composition:
- Allograft-Based: Utilizes cadaveric bone with or without other elements.
- Factor-Based: Includes natural and recombinant growth factors (e.g., rhBMP-2, rhBMP-7).
- Cell-Based: Utilizes expanded mesenchymal stem cells or osteoprogenitors to produce new bone.
- Ceramic-Based: Uses various synthetic ceramics (e.g., calcium phosphate, calcium sulfate, hydroxyapatite) as an osteoconductive scaffold.
- Polymer-Based: Uses biodegradable polymers alone or combined with other materials.
- Miscellaneous Group: Includes tissue derived from marine sources, such as coral (coralline hydroxyapatite) and sponge skeletons.
Allograft-Based Bone Graft Substitutes
Allograft bone is harvested from human cadaveric donors and processed extensively to remove immunogenic cellular material while preserving the extracellular matrix. It serves primarily as an osteoconductive scaffold. Allograft is available in numerous forms and is prepared via several distinct methods, including freeze-drying, irradiation (electron beam and gamma ray), and decalcification.
Structural vs. Morcelized Allograft
Freeze-dried and irradiated forms of cortical allograft provide excellent initial mechanical strength and are frequently used for structural support (e.g., intercalary segment reconstruction in tumor surgery). Conversely, cancellous allograft can be milled for special applications, such as packing into intervertebral cages, or morcelized to serve as a "bone extender" when mixed with autograft.
Demineralized Bone Matrix (DBM)
Demineralized bone matrix is the decalcified form of cortical allograft. The acid-extraction process removes the mineral phase of the bone, thereby exposing the matrix-bound osteoinductive proteins (primarily BMPs) that stimulate bone formation.
- Biological Profile: DBM is highly osteoconductive and moderately osteoinductive. However, because all viable cells are destroyed during processing, DBM possesses zero osteogenic capacity.
- Formulations: DBM is commercially supplied as a putty, injectable gel, paste, powder, strips, or a mixture of these. To enhance its handling characteristics, demineralized allograft is usually mixed with a carrier substance such as glycerol, calcium sulfate powder, sodium hyaluronate, or gelatin.
- Augmentation: To overcome its lack of osteogenesis, DBM is frequently mixed with the patient's own bone marrow aspirate (BMA), which introduces osteogenic pluripotent mesenchymal stem cells into the osteoinductive/osteoconductive DBM scaffold.
Surgical Warning: There is considerable variability in the osteoinductive potential between different commercially available DBM products. This variability is multifactorial, stemming from the donor source (age and health of the cadaver), the specific bone bank processing procedures, the physical form of the DBM, and the type of carrier used.
Sterilization and Disease Transmission
The primary concern with any allograft product is the risk of viral or bacterial disease transmission (e.g., HIV, Hepatitis C). To mitigate this, allografts undergo rigorous sterilization processes. While sterilization of DBM by gamma irradiation and ethylene oxide exposure drastically decreases the risk of disease transmission, these harsh processes can denature proteins and significantly decrease the inherent osteoinductive activity of the final graft material. Surgeons must balance the need for absolute sterility with the biological efficacy of the chosen substitute.
📚 Medical References
- bone grafting, Orthop Clin North Am 18:213, 1987.
- Lawrence TK, Garfin SR, Booth RE: Harvesting autogenous iliac bone grafts: a review of complications and techniques, Spine 14:1324, 1989.
- Lotem M, Maor P, Haimoff H, et al: Lumbar hernia at an iliac bone graft donor site: a case report, Clin Orthop Relat Res 80:130, 1971.
- Lundborg G, Rydevik B: The tourniquet in extremity surgery: how can complications be avoided? Acta Orthop Scand 54:669, 1983.
- Meeder PJ, Eggers C: Techniques for obtaining autogenous bone graft, Injury 1(suppl):5, 1994.
- Meyer DC, Jacob HAC, Pistoia W, et al: The use of acrylic bone cement for suture anchoring, Clin Orthop Relat Res 410:295, 2003.
- Wientroub S, Goodwin D, Khermosh O, et al: The clinical use of autologous marrow to improve osteogenic potential of bone grafts in pediatric orthopaedics, J Pediatr Orthop 9:186, 1989.
- Wolfe SA, Kawamoto HK: Taking the iliac-bone graft: a new technique, J Bone Joint Surg 60A:411, 1978.
- Younger ASE, McEwen JA, Inkpen K: Wide contoured cuffs and automated limb occlusion measurement allow lower tourniquet pressures, Clin Orthop Relat Res 428:286, 2004.
- [Surgical Approaches
General Banks SW, Laufman H: An atlas of surgical exposures of the extremities, Philadelphia, 1953, WB Saunders. Borges AF: Zigzag incisions for improved posture and scarring, Clin Orthop Relat Res 145:202, 1979.](https://pubmed.ncbi.nlm.nih.gov/?term=Surgical%20Approaches%0A%0AGeneral%20Banks%20SW%2C%20Laufman%20H%3A%20An%20atlas%20of%20surgical%20exposures%20of%20the%20extremities%2C%20Philadelphia%2C%201953%2C%20WB%20Saunders.%20Borges%20AF%3A%20Zigzag%20incisions%20for%20improved%20posture%20and%20scarring%2C%20Clin%20Orthop%20Relat%20Res%20145%3A202%2C%201979.)
- Henry AK: Extensile exposure, 2nd ed, Edinburgh, 1966, E & S Livingstone. Hoppenfeld S, deBoer P: Surgical exposures in orthopaedics: the anatomic approach , Philadelphia, 2003, Lippincott Williams & Wilkins. Kocher T: Textbook of operative surgery, 3rd ed, London, 1911, Adam & Charles Black (Translated by HJ Stiles, CB Paul). Kocher T: Chirurgische operationslehre, 5th ed, Edinburgh, 1911, Adam & Charles Black (Jena, Gustav Fischer; Translated by HJ Stiles). Stookey B: Surgical and mechanical treatment of peripheral nerves, Philadelphia, 1922, WB Saunders. Thompson JE: Anatomical methods of approach in operations on the long bones of the extremities, Ann Surg 68:309, 1918.
- [Calcaneus Kocher T: Textbook of operative surgery, 3rd ed, London, 1911, Adam & Charles Black (Translated by HJ Stiles, CB Paul).
Tarsus and Ankle Broomhead R: Discussion on fractures in the region of the ankle joint, Proc R Soc Med 25:1082, 1932.](https://pubmed.ncbi.nlm.nih.gov/?term=Calcaneus%20Kocher%20T%3A%20Textbook%20of%20operative%20surgery%2C%203rd%20ed%2C%20London%2C%201911%2C%20Adam%20%26%20Charles%20Black%20%28Translated%20by%20HJ%20Stiles%2C%20CB%20Paul%29.%0A%0ATarsus%20and%20Ankle%20Broomhead%20R%3A%20Discussion%20on%20fractures%20in%20the%20region%20of%20the%20ankle%20joint%2C%20Proc%20R%20Soc%20Med%2025%3A1082%2C%201932.)
- Colonna PC, Ralston EL: Operative approaches to the ankle joint, Am J Surg 82:44, 1951.
- Gatellier J, Chastang P: Access to fractured malleolus with piece chipped off at back, J Chir 24:513, 1924.
- Koenig F, Schaefer P: Osteoplastic surgical exposure of the ankle joint. In Forty-fi rst report of progress in orthopedic surgery, p 17. (Abstracted from Z Chir 215:196, 1929.) Nicola T: Atlas of surgical approaches to bones and joints, New York, 1945, Macmillan. Ollier P: Traite des resections, Paris, 1892.
- [(Quoted in Steindler A: A textbook of operative orthopedics, New York, 1925, D Appleton.)
Tibia and Fibula Banks SW, Laufman H: An atlas of surgical exposures of the extremities, Philadelphia, 1953, WB Saunders. Harmon PH: A simplifi ed surgical approach to the posterior tibia for bone-grafting and fi bular transference, J Bone Joint Surg 27:496, 1945.](https://pubmed.ncbi.nlm.nih.gov/?term=%28Quoted%20in%20Steindler%20A%3A%20A%20textbook%20of%20operative%20orthopedics%2C%20New%20York%2C%201925%2C%20D%20Appleton.%29%0A%0ATibia%20and%20Fibula%20Banks%20SW%2C%20Laufman%20H%3A%20An%20atlas%20of%20surgical%20exposures%20of%20the%20extremities%2C%20Philadelphia%2C%201953%2C%20WB%20Saunders.%20Harmon%20PH%3A%20A%20simpli%EF%AC%81%20ed%20surgical%20approach%20to%20the%20posterior%20tibia%20for%20bone-grafting%20and%20%EF%AC%81%20bular%20transference%2C%20J%20Bone%20Joint%20Surg%2027%3A496%2C%201945.)
- Henry AK: Exposures of long bones and other surgical methods, Bristol, England, 1927, John Wright & Sons. Phemister DB: Treatment of ununited fractures by onlay bone grafts without screw or tie fixation and without breaking down of the fi brous union, J Bone Joint Surg 29:946, 1947.
- Tochigi Y, Amendola A, Muir D, et al: Surgical approach for centrolateral talar osteochondral lesions with an anterolateral osteotomy, Foot Ankle Int 23:1038, 2002.
- Michelson JD, Perry M: Clinical safety and efficacy of calf tourniquets, Foot Ankle 17:573, 1996.
- Morbidity and Mortality Weekly Report: Transmission of HIV through bone transplantation: case report and public health recommendations, JAMA 260:2487, 1988.
- Muscolo DL, Caletti E, Schajowicz F, et al: Tissue-typing in human massive allografts of frozen bone, J Bone Joint Surg 69A:583, 1987.
- Nusbickel FR, Dell PC, McAndrew MP, et al: Vascularized autografts for reconstruction of skeletal defects following lower extremity trauma: a review, Clin Orthop Relat Res 243:65, 1989.
- Parikh SN: Bone graft substitutes in modern orthopedics, Orthopedics 25:1301, 2002.
- Patterson S, Klenerman L, Biswas M, et al: The effect of pneumatic tourniquets on skeletal muscle physiology, Acta Orthop Scand 52:171, 1981.
- Pedowitz RA, Gershuni DH, Botte MJ, et al: The use of lower tourniquet infl ation pressures in extremity surgery facilitated by curved and wide tourniquets and integrated cuff infl ation system, Clin Orthop Relat Res 287:237, 1993.
- Pelker RR, Friedlaender GE: Biomechanical aspects of bone autografts and allografts, Orthop Clin North Am 18:235, 1987.
- Reid HS, Camp RA, Jacob WH: Tourniquet hemostasis: a clinical study, Clin Orthop Relat Res 177:230, 1983.
- Reid RL: Hernia through an iliac bone-graft donor site: a case report, J Bone Joint Surg 50A:757, 1968.
- Rorabeck CH: Tourniquet-induced nerve ischemia: an experimental investigation, J Trauma 20:280, 1980.
- Rorabeck CH, Kennedy JC: Tourniquet-induced nerve ischemia complicating knee ligament surgery, Am J Sports Med 8:98, 1980.
- Sapega AA, Heppenstall RB, Chance B, et al: Optimizing tourniquet application and release times in extremity surgery: a biochemical and ultrastructural study, J Bone Joint Surg 67A:303, 1985.
- Saunders KC, Louis DL, Weingarden SI, et al: Effect of tourniquet time on postoperative quadriceps function, Clin Orthop Relat Res 143:194, 1979.
- Scarborough NL: Allograft bones and soft tissues: current procedures for banking allograft human bone, Orthopedics 15:1161, 1992.
- Shaw JA, Murray DG: The relationship between tourniquet pressure and underlying soft-tissue pressure in the thigh, J Bone Joint Surg 64A:1148, 1982.
- Simon MA, Mass DP, Zarins CK, et al: The effect of a thigh tourniquet on the incidence of deep venous thrombosis after operations on the fore part of the foot, J Bone Joint Surg 64A:188, 1982.
- Simonis RB, Shirall HR, Mayou B: Free vascularized fi bular grafts for congenital pseudoarthrosis of the tibia, J Bone Joint Surg 73B:211, 1991.
- Stevenson S: The immune response to osteochondral allografts in dogs, J Bone Joint Surg 69A:573, 1987.
- Tingart MJ, Apreleva M, Lehtinen J, et al: Anchor design and bone mineral density affect the pull-out strength of suture anchors in
