Advanced Magnetic Resonance Imaging of the Spine
Magnetic Resonance Imaging (MRI) of the spine accounts for a substantial percentage of radiological examinations at tertiary orthopedic and neurosurgical centers. MRI allows for a comprehensive, noninvasive evaluation of the osseous spine, spinal canal, and the delicate neural elements, including the spinal cord and exiting nerve roots. The anatomy of the spine, cord, nerve roots, and spinal ligaments is highly complex and biomechanically dynamic. Because these crucial structures are small and can be adequately imaged only with the use of specialized surface coils, the spine is typically divided into three distinct anatomical sections for imaging purposes: cervical, thoracic, and lumbar.
Thoracic and lumbar spine studies are routinely performed utilizing conventional or phased-array surface coils, which maximize the signal-to-noise ratio. The cervical spine, given its lordotic curvature and proximity to the cranium, is best studied with a specifically contoured posterior neck coil or a flat “license plate” coil. Standard spinal examinations must include series obtained in both axial and sagittal planes. Coronal images are particularly helpful in patients with complex spinal deformity, such as adolescent idiopathic scoliosis or adult degenerative scoliosis, to evaluate the fractional curve and lateral listhesis.
There is no single "correct" imaging construct; the makeup of the study depends on multiple factors, including the type and field strength of the magnet (1.5T vs. 3.0T), the availability of specific hardware and software, and the clinical preferences of the examining surgeon. However, all diagnostic studies must produce high-resolution images capable of detecting and defining pathological conditions of the spinal cord, thecal sac, vertebral bodies, and intervertebral discs.
Clinical Pearl: While MRI is the gold standard for soft tissue and neural element evaluation, it is highly susceptible to motion artifact and hardware distortion. In patients with prior spinal instrumentation, utilizing Metal Artifact Reduction Sequences (MARS) is critical for accurate postoperative evaluation.
Intervertebral Disc Disease: Pathophysiology and Imaging
The most common indication for MRI of the spine is the evaluation of intervertebral disc disease. MRI is currently the procedure of choice for screening patients presenting with axial low back pain, cervical radiculopathy, or sciatic pain. In the lumbar and thoracic spine, MRI has largely supplanted CT myelography because it is noninvasive, lacks ionizing radiation, and is highly cost-effective.
The combination of high soft-tissue contrast and high spatial resolution allows for the ideal evaluation of the intervertebral discs, traversing and exiting nerve roots, the posterior longitudinal ligament (PLL), and the intervertebral foramen. Furthermore, MRI provides excellent delineation of the spinal cord, allowing for the rapid identification of myelomalacia or cord edema.
Biomechanics and Degenerative Cascade
The intervertebral disc is a complex biomechanical structure consisting of a gelatinous nucleus pulposus surrounded by a highly organized, collagenous annulus fibrosus. Because of its high water and proteoglycan content, a normal, healthy disc exhibits signal hyperintensity (bright signal) on T2-weighted images. The natural aging process, combined with repetitive microtrauma, results in a gradual desiccation of the disc material, loss of hydrostatic pressure, and a subsequent loss of this T2 hyperintense signal—often referred to as "black disc disease."
Disc herniations or extrusions appear as convex or polypoid masses extending posteriorly into the ventral epidural space. They frequently maintain a signal intensity similar to that of the parent disc of origin.
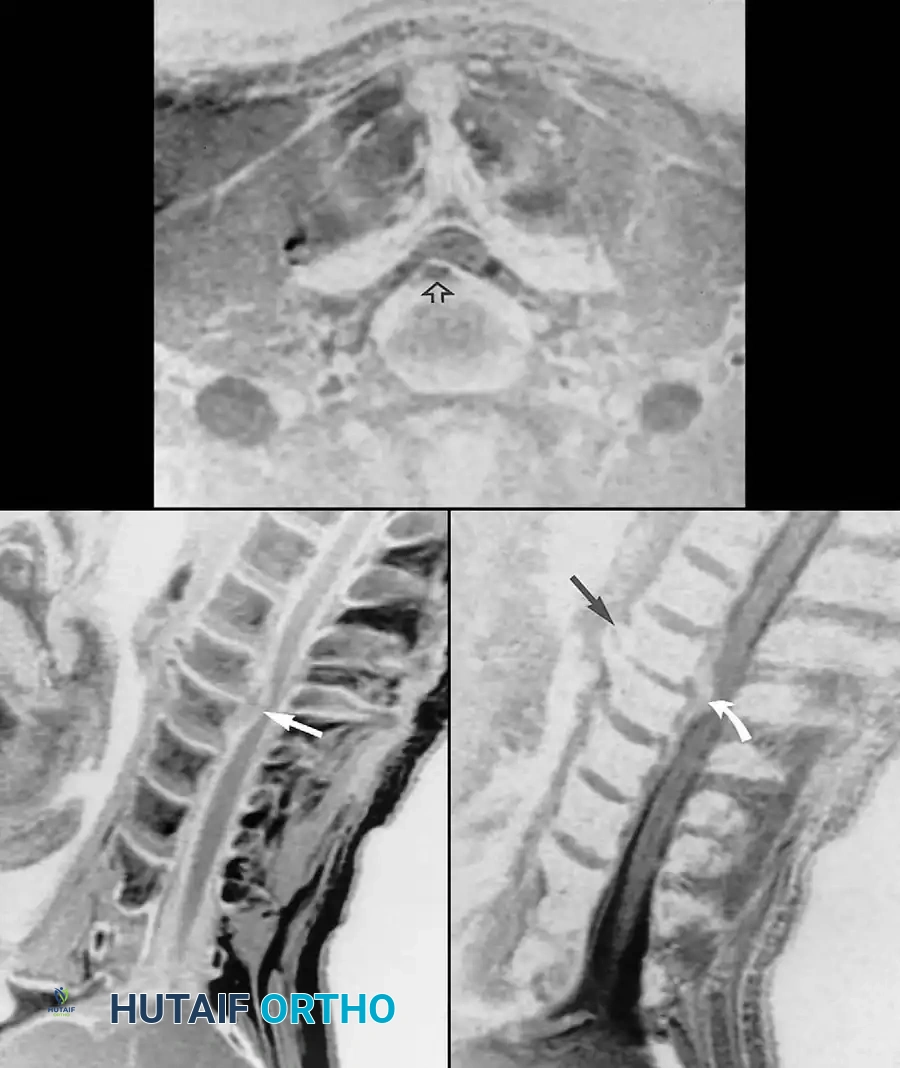
Fig. 2-25 Cervical disc extrusion (herniation). A, T1-weighted sagittal image of cervical spine reveals extruded C5-6 disc (arrow). B, Gradient-echo sagittal image produces “myelographic” effect, showing displaced disc material (black arrow) isointense to nucleus pulposus. Anterior osteophytes (white arrow) at this level are accentuated with gradient-echo technique. C, Gradient-echo axial image shows right paracentral extrusion (arrow) effacing cervical cord.
Sagittal T2-weighted or gradient-echo images create a “myelographic” effect, utilizing the hyperintense cerebrospinal fluid (CSF) to outline the neural elements, making them highly useful in evaluating compromise of the subarachnoid space. However, sagittal T1-weighted images must be examined closely to identify narrowing of the neuroforamina. The normal bright fat signal in the foramina provides excellent contrast to the darker, displaced disc material or hypertrophic facet arthropathy.
Far-lateral (extraforaminal) disc herniations are best visualized on selected axial images localized through the disc levels. Free disc fragments (sequestrations) appear discontinuous with the intervertebral disc, usually demonstrating intermediate T1-weighted signal in stark contrast to the hypointense CSF.
Standardized Nomenclature for Disc Pathology
The terminology surrounding pathological conditions of the intervertebral disc has historically been confusing. To standardize communication among radiologists and orthopedic surgeons, Jensen et al. proposed the following strict definitions:
* Bulge: A circumferential, symmetrical extension of the disc beyond the interspace around the vertebral end plates (involving >50% of the disc circumference).
* Protrusion: A focal or asymmetrical extension of the disc beyond the interspace, where the base against the disc of origin is broader than any other dimension of the protrusion.
* Extrusion: A more extreme extension of the disc beyond the interspace, where the base against the disc of origin is narrower than the diameter of the extruding material itself.
* Sequestration: Specifically refers to a disc fragment that has completely separated from the disc of origin, with no contiguous connection.
Surgical Approach: Lumbar Microdiscectomy
When conservative management fails, and MRI confirms a symptomatic disc extrusion correlating with the patient's radiculopathy, a lumbar microdiscectomy is indicated.
- Positioning: The patient is positioned prone on a radiolucent Jackson table or Wilson frame. The abdomen must hang free to decrease intra-abdominal pressure, thereby reducing epidural venous engorgement and intraoperative bleeding.
- Localization: Intraoperative fluoroscopy is utilized to localize the exact pathological disc space.
- Incision and Exposure: A 2-3 cm midline incision is made. The lumbodorsal fascia is incised, and the paraspinal muscles are subperiosteally elevated off the spinous process and lamina on the ipsilateral side.
- Microsurgical Decompression: A tubular retractor or Taylor retractor is placed. Using an operating microscope, a minimal laminotomy is performed using a high-speed burr and Kerrison rongeurs. The ligamentum flavum is excised to expose the dura and the traversing nerve root.
- Discectomy: The nerve root is gently retracted medially. The extruded disc fragment is identified and removed using pituitary rongeurs. The annulotomy site is probed to ensure no loose fragments remain within the disc space.
- Postoperative Protocol: Patients are typically mobilized on the day of surgery. Bending, lifting, and twisting are restricted for 4-6 weeks to minimize the risk of recurrent herniation.
Evaluating Postoperative Back Pain
In a patient presenting with persistent or recurrent postoperative back pain (Failed Back Surgery Syndrome), the differential diagnosis must include residual or recurrent disc herniation, epidural hematoma, epidural abscess, and postoperative discitis.
Before the advent of intravenous gadolinium contrast agents, making a definitive distinction between recurrent disc material and postoperative epidural scar tissue (fibrosis) was nearly impossible with CT myelography or non-contrast MRI. The introduction of gadolinium has revolutionized the postoperative evaluation of the spine.
Differentiating Scar Tissue from Recurrent Disc
After intravenous gadolinium administration, repeat T1-weighted images typically demonstrate robust enhancement of vascularized scar tissue or epidural fibrosis. Conversely, beyond the immediate postoperative period, avascular disc material does not enhance.
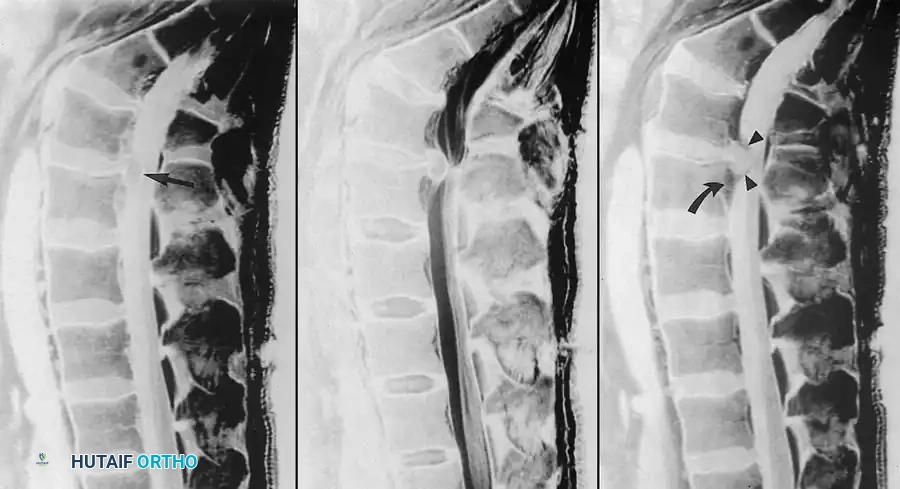
Fig. 2-26 Recurrent lumbar disc extrusion (herniation). A, Sagittal T1-weighted image shows intermediate signal intensity in L4-5 disc material (arrow) surrounded by hypointense cerebrospinal fluid. B, Sagittal T2-weighted image shows displaced disc material contiguous with intervertebral disc. Hyperintense cerebrospinal fluid provides improved contrast. C, Sagittal T1-weighted image after gadolinium administration shows enhancement of epidural venous plexus (curved arrow) and overlying granulation tissue (arrowheads), but no enhancement of disc material.
Surgical Warning: Operating on epidural fibrosis carries a high risk of incidental durotomy (dural tear) and nerve root injury. If MRI confirms that the patient's symptoms are due to scar tissue tethering the nerve root rather than a recurrent compressive disc fragment, revision decompression is generally contraindicated, and spinal cord stimulation or pain management should be considered.
Postoperative Complications: Hematoma and Infection
Epidural hematomas and abscesses appear as loculated collections within the spinal canal, typically demonstrating peripheral rim enhancement with gadolinium on T1-weighted images.
Gadolinium contrast is also indispensable in the postoperative evaluation of the spine for discitis. While reactive changes in the disc space and adjacent vertebral end plates (Modic changes) are frequently seen after uncomplicated spinal surgery, the classic MRI triad for postoperative discitis includes:
1. Vertebral body end plate enhancement.
2. Disc space enhancement.
3. Enhancement of the posterior longitudinal ligament.
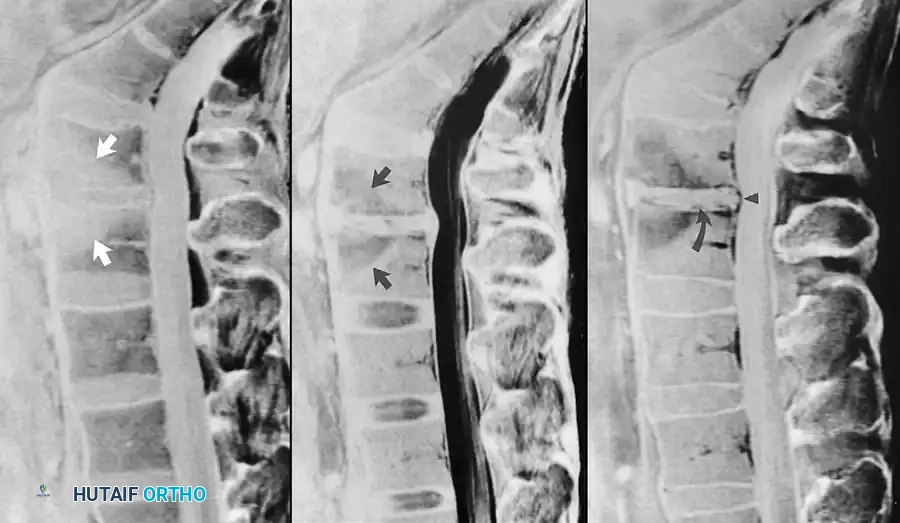
Fig. 2-27 Postoperative discitis. A, Sagittal T1-weighted image exhibits reduced marrow signal adjacent to narrowed L4-5 intervertebral disc (arrows). B, Sagittal T2-weighted image reveals corresponding hyperintense areas of vertebral marrow edema (arrows). C, After administration of gadolinium, sagittal T1-weighted image exhibits enhancement of vertebral marrow, intervertebral disc (curved arrow), and posterior longitudinal ligament (arrowhead).
Clinical correlation with inflammatory markers (Erythrocyte Sedimentation Rate [ESR] and C-Reactive Protein [CRP]), gallium or tagged white blood cell radionuclide imaging, and CT-guided percutaneous aspiration is often necessary to isolate the offending organism.
Differentiating Pyogenic vs. Granulomatous Infection
Although diagnosing a disc space infection in a surgically naive patient is generally straightforward, the MRI appearance of severe degenerative disc disease can mimic infection. While vertebral end plate edema (Modic Type 1) and mild enhancement can occur in severe degeneration, the presence of frank disc space enhancement strongly suggests infection.
Pyogenic infections (e.g., Staphylococcus aureus) and granulomatous infections (fungal or tuberculous) are frequently associated with epidural and paraspinal abscesses. In the lumbar spine, extension into the adjacent psoas muscles is best demonstrated on axial T2-weighted sequences, where hyperintense fluid and edema invade the normally hypointense musculature.
Subligamentous spread of infection across multiple vertebral levels, with relative sparing of the intervertebral discs, should immediately raise the clinical suspicion of tuberculous spondylitis (Pott's disease). Both pyogenic and tuberculous infections show abnormal enhancement with gadolinium. Abscesses, lacking central perfusion, will enhance only at their periphery.
Spinal Tumors and Marrow Pathologies
MRI has proven exceptionally valuable in the assessment of spinal neoplasms. The excellent delineation of vertebral body marrow allows for the detection of primary and metastatic disease with high sensitivity, particularly on T1-weighted sequences.
Marrow Conversion and Signal Interpretation
Normally, vertebral body marrow signal progressively increases with age on T1-weighted images. This is a reflection of the physiological conversion from hematopoietic (red) marrow to fatty (yellow) marrow. Systemic diseases such as chronic anemia or heavy smoking can result in a compensatory reconversion to a higher percentage of hematopoietic marrow, which diffusely diminishes the T1-weighted signal.
Vertebral tumor foci typically appear as discrete, focal areas of diminished T1 signal replacing the normal bright fat. As is typical with most solid tumors, these lesions become hyperintense relative to surrounding marrow on T2-weighted studies and demonstrate robust enhancement following gadolinium contrast administration.
Neoplasms that diffusely infiltrate the vertebral marrow, such as multiple myeloma or diffuse metastatic prostate carcinoma, present a diagnostic challenge because differentiating diffuse tumor infiltration from hyperplastic hematopoietic marrow is sometimes difficult. In these cases, STIR (Short Tau Inversion Recovery) sequences and diffusion-weighted imaging (DWI) can provide additional diagnostic clarity.
Surgical Considerations for Spinal Tumors
When evaluating a patient with a spinal neoplasm, the Spinal Instability Neoplastic Score (SINS) is utilized to guide surgical decision-making.
* Indications for Surgery: Intractable pain due to mechanical instability, progressive neurological deficit due to epidural spinal cord compression (ESCC), or the need for an open biopsy when percutaneous methods fail.
* Surgical Approach: Depending on the tumor's location and the patient's life expectancy, surgical options range from palliative posterior decompression and instrumented stabilization to en bloc spondylectomy for isolated primary bone tumors (e.g., chordoma, chondrosarcoma).
Spinal Trauma: Imaging Protocols and Surgical Management
While MRI provides unparalleled soft-tissue detail, non-contrast Computed Tomography (CT) remains the most useful advanced imaging technique for acute spinal trauma. The inherent contrast provided by cortical bone and the unmatched spatial resolution make CT the preferred initial examination to rapidly identify fractures, subluxations, and osseous compromise of the spinal canal.
However, MRI is indispensable in trauma patients with suspected spinal cord injury (SCI), epidural hematoma, or traumatic disc herniation. Soft-tissue injuries, particularly tears of the Posterior Ligamentous Complex (PLC)—which includes the supraspinous ligament, interspinous ligament, ligamentum flavum, and facet capsules—can be definitively identified in the acute stage. Discontinuity of these normally hypointense ligaments, accompanied by hemorrhage and edema, is clearly visualized on sagittal STIR and T2-weighted images.
Indications for MRI in Spinal Trauma
In the acute trauma setting, MRI is specifically indicated for:
1. Neurologically impaired patients whose CT examinations are negative (to evaluate for Spinal Cord Injury Without Radiographic Abnormality - SCIWORA).
2. Patients with cervical facet dislocations prior to closed reduction (if the patient is obtunded) or open reduction, to exclude an associated traumatic disc herniation that could be retropulsed into the cord during reduction maneuvers.
3. Evaluating the integrity of the PLC to determine the need for surgical stabilization in thoracolumbar burst fractures.
Differentiating Osteoporotic vs. Pathologic Fractures
The role of MRI in evaluating non-traumatic or low-energy compressed vertebrae is crucial to exclude underlying pathological conditions (e.g., metastasis).
* Benign Osteoporotic Fracture: Preservation of normal marrow signal in a portion of the compressed vertebral body, especially with a linear, band-like pattern of signal abnormality parallel to the endplate, suggests a benign osteoporotic process.
* Pathologic Fracture: Complete marrow replacement, or incomplete replacement with focal abnormal marrow signal at other non-fractured levels, should prompt immediate consideration of a CT-guided core needle biopsy. Furthermore, the presence of an irregular, asymmetrical paraspinal soft-tissue mass or a convex bulging of the dorsal vertebral cortex into the spinal canal strongly suggests an underlying neoplasm.
