Patient Presentation & History
We present the case of a male newborn, delivered at 40 weeks gestation, weighing 4.2 kg, a macrosomic infant. The pregnancy was complicated by gestational diabetes mellitus requiring dietary control. Labor was protracted, lasting over 18 hours, culminating in a difficult vaginal delivery with a documented shoulder dystocia requiring McRoberts maneuver and suprapubic pressure. Apgar scores were 8 and 9 at 1 and 5 minutes, respectively.
Immediately post-delivery, the neonatal team noted flaccidity of the right upper extremity, with an absent Moro reflex on the affected side. The arm was held in an adducted, internally rotated, and pronated position. There was no gross deformity or skin discoloration noted. There was no history of maternal fever, trauma during pregnancy, or other known congenital anomalies. Initial pediatrician assessment diagnosed a right brachial plexus injury and referred the patient to our orthopedic trauma service for further evaluation.
Clinical Examination
Upon initial assessment at 24 hours of life, the patient was alert and crying, demonstrating appropriate systemic responses.
Inspection
The right upper extremity maintained a characteristic "waiter's tip" posture: adducted at the shoulder, internally rotated, elbow extended, forearm pronated, and wrist flexed. There was no obvious swelling, ecchymosis, or crepitus around the clavicle or humerus. Skin integrity was intact, with no signs of laceration or abrasion. The palmar crease was normal, and the hand appeared proportional. The left upper extremity and lower extremities demonstrated full spontaneous movement.
Palpation
Gentle palpation of the right clavicle, humerus, and shoulder joint revealed no tenderness, crepitus, or focal swelling, effectively ruling out a gross clavicular or humeral fracture as the primary injury source. No palpable masses were identified in the supraclavicular fossa.
Range of Motion
Passive range of motion (ROM) was assessed and compared to the contralateral limb:
*
Shoulder:
Full passive ROM in all planes (flexion, extension, abduction, adduction, internal and external rotation). No mechanical block or apprehension was noted.
*
Elbow:
Full passive flexion and extension.
*
Forearm:
Full passive pronation and supination.
*
Wrist:
Full passive flexion and extension.
*
Digits:
Full passive flexion and extension of all metacarpophalangeal and interphalangeal joints.
Neurological Assessment
A detailed neurological examination, adapted for a newborn, was performed, focusing on motor function, reflexes, and observation for autonomic involvement. The Active Movement Scale (AMS) was utilized, though baseline function in a newborn is challenging to quantify definitively without a comparator.
-
Motor Function (Right Upper Extremity):
- Shoulder Abduction/External Rotation (C5-C6): Absent spontaneous movement. No anti-gravity movement against gravity.
- Elbow Flexion (C5-C6): Absent spontaneous elbow flexion. The biceps muscle was flaccid.
- Elbow Extension (C7-C8): Present, suggesting relative sparing of the triceps.
- Wrist Extension (C6-C7): Absent spontaneous movement.
- Wrist Flexion (C7-T1): Present, with some spontaneous movement.
- Finger Flexion/Extension (C7-T1): Full spontaneous movement of fingers and thumb. Grip reflex present, though weak compared to the contralateral side.
- Intrinsic Hand Muscles (T1): Appears intact with spontaneous finger splaying and adduction.
-
Reflexes:
- Moro Reflex: Asymmetrical. Absent on the right, present on the left.
- Biceps Reflex (C5-C6): Absent on the right, present on the left.
- Triceps Reflex (C7-C8): Present on both sides.
- Grasp Reflex (C7-T1): Present on the right, but weaker than the left.
- Sensory Examination: Challenging in a newborn. No withdrawal to noxious stimuli (pinprick) in the lateral upper arm (C5-C6 dermatome). Intact withdrawal reflex in the hand (C8-T1 dermatome).
- Autonomic (Horner's Syndrome): No ptosis, miosis, or anhidrosis was observed, indicating a low likelihood of C8-T1 preganglionic involvement with sympathetic chain disruption.
Vascular Assessment
Distal radial and ulnar pulses were palpable and symmetrical. Capillary refill was brisk (<2 seconds) in all digits of the right hand. No color or temperature discrepancies were noted.
Associated Injuries
A comprehensive examination to rule out other birth-related injuries was performed. Facial nerve palsy was absent. Abdominal examination did not reveal any distension or organomegaly. Respiratory assessment found clear breath sounds bilaterally, with no paradoxical abdominal breathing, suggesting an intact phrenic nerve.
Overall, the clinical picture was consistent with a right upper trunk brachial plexus birth palsy, predominantly involving C5 and C6 nerve roots (Erb-Duchenne palsy).
Imaging & Diagnostics
Radiographs
Initial radiographs were obtained to rule out bony injuries commonly associated with brachial plexus palsy.
*
Right Shoulder and Humerus X-rays (AP and Lateral views):
Demonstrated no evidence of clavicular fracture, humeral shaft fracture, or proximal humerus physeal injury. The glenohumeral joint appeared anatomically reduced.
*
Chest X-ray (AP view):
Excluded rib fractures and demonstrated a normal diaphragm contour, ruling out phrenic nerve palsy (C3-C5 involvement).
Magnetic Resonance Imaging (MRI)
An MRI of the cervical spine and brachial plexus was performed at 3 weeks of age, following the initial period of observation for spontaneous recovery. This timing allows for resolution of edema and early fibrotic changes, enhancing diagnostic accuracy for nerve root integrity.
Indications for MRI:
* To visualize the integrity of the brachial plexus roots, trunks, and divisions.
* To differentiate preganglionic (root avulsion from spinal cord) from postganglionic injuries (rupture or neuroma in continuity).
* To identify pseudomeningoceles, which are highly suggestive of root avulsions.
* To guide surgical planning by mapping the exact level and nature of the injury.
MRI Findings (Cervical Spine and Brachial Plexus, T1 and T2 weighted sequences with fat suppression):
*
Sagittal and Axial T2-weighted images:
Demonstrated T2 hyperintensity and thickening of the right C5 and C6 nerve roots in the supraclavicular fossa, consistent with significant stretch injury and edema, likely representing neuromas-in-continuity or ruptures.
*
No clear evidence of pseudomeningoceles
in the cervical spinal canal, which reduces the likelihood of complete preganglionic C5/C6 avulsion, but does not definitively rule it out.
* The C7, C8, and T1 nerve roots appeared to be structurally intact, with normal signal intensity and caliber.
* The overall architecture of the cervical spinal cord and vertebral bodies was normal.
* No significant paraspinal hematoma or other soft tissue abnormalities were observed.
Interpretation: The MRI findings suggested a severe postganglionic injury to the C5 and C6 nerve roots, either a complete rupture or a neuroma-in-continuity, rather than a definitive preganglionic avulsion. This information is crucial for determining the potential for nerve repair (grafting) versus nerve transfer.
Figure 1: Representative MRI image (T2-weighted sagittal view) of a brachial plexus, illustrating typical findings such as T2 hyperintensity and thickening indicative of neuroma-in-continuity (not specific to this patient, but illustrative of a common finding in BPBP).
Electrophysiological Studies (EMG/NCS)
Nerve Conduction Studies (NCS) and Electromyography (EMG) were deferred at 3 weeks given the young age of the patient and the clarity provided by MRI. Typically, these studies are more informative at 6-8 weeks when Wallerian degeneration is complete, to confirm the extent of axonal loss and reinnervation. However, given the strong clinical picture of severe C5-C6 palsy and concerning MRI findings, the decision was made to proceed with surgical planning without waiting for full electrophysiological maturation, to avoid missing the critical window for nerve repair.
Differential Diagnosis
A thorough differential diagnosis is crucial in a newborn presenting with arm paralysis. The following table outlines key considerations:
| Condition | Key Features | Diagnostic Tests | Differentiating Factors |
|---|---|---|---|
| Brachial Plexus Birth Palsy (BPBP) | Unilateral arm paralysis (flaccidity), often "waiter's tip" posture. Absent Moro reflex on affected side. Intact clavicle/humerus. Specific dermatomal/myotomal deficits. | Clinical exam, MRI brachial plexus (nerve root integrity, avulsions, neuromas), EMG/NCS (later). X-rays (rule out fracture). | Clinical presentation (posture, specific nerve deficits), absence of bony injury, MRI findings confirming nerve injury. |
| Clavicle Fracture | Most common birth injury. Pseudoparalysis (arm held still due to pain), focal tenderness, crepitus, swelling over clavicle. Moro reflex often present but guarded. | X-ray of clavicle (confirms fracture). | Pain response (pseudoparalysis), focal bony tenderness/deformity, specific X-ray finding. Neurological exam often normal distal to fracture. |
| Humeral Fracture | Pseudoparalysis, focal tenderness/swelling/deformity over humerus, crepitus. May have associated nerve injury (e.g., radial nerve). | X-ray of humerus (confirms fracture). | Pain response, focal bony findings, X-ray confirmation. Neurological deficits might be isolated to a single peripheral nerve (e.g., radial) rather than plexus pattern. |
| Septic Arthritis of Shoulder | Pseudoparalysis, fever, signs of systemic infection, severe pain with passive ROM, warmth/redness over joint. | Blood tests (CBC, CRP, ESR), Aspiration of joint fluid (culture, cell count), X-ray (joint effusion, periarticular osteopenia), Ultrasound (effusion). | Systemic signs of infection, severe pain on passive ROM, elevated inflammatory markers, positive joint aspirate. Rare in neonates without specific risk factors. |
| Central Nervous System Injury (e.g., stroke, hemiparesis) | Often associated with altered mental status, seizures, other neurological deficits (e.g., facial asymmetry, lower limb involvement). Weakness may be spastic rather than flaccid (later). | Cranial Ultrasound, MRI Brain. | Global neurological findings, often affecting multiple limbs or cranial nerves. Lack of specific peripheral nerve pattern. |
| Congenital Muscular Torticollis | Head tilt with chin rotated to opposite side, palpable mass (SCM tumor) in sternocleidomastoid. Not primarily arm paralysis. | Clinical exam, Ultrasound of SCM (muscle thickness, mass). | Distinct head posture, SCM mass, no arm paralysis. |
In our patient, the absence of bony tenderness, crepitus, and the specific pattern of neurological deficit (C5-C6 paralysis with sparing of lower roots) along with the confirmatory MRI findings firmly established the diagnosis of Brachial Plexus Birth Palsy, an Erb-Duchenne type lesion. The lack of systemic signs or focal joint findings ruled out septic arthritis. The isolated upper extremity paralysis without other CNS signs made a central lesion less likely.
Surgical Decision Making & Classification
Non-Operative Management Considerations
Initial management of brachial plexus birth palsy is often non-operative, involving physical therapy and observation, as up to 70-80% of cases demonstrate spontaneous recovery, especially those with neurapraxia or mild axonotmesis. This observation period typically lasts for 3 months.
However, specific indicators predict a poor prognosis for spontaneous recovery and necessitate surgical intervention:
*
Absence of biceps function by 3 months of age:
This is the most critical prognostic indicator. If no antigravity elbow flexion is present by this time, spontaneous functional recovery is unlikely.
*
Presence of a true avulsion (preganglionic injury) on MRI:
These injuries do not recover spontaneously.
*
Persistent global palsy:
While our patient had Erb's palsy, if multiple roots (C5-T1) were involved with no recovery, surgery would be strongly indicated.
In our case, the patient, at 3 weeks, showed no signs of active biceps function. The MRI, while not definitively showing avulsions, indicated severe injury (neuromas-in-continuity) to C5 and C6 roots. Given the macrosomic birth and shoulder dystocia, a severe stretch injury was highly suspected. The decision was made to prepare for early surgical intervention, recognizing the critical window for nerve regeneration and reinnervation.
Classification
For surgical planning and prognostication, several classification systems are used:
-
Narakas Classification (Based on nerve root involvement):
- Type I (Erb-Duchenne): C5-C6 involvement (most common, ~45-50%). Characterized by shoulder abduction/external rotation and elbow flexion weakness.
- Type II: C5-C7 involvement (~30-35%). Similar to Type I with added wrist extension weakness.
- Type III (Total Palsy with Intact Hand): C5-C8 involvement (~10%). Upper and forearm weakness, hand function generally preserved.
- Type IV (Total Palsy with Hand Involvement): C5-T1 involvement (~5%). Flail arm, often associated with Horner's syndrome.
- Our patient presented as a Narakas Type I injury, primarily affecting C5 and C6 roots.
-
Mallet Score (Functional Assessment):
This score assesses shoulder function post-injury, particularly useful for longitudinal follow-up and evaluating surgical outcomes. It rates shoulder abduction, external rotation, hand-to-neck, hand-to-spine, and hand-to-mouth movements on a scale of 1-5 (1=no function, 5=normal). This was used pre-operatively to establish a baseline (expected very low scores for C5-C6 movements) and would be critical in post-operative evaluation. -
Sunderland Classification (Pathological Severity):
While primarily a histological classification, it conceptually guides surgical indications.- Grade 1 (Neurapraxia): Temporary conduction block, full recovery expected.
- Grade 2 (Axonotmesis): Axon damage, intact endoneurium. Good recovery expected.
- Grade 3 (Neurotmesis): Axon and endoneurium damaged, intact perineurium. Incomplete recovery, often requires surgery.
- Grade 4 (Neurotmesis): Axon, endoneurium, perineurium damaged, intact epineurium. Requires surgical repair.
-
Grade 5 (Neurotmesis):
Complete transection of the nerve. Requires surgical repair.
Our patient's MRI findings, specifically the neuromas-in-continuity, suggested at least a Sunderland Grade 3 or 4 injury, indicating the need for exploration and potential repair/grafting.
Surgical Decision
Given the severe C5-C6 palsy, the lack of any clinical signs of recovery at 3 weeks, and MRI evidence of significant injury (neuroma-in-continuity), a surgical exploration and nerve reconstruction were indicated. The critical window for nerve repair in BPBP is generally considered to be between 3 and 9 months of age, with optimal outcomes for primary repair often within 3-6 months. We aimed for surgical intervention at 3-4 months if no definitive recovery of biceps function was observed, to allow for a brief period of observation for potential minimal spontaneous recovery and to ensure the infant's physiological readiness for surgery.
The specific surgical plan was for neurolysis, nerve grafting, and/or nerve transfer to restore shoulder abduction, external rotation, and elbow flexion.
Surgical Technique / Intervention
The patient underwent surgical exploration and nerve reconstruction at 4 months of age, as no significant recovery of biceps function (AMS 0/7) had occurred by 3 months, confirming the need for intervention.
Patient Positioning and Preparation
The patient was positioned supine on the operating table. The head was turned gently to the left and secured in a neutral position to expose the right supraclavicular and infraclavicular regions. The right arm was draped free to allow for intraoperative manipulation and assessment. A sterile field extending from the neck to the mid-chest and down the entire right upper extremity was prepared. The left lower extremity was also prepped for potential sural nerve graft harvest.
Surgical Approach and Exposure
A standard supraclavicular incision was made, extending from the posterior border of the sternocleidomastoid muscle at the level of the cricoid cartilage laterally across the clavicle towards the trapezius. The platysma muscle was divided. The sternocleidomastoid muscle was retracted medially. The omohyoid muscle was identified and often divided for better exposure.
The key anatomical landmarks identified were the phrenic nerve (medial to the anterior scalene), the transverse cervical artery (often ligated for clarity), and the anterior scalene muscle. The brachial plexus was meticulously dissected from the surrounding fascia and fat. Careful identification of the individual nerve roots (C5, C6, C7, C8, T1) was performed as they emerged from between the anterior and middle scalene muscles.
Intraoperative Findings
Upon exposure, the C5 and C6 nerve roots were found to be severely attenuated, with a large, firm neuroma-in-continuity involving both roots, extending distally towards the upper trunk. Neurolysis was attempted, but significant fibrosis and scar tissue within the neuroma made simple decompression unlikely to yield recovery. Intraoperative nerve action potential (SNAP/CMAP) studies were performed across the neuroma, demonstrating no conductive signal, confirming a complete disruption (Sunderland Grade 4 or 5 equivalent). The C7 root appeared less involved, but was also thin and pale. The C8 and T1 roots appeared healthy and demonstrated good intraoperative stimulation responses. There was no evidence of root avulsion from the spinal cord, confirming postganglionic injury.
Surgical Reconstruction
The decision was made to resect the neuromatous segments of C5 and C6, and perform interpositional nerve grafting, along with a nerve transfer for robust elbow flexion.
-
Resection of Neuroma and Graft Harvest: The non-conducting, scarred segments of C5 and C6 roots were sharply resected, revealing healthy fascicles proximally and distally. The resultant gaps were measured to be approximately 3 cm for C5 and 2.5 cm for C6. The ipsilateral sural nerve was harvested from the calf as a multi-fascicular cable graft, providing sufficient length and caliber.
-
Nerve Grafting (C5 and C6):
- The sural nerve grafts were reversed to ensure correct fascicular orientation (proximal end of graft to proximal nerve stump, distal end of graft to distal nerve stump, as nerve growth factor gradient is proximodistal).
- Under high magnification with a surgical microscope, the harvested sural nerve grafts were coapted to the resected C5 and C6 nerve stumps using 10-0 monofilament nylon sutures. Epineurial repair was performed, ensuring tension-free coaptation and precise alignment of the fascicles.
- The proximal C5 stump was grafted to the distal C5 upper trunk fascicles.
-
The proximal C6 stump was grafted to the distal C6 upper trunk fascicles.
This was primarily intended to restore shoulder abduction/external rotation.
-
Nerve Transfer (Oberlin Transfer for Elbow Flexion):
To ensure reliable reinnervation of elbow flexion (biceps), a direct nerve transfer was performed. The medial pectoral nerve, which is a expendable motor nerve, was identified arising from the medial cord. A fascicle of the ulnar nerve destined for the brachialis/biceps was identified and sectioned distally. The medial pectoral nerve was then coapted to the distal stump of the musculocutaneous nerve (or a specific fascicle leading to biceps) under microscopic guidance, again using 10-0 nylon sutures. This "Oberlin transfer" provides a strong, reliable source of axons to the biceps, prioritizing restoration of active elbow flexion, which is critical for hand-to-mouth function.
Figure 2: Intraoperative image showing microsurgical nerve coaptation using fine sutures (e.g., 10-0 nylon) under magnification during nerve grafting (not specific to this patient, but representative of the technique).
-
Additional Nerve Transfers (Spinal Accessory to Suprascapular):
To further augment shoulder external rotation and abduction (supraspinatus and infraspinatus muscles), the spinal accessory nerve was identified and a portion of it (or an accessory branch if available) was transferred to the suprascapular nerve. The suprascapular nerve was identified and sectioned proximally, and the chosen donor fascicle of the spinal accessory nerve was coapted to its distal stump. This transfer improves the outcome of shoulder function significantly.
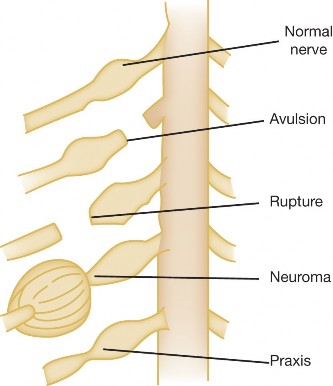
Figure 3: Intraoperative view of the brachial plexus after careful dissection, showing individual nerve roots and trunks prior to nerve grafting and transfer procedures (not specific to this patient, but illustrative of the surgical field).
Closure
After ensuring hemostasis, a small closed suction drain was placed. The platysma was approximated, and the skin was closed in layers using absorbable sutures for the dermis and a running subcuticular suture for the skin. A sterile dressing was applied.
Post-Operative Protocol & Rehabilitation
Immediate Post-Operative Period
- Immobilization: The patient's right arm was placed in an airplane splint or a long arm cast with the shoulder abducted to 90 degrees, externally rotated to 30 degrees, and the elbow flexed to 90 degrees. This position minimizes tension on the nerve repair sites, particularly the grafted segments and the Oberlin transfer. The splint was custom-made and maintained for 4-6 weeks.
- Pain Management: Opioid analgesia (e.g., oral morphine) was administered initially, transitioning to acetaminophen as appropriate for an infant. Local anesthetic infiltration at the surgical site and regional blocks were utilized intraoperatively to minimize immediate post-operative pain.
- Monitoring: Regular neurovascular checks of the hand and fingers were performed to ensure adequate perfusion and detect any signs of compression. Wound inspection for infection or hematoma was done daily.
Rehabilitation (After Splint Removal)
The long-term success of brachial plexus surgery relies heavily on a dedicated and consistent rehabilitation program.
-
Phase 1: Early Passive Range of Motion (Weeks 4-12 post-op):
- After splint removal, a specialized pediatric physical therapist initiated gentle passive range of motion (PROM) exercises to all joints of the right upper extremity (shoulder, elbow, wrist, hand). The goal was to prevent contractures and maintain joint mobility without stressing the nerve repair sites.
- Parents were extensively educated on proper PROM techniques and the importance of daily home exercises.
- Sensory re-education was initiated, focusing on tactile stimulation and desensitization of the affected limb.
-
Phase 2: Active Movement and Strengthening (Months 3-12 post-op):
- As nerve regeneration progressed, typically evidenced by muscle twitching (Tinel's sign advancement) and initial signs of active muscle contraction, active assistive ROM and then active ROM exercises were introduced.
- Focus was placed on early re-establishment of elbow flexion and shoulder external rotation.
- Strengthening exercises using gravity and light resistance were progressively introduced. These were often play-based to engage the infant.
- Mallet scoring was performed every 3 months to objectively track improvements in shoulder function. The Active Movement Scale (AMS) was used for specific muscle groups.
-
Phase 3: Functional Integration and Addressing Secondary Deformities (Beyond 12 months):
- Emphasis shifted to integrating the affected limb into daily activities. Occupational therapy played a crucial role.
- Regular follow-up appointments (every 3-6 months) were scheduled to monitor motor recovery, identify potential contractures, and assess for glenohumeral dysplasia, a common sequela.
-
Management of Secondary Procedures:
- Shoulder internal rotation contractures often develop due to persistent muscle imbalance (stronger internal rotators). If conservative measures fail, secondary procedures like Latissimus Dorsi and Teres Major tendon transfers (for external rotation) or subscapularis release may be considered, typically after 2-3 years of age, or once nerve regeneration plateaus.
- Humeral derotational osteotomy may be necessary in older children with persistent internal rotation deformity and functional deficits.
- Elbow flexion contractures or persistent elbow extension weakness may require triceps to biceps transfer.
Monitoring and Follow-up
- Close monitoring of motor recovery using the AMS and Mallet score.
- Regular pediatric orthopedic and neurosurgical team evaluations.
- Serial X-rays of the shoulder to monitor for glenohumeral joint incongruity or dysplasia (e.g., posterior subluxation of the humeral head, glenoid retroversion).
- Psychological support for parents and child as needed.
Pearls & Pitfalls (Crucial for FRCS/Board Exams)
Pearls
- Early Diagnosis & Referral: Prompt recognition of arm paralysis at birth is paramount. Early referral to a specialized brachial plexus center is critical for timely evaluation and optimal management.
- "Biceps at 3 Months" Rule: The most important prognostic indicator for spontaneous recovery. Absence of antigravity biceps function by 3 months is a strong indication for surgical consideration. Don't wait beyond 6 months for surgical intervention in severe cases.
- MRI is King for Surgical Planning: High-resolution MRI of the brachial plexus is indispensable for visualizing nerve root integrity, identifying avulsions (pseudomeningoceles), neuromas-in-continuity, and guiding surgical strategy (grafting vs. transfer). Perform early (3-4 weeks) if severe palsy is suspected.
- Prioritize Function: Surgical goals prioritize key functions: elbow flexion (hand-to-mouth), followed by shoulder abduction/external rotation. These contribute most significantly to quality of life.
- Multidisciplinary Team: Optimal care involves a team approach including pediatric orthopedics, neurosurgery, hand surgery, physical and occupational therapists, neurologists, and specialized nurses.
- Parental Education & Realistic Expectations: Thoroughly educate parents about the injury, the surgical process, the extensive rehabilitation required, and the often protracted timeline for recovery. Manage expectations regarding full functional recovery.
- Sural Nerve is the Workhorse Graft: The sural nerve is the preferred donor nerve for grafting due to its length, fascicular pattern, and minimal donor site morbidity.
- Oberlin Transfer for Biceps: The medial pectoral to musculocutaneous nerve transfer (Oberlin transfer) is highly effective for restoring elbow flexion with excellent predictability and minimal donor morbidity.
- Spinal Accessory to Suprascapular: This transfer is crucial for improving shoulder abduction and external rotation.
Pitfalls
- Delayed Diagnosis/Referral: Missing the critical window for nerve repair (ideally 3-6 months, definitely before 9-12 months) significantly compromises outcomes due to irreversible muscle atrophy and target organ degeneration.
- Misdiagnosis of Associated Bony Injuries: Failing to rule out clavicle or humeral fractures can lead to inappropriate management and worsening of the overall outcome. Always obtain radiographs.
- Over-reliance on Spontaneous Recovery: While many cases recover, severe injuries, particularly those involving C5-C6 with no biceps function at 3 months, warrant surgical intervention. Assuming "wait and see" for too long is a common pitfall.
- Incomplete Assessment of Injury Extent: Failing to identify all involved nerve roots or distinguishing preganglionic (avulsion) from postganglionic (rupture/neuroma) injuries can lead to inappropriate surgical techniques.
- Poor Surgical Technique: Inadequate microsurgical coaptation, tension at repair sites, or misidentification of donor/recipient nerves can lead to graft failure or poor reinnervation.
- Lack of Adherence to Rehabilitation: Nerve regeneration is slow, and consistent, long-term physical therapy is essential. Poor compliance from families or inadequate access to specialized therapy will severely limit functional outcomes.
- Ignoring Secondary Deformities: Failure to address persistent muscle imbalances leading to contractures (e.g., shoulder internal rotation contracture) or glenohumeral dysplasia will limit functional gains even after successful nerve repair. These often require secondary procedures.
- Underestimating T1 Involvement: Missing a lower trunk injury (C8-T1) can lead to unrecognized hand dysfunction or Horner's syndrome, indicating a more severe global palsy.
