GENERAL PRINCIPLES AND RESULTS OF PERIPHERAL NERVE OPERATION
The precise determination of outcomes following peripheral nerve procedures, such as neurolysis and partial neurorrhaphy, remains one of the most complex challenges in operative orthopaedics and microsurgery. Historically and clinically, it is a well-established axiom that primary neurorrhaphy is almost never followed by a complete, 100% return of both motor and sensory function. The biological limitations of Wallerian degeneration, axonal sprouting, and end-organ atrophy dictate that some degree of permanent deficit is inevitable.
Rarely, a near-full return of function is approached after the suture of the radial nerve—largely due to its predominantly motor composition and the relatively straightforward biomechanical actions of the extensor musculature it innervates. Occasionally, excellent functional recovery is also observed after the suture of the median nerve in pediatric populations, owing to the robust neuroplasticity and shorter axonal regeneration distances inherent to children.
A useful degree of recovery often occurs when the physiological and surgical factors that influence regeneration are favorable. These factors include the patient's age, the mechanism of injury (sharp transection versus high-energy crush), the delay between injury and repair, and the surgical technique employed.
Clinical Pearl: The degree of recovery varies significantly from nerve to nerve and is heavily dependent on the relative extent of damage to the motor and sensory components within each mixed nerve. Pure motor nerves (e.g., the spinal accessory nerve) or pure sensory nerves generally exhibit superior recovery profiles compared to mixed nerves, as the risk of axonal misdirection (sensory fibers growing into motor endplates, or vice versa) is eliminated.
It is critical for the operating surgeon to recognize that the recovery of function of the limb as a whole is not strictly proportionate to isolated neurological recovery. A patient may recover fairly good neurological function (e.g., M4 motor strength and S3+ sensory recovery), but because of other concomitant defects in the limb—such as joint contractures, tendon adhesions, or ischemic muscle fibrosis—the overall functional recovery may remain unsatisfactory. Therefore, comprehensive rehabilitation and the prevention of secondary musculoskeletal contractures are as vital as the microsurgical repair itself.
SURGICAL ANATOMY OF THE CERVICAL PLEXUS
A profound understanding of the cervical plexus is mandatory for surgeons operating in the anterior and posterior triangles of the neck. The cervical plexus is formed by the union of the anterior primary rami of the first four cervical nerves (C1, C2, C3, and C4). It lies deep to the internal jugular vein and the sternocleidomastoid (SCM) muscle, resting upon the levator scapulae and middle scalene muscles.
Sensory Branches
The sensory fibers from the upper two or three cervical segments emerge around the posterior border of the SCM (the punctum nervosum or Erb's point) and course through several distinct nerves:
* Lesser Occipital Nerve (C2): Supplies the skin of the neck and scalp posterosuperior to the auricle.
* Greater Auricular Nerve (C2, C3): Ascends across the SCM to supply the skin over the parotid gland, the mastoid process, and the auricle.
* Transverse Cervical Nerve / Anterior Cutaneous Nerve of the Neck (C2, C3): Passes anteriorly to supply the skin of the anterior cervical triangle.
* Supraclavicular Nerves (C3, C4): Sensory fibers from the lower two segments of the plexus course inferiorly to supply the skin over the clavicle, the upper thorax, and the deltoid region.
Motor Branches
The muscular branches of the cervical plexus are critical for the function of the anterior neck and respiratory mechanics:
* Ansa Hypoglossi (Ansa Cervicalis): Muscular branches from C1, C2, and C3 join to form this loop, which innervates the infrahyoid "strap" muscles: the thyrohyoid, geniohyoid, omohyoid, sternothyroid, and sternohyoid muscles.
* Phrenic Nerve: Branches from C3, C4, and C5 unite to form the phrenic nerve, the sole motor supply to the diaphragm. Iatrogenic injury to this nerve during deep cervical dissections results in ipsilateral diaphragmatic paralysis.
THE SPINAL ACCESSORY NERVE (CRANIAL NERVE XI)
Anatomical Course
The spinal accessory nerve possesses a unique and complex anatomical trajectory. Fibers arising from the lateral aspect of the anterior horns of the upper five cervical segments (C1-C5) unite to form the spinal root of the accessory nerve. This root ascends into the cranial cavity through the foramen magnum.
Within the cranium, it is joined by its cranial part, which consists primarily of rootlets destined to pass with the vagus nerve (CN X). These cranial rootlets diverge from the spinal accessory nerve immediately after its exit from the skull via the jugular foramen, thereafter coursing with the vagus fibers.
The remaining spinal accessory nerve descends in the neck beneath the posterior belly of the digastric muscle. It receives communicating branches from the anterior primary rami of C2, C3, and C4, and provides motor branches to innervate the sternocleidomastoid muscle. It then exits the posterior aspect of the SCM and descends obliquely across the posterior cervical triangle to innervate the superior third of the trapezius muscle.
Mechanisms of Injury and Clinical Presentation
The spinal accessory nerve may be injured at any point along its course. However, because of its highly superficial location within the posterior cervical triangle (between the SCM and the trapezius), it is exceptionally susceptible to damage.
* Penetrating Trauma: Lacerations to the lateral neck frequently sever the nerve.
* Iatrogenic Injury: The nerve is at high risk during surgical operations in the posterior triangle, most notably during lymph node biopsies, excision of benign masses, or radical neck dissections.
Injury results in profound dysfunction of the shoulder girdle. Patients present with a drooping shoulder, weakness in forward elevation and abduction above 90 degrees, and a characteristic lateral winging of the scapula (which must be differentiated from the medial winging seen in long thoracic nerve palsy).
SURGICAL ANATOMY OF THE BRACHIAL PLEXUS
The brachial plexus is a complex neural network that supplies motor and sensory innervation to the upper extremity. It is formed by the anterior primary rami of C5, C6, C7, C8, and T1.
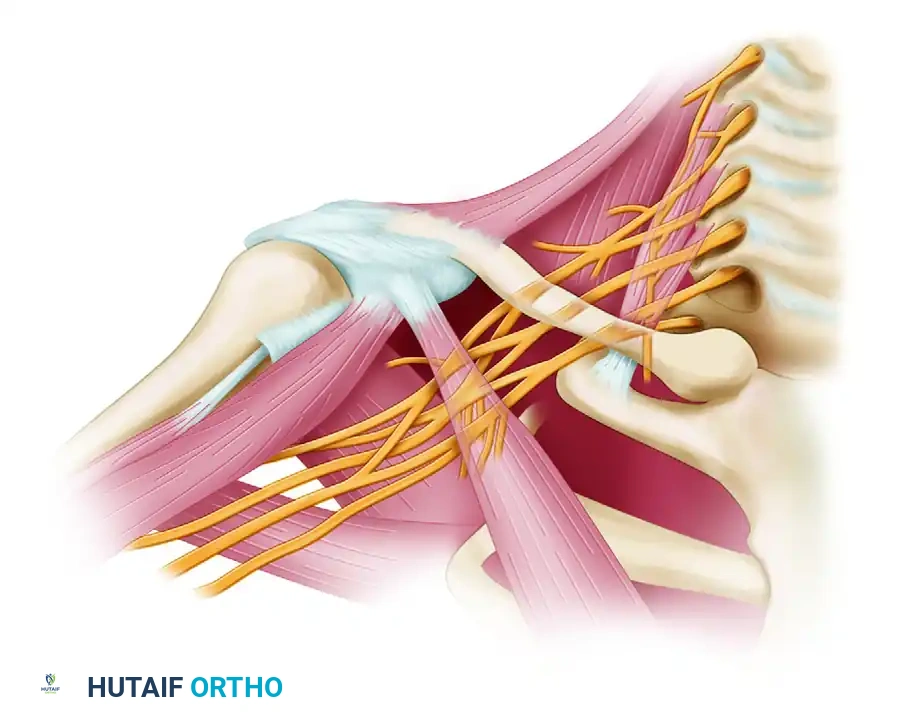
Roots, Trunks, and Divisions
The roots emerge from the intervertebral foramina between the anterior and middle scalene muscles. They quickly unite to form three trunks:
1. Upper Trunk: Formed by the union of C5 and C6.
2. Middle Trunk: A direct continuation of C7.
3. Lower Trunk: Formed by the union of C8 and T1.
These three trunks proceed inferolaterally behind the clavicle. As they pass the clavicle, each trunk divides into anterior and posterior divisions, representing the separation of innervation to the flexor (anterior) and extensor (posterior) compartments of the upper limb.
Cords and Terminal Branches
The divisions unite to form three cords, which are named based on their anatomical relationship to the axillary artery:
* Posterior Cord: Formed by the union of all three posterior divisions. It gives rise to the radial and axillary nerves.
* Lateral Cord: Formed by the union of the anterior divisions of the upper and middle trunks. It gives rise to the musculocutaneous nerve and the lateral root of the median nerve.
* Medial Cord: Formed by the continuation of the anterior division of the lower trunk. It gives rise to the ulnar nerve and the medial root of the median nerve.
Surgically Important Supraclavicular Branches
Several surgically significant nerves arise directly from the roots and trunks of the brachial plexus, proximal to the divisions:
- Long Thoracic Nerve (C5, C6, C7): Arises immediately after the roots emerge from the intervertebral foramina. It traverses the neck posterior to the brachial plexus, continues distally along the lateral aspect of the thoracic wall, and innervates the serratus anterior muscle. Injury results in classic medial scapular winging.
- Dorsal Scapular Nerve (C5): Arises from the C5 root just lateral to its contribution to the long thoracic nerve. It also traverses the neck posterior to the brachial plexus. As it courses to the medial border of the scapula, it innervates the levator scapulae, the rhomboideus major, and the rhomboideus minor.
- Suprascapular Nerve (C5, C6): This is the only surgically significant nerve to arise from a trunk. It originates from the lateral aspect of the upper trunk well superior to the clavicle. This is typically the first important branch identified when the plexus is explored superior to the clavicle. The nerve proceeds distally, passing through the suprascapular notch beneath the superior transverse scapular ligament to the posterior aspect of the scapula. Here, it supplies the supraspinatus muscle, and after proceeding around the lateral border of the scapular spine (spinoglenoid notch), it supplies the infraspinatus muscle.
Surgical Warning: No branches arise from the divisions of the brachial plexus. Any branch identified during supraclavicular exploration must originate from either a root, a trunk, or a cord.
OPERATIVE TECHNIQUES: NEURORRHAPHY AND NERVE GRAFTING
When addressing peripheral nerve transections, the ultimate goal is a tension-free, well-vascularized coaptation of healthy nerve fascicles.
The Fallacy of Extreme Positioning
Historically, surgeons utilized extreme joint positioning (e.g., severe cervical flexion or shoulder adduction) combined with rigid postoperative casting to allow the suturing of a shortened nerve without immediate tension. This practice should be strictly avoided.
While extreme positioning may allow primary coaptation in the operating room, the subsequent mobilization of the joint inevitably places massive traction forces across the repair site. This leads to intraneural ischemia, extensive scar formation, and ultimate failure of axonal regeneration.
Interfascicular Nerve Grafting
If a primary line of suture cannot be achieved without tension in a neutral anatomical position, interfascicular nerve grafting is the mandatory and highly satisfactory alternative.
1. Preparation: The proximal and distal nerve stumps are resected back to healthy, unscarred fascicular tissue (visualized under the operating microscope).
2. Harvesting: Autologous nerve grafts (most commonly the sural nerve) are harvested.
3. Coaptation: The grafts are reversed (to prevent axonal escape through branching points) and interposed between the stumps. They are secured using 8-0 or 9-0 non-absorbable monofilament epineurial sutures, often supplemented with fibrin glue.
POSTOPERATIVE PROTOCOLS AND REHABILITATION
The postoperative management of nerve repairs requires a delicate balance between protecting the microvascular repair and preventing joint stiffness.
If the line of suture is confirmed to be completely under no tension, the shoulder and neck should be immobilized in a Velpeau bandage or a customized orthosis for 3 to 4 weeks.
* Phase I (0 to 3-4 Weeks): Strict immobilization to allow the initial fibrin clot and early collagen cross-linking to stabilize the coaptation site.
* Phase II (3 to 6 Weeks): The Velpeau bandage is discontinued. Gentle, progressive active and active-assisted range-of-motion (ROM) exercises are initiated. Passive stretching that places direct tension on the nerve tract is strictly prohibited.
* Phase III (6 to 8 Weeks): Normal daily activities are gradually resumed. Progressive resistance exercises are introduced as motor reinnervation becomes clinically apparent (often months later, depending on the distance from the repair to the motor endplate).
RESULTS OF SUTURE OF THE SPINAL ACCESSORY NERVE
While large-scale, statistically significant randomized controlled trials on the results of suturing the spinal accessory nerve are scarce, extensive clinical experience provides clear prognostic guidelines.
It is universally accepted that neurolysis (for continuity lesions with scar tethering) or primary repair/grafting (for transections) can dramatically relieve neuropathic pain and restore shoulder mechanics.
Prognostic Pearl: Excellent functional results may be expected following the repair of the spinal accessory nerve. Because it is a purely motor nerve, the regenerating axons are not confounded by the presence of sensory pathways. If the repair is performed meticulously and without tension, the axons will reliably navigate to the motor endplates of the trapezius.
Early exploration (within 3 to 6 months of injury) is highly recommended if spontaneous clinical and electromyographic (EMG) recovery is not observed. Delayed repairs (beyond 12-18 months) suffer from irreversible motor endplate degradation and muscle atrophy, necessitating salvage procedures such as the Eden-Lange muscle transfer rather than primary nerve repair.
