Introduction & Epidemiology
Spinal degenerative disorders represent a significant burden on healthcare systems globally, with intervertebral disc pathology being a predominant etiology of axial and radicular pain, as well as myelopathy. As orthopedic surgeons, a comprehensive understanding of the intricate relationship between the intervertebral disc and associated neural tissue is paramount for accurate diagnosis, appropriate management, and successful surgical outcomes. Disc herniation, degeneration, and instability frequently lead to direct mechanical compression, chemical irritation, and vascular compromise of neural structures, manifesting as radiculopathy, myelopathy, or cauda equina syndrome. The prevalence of symptomatic disc herniation is estimated to be 1-3% of the population, peaking in the fourth and fifth decades of life. Lumbar disc herniation accounts for approximately 95% of cases at L4-L5 and L5-S1, while cervical disc herniation most commonly occurs at C5-C6 and C6-C7. Myelopathy secondary to cervical or thoracic disc pathology, though less common, carries a higher morbidity risk due to potential irreversible spinal cord damage. This review will delineate the vital link between neural tissue and the intervertebral disc, from basic neuroanatomy to advanced surgical principles.
Surgical Anatomy & Biomechanics
Understanding the fundamental anatomy and physiology of neural tissue, particularly the peripheral nervous system (PNS) and its interaction with the central nervous system (CNS) elements within the spinal canal, is crucial for any spinal surgeon.
Peripheral Nervous System (PNS) Fundamentals
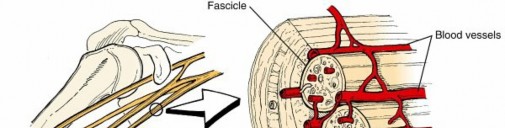
The PNS comprises all neural structures outside the brain and spinal cord, including cranial nerves, spinal nerves, and their associated ganglia. Its components are directly susceptible to mechanical compression and chemical irritation from disc pathology.
Neuron
The fundamental unit of the nervous system, responsible for transmitting electrical and chemical signals.
*
Cell body (Soma):
The metabolic center, housing the nucleus and organelles. It represents approximately 10% of the overall size of a neuron.
*
Axon hillock:
A specialized region where the cell body tapers into the axon, serving as the site of action potential initiation.
*
Axons:
Typically one long process, sometimes with collateral branches, that propagates nerve impulses away from the cell body. They connect the neuron to the spinal cord (for sensory afferents) or end-organs (for motor efferents).
*
Dendrites:
Multiple, tree-like processes extending from the cell body that receive signals from surrounding nerve cells, acting as the primary receptive surfaces.
*
Myelin sheath:
A lipid-rich insulating layer that surrounds many axons.
Neurophysiology of Myelin
The myelin sheath is critical for efficient nerve conduction.
*
Composition:
Primarily composed of galactocerebroside, sphingomyelin, and cholesterol.
*
Function:
Significantly speeds wave propagation or conduction.
Thicker myelin sheaths directly correlate with increased conduction velocity.
*
Formation in PNS:
Produced by Schwann cells.
*
Schwann cells:
* Originate from the neural crest during embryonic development.
* Play a critical role in post-traumatic nerve regeneration due to their ability to produce various neurotrophic factors and guide axonal regrowth.
*
Secretions:
Produce nerve growth factor-β (NGF-β), brain-derived neurotrophic factor (BDNF), insulin-like growth factor 1 (IGF-1), and erythropoietin – all vital for neuronal survival, growth, and repair.
*
Myelinated fibers:
A single Schwann cell ensheathes a single axon, spiraling around it multiple times. Each Schwann cell covers an axonal footprint of approximately 100 µm.
*
Nodes of Ranvier:
Unmyelinated gaps between adjacent Schwann cells. These nodes are characterized by a high concentration of voltage-gated Na+ channels, which are essential for saltatory conduction.
*
Saltatory conduction:
The action potential "jumps" from one node of Ranvier to the next, dramatically increasing the speed of impulse propagation compared to continuous conduction in unmyelinated fibers.
*
Unmyelinated fibers:
A single Schwann cell typically surrounds multiple axons, but without forming a spiraled myelin sheath.
*
Axolemma:
The specialized membrane that surrounds the axon. It is crucial for maintaining the neuron's membrane potential.
*
Resting potential:
Maintained by the Na-K pumps, which actively transport 3 Na+ ions out and 2 K+ ions into the cell. This electrogenic pump, along with differential ion permeability, establishes a resting membrane potential of approximately −70 mV, with the cell interior having a relative negative charge.

Depiction of a typical neuron illustrating the cell body, dendrites, axon, and myelin sheath.

Illustration of Schwann cells forming myelin around a peripheral axon, highlighting the nodes of Ranvier.
Spinal Cord and Nerve Roots
The spinal cord, a continuation of the brainstem, extends from the foramen magnum to the conus medullaris (typically L1-L2). It serves as the primary conduit for sensory and motor information between the brain and the periphery.
*
Gray Matter:
Butterfly-shaped central region containing neuronal cell bodies, dendrites, and unmyelinated axons. Divided into dorsal (sensory), ventral (motor), and lateral (autonomic) horns.
*
White Matter:
Surrounds the gray matter, composed of myelinated axons organized into ascending (sensory) and descending (motor) tracts.
*
Spinal Nerves:
31 pairs of spinal nerves emerge from the spinal cord, each formed by the union of a dorsal (sensory) root and a ventral (motor) root. These roots traverse the subarachnoid space, protected by the dura mater and arachnoid mater, before exiting the spinal canal through the intervertebral foramina.
*
Dorsal Root Ganglia (DRG):
Located within the intervertebral foramen, these ganglia contain the cell bodies of sensory neurons. The DRG is particularly sensitive to compression and inflammation, contributing significantly to radicular pain.
*
Cauda Equina:
Below the conus medullaris, the spinal canal contains a bundle of lumbar, sacral, and coccygeal nerve roots, resembling a horse's tail. Compression of these roots can lead to cauda equina syndrome, a neurosurgical emergency.

Cross-section of the spinal cord showing gray and white matter, and the emergence of spinal nerve roots.
Intervertebral Disc Anatomy and Innervation
The intervertebral disc is a complex fibrocartilaginous structure between adjacent vertebral bodies, composed of the annulus fibrosus, nucleus pulposus, and cartilaginous endplates.
*
Annulus Fibrosus:
Concentric lamellae of collagen fibers (Type I and II) that encase the nucleus pulposus. The outer layers are richly innervated, primarily by the sinuvertebral nerve (recurrent meningeal nerve) and branches from the gray rami communicantes, contributing to discogenic pain.
*
Nucleus Pulposus:
A gel-like core composed of proteoglycans, water, and collagen fibers (Type II), acting as a hydrostatic shock absorber. It is typically avascular and aneural in healthy adults, but neovascularization and innervation can occur with degeneration.
*
Cartilaginous Endplates:
Thin layers of hyaline cartilage that connect the disc to the vertebral bodies, allowing nutrient diffusion.
Biomechanics of Disc-Neural Interaction
Degenerative processes in the intervertebral disc can lead to structural compromises that directly impinge upon or chemically irritate neural elements.
*
Disc Herniation:
Protrusion or extrusion of nuclear material into the spinal canal or intervertebral foramen.
*
Mechanical Compression:
Direct pressure on the spinal cord, cauda equina, or exiting nerve roots (e.g., in subarticular, foraminal, or far-lateral herniations). This can lead to focal demyelination, axonal injury, and impaired axonal transport.
*
Chemical Irritation:
The nucleus pulposus contains inflammatory mediators (e.g., TNF-α, IL-1β, prostaglandin E2, nitric oxide, phospholipase A2) that, upon exposure to neural tissue, induce an inflammatory response, edema, and nociceptive stimulation, exacerbating neural dysfunction and pain.
*
Vascular Compromise:
Compression can compromise the microvasculature supplying the nerve root or spinal cord (e.g., radicular arteries, anterior spinal artery), leading to ischemia and hypoxia, further contributing to neural injury.
*
Disc Degeneration and Instability:
Loss of disc height, annular tears, and osteophyte formation can lead to segmental instability, narrowing of the spinal canal (spinal stenosis) and intervertebral foramina. This dynamic compression further compromises neural elements.
*
Myelopathy:
Spinal cord compression, most commonly in the cervical spine, resulting in upper motor neuron signs (spasticity, hyperreflexia, gait disturbance, fine motor deficits).
*
Radiculopathy:
Nerve root compression, resulting in sensory (pain, paresthesia, numbness) and/or motor deficits (weakness, atrophy) in a dermatomal/myotomal distribution.

Sagittal view depicting a disc herniation compressing the adjacent neural elements, specifically a nerve root.
Neural Imaging Considerations
High-resolution Magnetic Resonance Imaging (MRI) is the gold standard for evaluating disc-neural conflicts.
*
T1-weighted images:
Excellent for anatomical detail, showing fat bright, water dark, and cortical bone dark. Useful for assessing neural element morphology.
*
T2-weighted images:
Demonstrates fluid (including CSF and edematous tissue) as bright, ideal for visualizing disc herniations, spinal cord edema (myelomalacia), and CSF surrounding neural structures.
*
Myelography/CT Myelography:
Utilized when MRI is contraindicated or insufficient, providing dynamic information on spinal canal patency and nerve root impingement.
Indications & Contraindications
The decision-making process for intervention in disc-related neural pathology is complex, weighing neurological deficit, symptom severity, duration, and patient comorbidities.
Non-Operative Indications
- Acute radiculopathy without progressive neurological deficit: Most acute disc herniations resolve with conservative management.
- Mild to moderate pain: Manageable with analgesics, anti-inflammatory medications (NSAIDs, corticosteroids), and physical therapy.
- Absence of neurological deficits: Or stable, non-progressive deficits.
- Successful response to epidural steroid injections: For radicular pain refractory to oral medications and physical therapy.
- Patient preference: In carefully selected cases where surgical risks outweigh potential benefits.
Operative Indications
- Progressive neurological deficit: Worsening motor weakness, sensory loss, or myelopathy.
- Cauda Equina Syndrome: (CES) – characterized by bilateral radicular pain, saddle anesthesia, bowel/bladder dysfunction, and variable lower extremity weakness. This is a surgical emergency requiring urgent decompression.
- Intractable radicular pain: Severe pain refractory to a prolonged course (typically 6-12 weeks) of conservative management.
- Cervical Myelopathy: Clear evidence of spinal cord compression with clinical myelopathic signs and symptoms.
- Severe spinal stenosis: With disabling claudication or radiculopathy.
Contraindications
-
Absolute Contraindications:
- Active systemic infection or local infection at the surgical site.
- Uncontrolled coagulopathy.
- Severe cardiopulmonary disease precluding general anesthesia.
- Patient refusal or inability to comply with post-operative rehabilitation.
-
Relative Contraindications:
- Significant psychological comorbidities.
- Uncontrolled diabetes or other systemic diseases that increase surgical risk.
- Extreme obesity.
- Extensive prior surgery with significant scarring (may increase complexity and risk).
- Lack of clear anatomical correlation between symptoms and imaging findings.
Operative vs. Non-Operative Indications
| Indication Category | Non-Operative Management | Operative Management |
|---|---|---|
| Pain | Mild to moderate radicular/axial pain | Intractable radicular pain refractory to 6-12 weeks conservative care |
| Neurological Deficit | No deficit or stable, non-progressive sensory deficit | Progressive motor weakness, worsening sensory deficit, or bowel/bladder dysfunction |
| Spinal Cord | No myelopathy | Any degree of myelopathy (cervical/thoracic) |
| Cauda Equina | N/A (CES is always an operative indication) | Cauda Equina Syndrome (urgent decompression) |
| Imaging | Disc bulge/protrusion with good spinal canal reserve | Significant disc herniation (extrusion/sequestration) with neural compression |
| Duration | Acute symptoms (<6 weeks), often improving spontaneously | Chronic symptoms (>12 weeks) with persistent neural compromise |
| Functional Impairment | Mild to moderate, manageable with daily activities | Severe functional impairment, inability to perform ADLs |
Pre-Operative Planning & Patient Positioning
Meticulous pre-operative planning is critical to optimize surgical outcomes and minimize complications related to neural tissue.
Pre-Operative Planning
- Comprehensive Clinical Evaluation: Detailed history of pain characteristics, neurological symptoms, and functional limitations. Thorough neurological examination including motor strength, sensation, reflexes, and gait.
-
Imaging Review:
- MRI: Review T1, T2, STIR, and sometimes post-contrast sequences. Identify the exact level and nature of disc pathology (protrusion, extrusion, sequestration), extent of neural compression (spinal cord, cauda equina, nerve roots), presence of edema (myelomalacia), and foraminal stenosis.
- CT: Useful for bony anatomy, osteophyte formation, calcified discs, and assessment of spinal canal dimensions. Also used for 3D reconstruction in complex cases.
- Plain Radiographs: Evaluate spinal alignment, stability (flexion-extension views if indicated), disc height, and degenerative changes.
- Electrophysiological Studies (EMG/NCS): May be indicated in cases of diagnostic uncertainty or to differentiate radiculopathy from peripheral neuropathy. Not typically required for straightforward disc herniations with clear neurological findings.
- Medical Optimization: Address comorbidities (diabetes, hypertension, cardiac disease) to reduce perioperative risks. Smoking cessation counseling is vital for fusion cases.
- Informed Consent: Detailed discussion with the patient about surgical rationale, expected outcomes, and potential complications, particularly those related to neural injury.
-
Intraoperative Neuromonitoring (IONM):
Highly recommended for all spinal surgeries involving neural elements, especially for myelopathy or complex revisions. Modalities include:
- Somatosensory Evoked Potentials (SSEPs): Assess the integrity of posterior column sensory pathways.
- Motor Evoked Potentials (MEPs): Assess corticospinal tract motor pathway integrity.
- Electromyography (EMG): Monitors nerve root function and detects spontaneous electrical activity (neurotonic discharges) indicative of irritation or injury.
Patient Positioning
Proper patient positioning is paramount to ensure surgical access, maintain physiological homeostasis, and protect vulnerable neural and vascular structures.
*
General Principles:
*
Padding:
All pressure points (heels, knees, elbows, shoulders, face) must be meticulously padded to prevent neuropathies (e.g., ulnar, peroneal nerve palsy) or skin breakdown.
*
Alignment:
Maintain physiological spinal alignment to prevent undue tension on neural structures.
*
Vascular Access:
Ensure adequate venous access and arterial line placement if indicated.
*
Specific Positions:
*
Prone Position (for Posterior Lumbar/Thoracic/Cervical Approaches):
* Utilize a frame (e.g., Jackson table, Wilson frame) or bolsters to allow the abdomen to hang freely, reducing intra-abdominal pressure and epidural venous bleeding.
* Head in a neutral or slightly flexed position, secured in a Mayfield head clamp for cervical spine cases, ensuring no undue tension on the brachial plexus.
* Arms abducted less than 90 degrees and padded to prevent brachial plexus stretch injury.
* Legs slightly flexed at hips and knees, with adequate padding.
*
Supine Position (for Anterior Cervical/Lumbar Approaches):
* Cervical: Head in neutral position, slightly extended with a shoulder roll to optimize exposure. Ensure careful neck hyperextension to avoid brachial plexus or spinal cord stretch.
* Lumbar (ALIF): Patient flat, ensuring adequate abdominal exposure, often with a slight Trendelenburg position.
*
Lateral Decubitus Position (for Lateral Lumbar Approaches - LLIF/DLIF):
* Patient secured to the operating table. Axillary roll to protect the brachial plexus.
* Top leg slightly flexed, bottom leg extended with padding between.
* Ensure adequate padding to the dependent arm and leg.
Detailed Surgical Approach / Technique
Surgical intervention for disc-neural pathology aims to achieve neural decompression, often coupled with stabilization. The choice of approach depends on the level, pathology, and surgeon preference.
Microdiscectomy (Lumbar)
The gold standard for lumbar radiculopathy secondary to disc herniation.
1.
Incision:
Small (2-3 cm) midline or paramedian skin incision, typically over the affected interspace.
2.
Muscle Dissection:
Subperiosteal dissection of the paraspinal muscles from the spinous process and lamina, typically on one side.
3.
Exposure:
Retraction of muscles to expose the ligamentum flavum and underlying lamina.
4.
Ligamentum Flavum Resection:
A small laminotomy (bone window) or partial hemilaminectomy may be performed. The ligamentum flavum is resected using Kerrison rongeurs and pituitary rongeurs to expose the dura and nerve root.
5.
Neural Decompression & Retraction:
The epidural fat is gently mobilized, and the nerve root (typically exiting under the pedicle of the superior vertebra) is identified. A nerve root retractor is used to carefully retract the nerve root medially, exposing the disc space.
6.
Discectomy:
The posterior annulus is incised. Pituitary rongeurs are used to remove the herniated disc fragment. Care is taken to identify and remove any free fragments (sequestration) from the epidural space. Over-aggressive curettage of the disc space is generally avoided to reduce the risk of future instability or collapse.
7.
Inspection:
The nerve root and dura are inspected for adequate decompression.
8.
Closure:
Hemostasis, layered closure of fascia, subcutaneous tissue, and skin.
Anterior Cervical Discectomy and Fusion (ACDF)
Commonly performed for cervical radiculopathy or myelopathy caused by disc herniation or osteophyte formation.
1.
Incision:
Transverse skin incision in a skin crease (typically at C5-C6 or C6-C7), usually on the right side to avoid the recurrent laryngeal nerve.
2.
Muscle Dissection:
Subplatysmal dissection. The medial border of the sternocleidomastoid muscle is identified. The interval between the carotid sheath laterally and the trachea/esophagus medially is developed.
3.
Exposure:
The prevertebral fascia is opened, exposing the longus colli muscles and the anterior cervical spine. The self-retaining retractors are carefully placed to protect the recurrent laryngeal nerve (left side), esophagus, trachea, and carotid sheath.
4.
Disc Excision:
The anterior annulus is incised. The disc material (nucleus pulposus and degenerated annulus) is removed using pituitary rongeurs and curettes. Special care is taken to remove posterior osteophytes and disc material impinging on the spinal cord or nerve roots using a high-speed burr and Kerrison rongeurs. The posterior longitudinal ligament is typically removed to ensure complete decompression.
5.
Decompression & Endplate Preparation:
The spinal cord and nerve roots are visually inspected for full decompression. The cartilaginous endplates of the vertebral bodies are prepared to facilitate fusion, removing cartilage but preserving the cortical bone.
6.
Fusion:
An interbody cage, often filled with autograft or allograft, is impacted into the disc space.
7.
Plate Fixation:
An anterior cervical plate is applied to provide immediate stability and enhance fusion rates.
8.
Closure:
Hemostasis, layered closure.
Posterior Lumbar Interbody Fusion (PLIF) / Transforaminal Lumbar Interbody Fusion (TLIF)
Performed for degenerative disc disease, spondylolisthesis, or recurrent disc herniation requiring decompression and stabilization.
1.
Incision & Exposure:
Midline incision. Subperiosteal dissection of paraspinal muscles bilaterally to expose lamina, facet joints, and transverse processes.
2.
Laminectomy/Hemilaminectomy:
Performed to decompress the spinal canal. The inferior facet of the superior vertebra and the superior facet of the inferior vertebra may be trimmed (facetectomy) to facilitate nerve root decompression and cage insertion.
3.
Nerve Root Decompression:
Identification and careful decompression of the nerve roots. In TLIF, a unilateral approach is used, preserving more paraspinal musculature and contralateral facet.
4.
Discectomy:
The annulus is incised, and the disc material is removed from the intervertebral space. In PLIF, disc removal is bilateral. In TLIF, disc removal is unilateral through the excised facet joint. The endplates are prepared for fusion.
5.
Interbody Graft/Cage Insertion:
One or two interbody cages (PLIF) or a single banana-shaped cage (TLIF) filled with bone graft material are impacted into the disc space to restore disc height and promote fusion.
6.
Pedicle Screw Instrumentation:
Pedicle screws are placed into the vertebral bodies above and below the fused segment, connected by rods to provide rigid internal fixation and stability.
7.
Posterolateral Fusion (PLF):
Autograft or allograft bone may be placed posterolaterally over the decorticated transverse processes and lamina to augment fusion.
8.
Closure:
Hemostasis, layered closure.
Intraoperative Neural Monitoring (IONM) Integration
During all these procedures, IONM plays a critical role. Persistent EMG activity during nerve root manipulation, changes in SSEP or MEP waveforms (amplitude reduction or latency prolongation) can indicate neural compromise, prompting the surgeon to modify the technique (e.g., adjust retraction, re-evaluate decompression) to prevent permanent injury.
Complications & Management
Despite meticulous surgical technique, complications can arise, particularly involving neural elements.
Intraoperative Complications
-
Dural Tear and CSF Leak:
Incidence varies from 1-10%.
- Management: Primary repair with fine non-absorbable sutures (e.g., 5-0 or 6-0 Prolene), often reinforced with dural sealants (e.g., DuraSeal, Tisseel), fat graft, or muscle patch. Post-operatively, strict bed rest, head elevation, and lumbar drain (if large tear) may be required.
-
Nerve Root Injury:
Direct trauma from instruments, excessive retraction, or thermal injury (burr). Can lead to persistent pain, numbness, or motor weakness. Incidence <1%.
- Management: Immediate recognition is key. Release compression, careful inspection of the nerve. Post-operatively, manage with corticosteroids, gabapentinoids, and physical therapy. Severe cases may require nerve exploration or repair by a peripheral nerve specialist.
-
Spinal Cord Injury:
Most severe complication, leading to partial or complete paralysis (myelopathy) or cauda equina syndrome. More common in cervical/thoracic spine surgery. Incidence <0.1%.
- Management: Immediate cessation of offending maneuver, ensure adequate decompression, correct any malpositioning. High-dose steroids may be administered, though evidence for efficacy is limited. Neurological assessment and urgent re-exploration if deficits persist.
-
Vascular Injury:
E.g., great vessels (aorta, vena cava) in lumbar anterior approaches; vertebral artery or carotid artery in cervical approaches. Incidence <0.1%.
- Management: Immediate pressure, vascular surgery consultation for repair.
-
Hemorrhage:
Epidural hematoma potentially compressing neural elements.
- Management: Meticulous hemostasis intraoperatively. If symptomatic post-op, urgent evacuation.
Post-Operative Complications
-
Infection (Surgical Site Infection):
Superficial or deep. Incidence 0.5-5%.
- Management: Superficial infections may respond to oral antibiotics. Deep infections often require surgical debridement, washout, and prolonged intravenous antibiotics.
-
Recurrent Disc Herniation:
Re-herniation at the same level. Incidence 5-15%.
- Management: Initial conservative management. If symptomatic and persistent, revision surgery (re-discectomy, fusion) may be indicated.
-
Failed Back Surgery Syndrome (FBSS):
Persistent or new pain after spinal surgery. Multifactorial etiology (inadequate decompression, recurrent pathology, arachnoiditis, psychosocial factors). Incidence 10-40%.
- Management: Thorough diagnostic workup to identify specific etiology. May involve conservative measures, injections, revision surgery, spinal cord stimulation, or intrathecal drug delivery systems.
-
Hardware-Related Complications:
Screw malposition, loosening, breakage.
- Management: Revision surgery for symptomatic hardware or loss of stability.
-
Non-Union/Pseudarthrosis (in fusion cases):
Failure of bone graft to heal across the interbody or posterolateral space. Incidence 5-20%.
- Management: Conservative management for asymptomatic cases. Revision surgery with additional grafting, instrumentation, or different fusion technique for symptomatic non-union.
-
Adjacent Segment Disease (ASD):
Degenerative changes at levels adjacent to a fused segment. Incidence up to 25% over 10 years.
- Management: Initial conservative treatment. If symptomatic with neural compression or instability, extension of fusion or motion-preserving surgery may be considered.
Common Complications & Salvage Strategies
| Complication | Incidence (Approx.) | Salvage Strategies |
|---|---|---|
| Dural Tear / CSF Leak | 1-10% | Primary suture repair, dural sealant, fat/muscle graft, lumbar drain, strict bed rest. |
| Nerve Root Injury | <1% | Intraoperative recognition and release, post-op conservative management, nerve exploration for severe cases. |
| Spinal Cord Injury | <0.1% | Immediate cessation of compression, re-evaluation of decompression, high-dose steroids (controversial). |
| Surgical Site Infection | 0.5-5% | Debridement, washout, IV antibiotics. |
| Recurrent Disc Herniation | 5-15% | Conservative management initially, revision microdiscectomy or fusion if symptomatic. |
| Failed Back Surgery Syndrome | 10-40% | Multidisciplinary approach: re-evaluation, injections, SCS, intrathecal pumps, revision surgery. |
| Non-Union / Pseudarthrosis | 5-20% | Conservative management for asymptomatic. Revision fusion, additional instrumentation/graft for symptomatic. |
| Adjacent Segment Disease | Up to 25% (10 yrs) | Conservative care, injections. If symptomatic, extension of fusion or disc arthroplasty at adjacent level. |
| Hematoma (Epidural) | <1% | Urgent surgical evacuation if symptomatic. |
| Vascular Injury | <0.1% | Immediate pressure, emergent vascular surgery consultation and repair. |
Post-Operative Rehabilitation Protocols
Post-operative rehabilitation is crucial for optimizing neural recovery, strengthening core musculature, and facilitating a safe return to functional activities. Protocols vary depending on the surgical procedure, patient specific factors, and the presence of any neurological deficit.
Immediate Post-Operative Phase (Days 0-7)
- Pain Management: Multimodal analgesia (opioids, NSAIDs, acetaminophen, muscle relaxants, gabapentinoids).
- Neurological Monitoring: Frequent neurological checks to detect any new or worsening deficits.
-
Mobilization:
- Microdiscectomy: Early mobilization (out of bed, short walks) is encouraged within hours post-op, emphasizing spinal precautions (avoiding flexion, extension, and rotation).
- Fusion Procedures: Brace may be prescribed depending on the extent of fusion, patient's bone quality, and surgeon preference. Early ambulation, but activity is more restricted. Log rolling technique for bed mobility.
- Wound Care: Monitor for signs of infection or CSF leak.
- Patient Education: Reinforce spinal precautions, proper body mechanics, and activity restrictions.
Early Rehabilitation Phase (Weeks 1-6)
- Spinal Precautions: Continue to avoid bending, lifting, and twisting (BLT restrictions) for lumbar procedures. Cervical procedures typically have restrictions on active neck flexion/extension/rotation.
-
Physical Therapy:
- Gentle range of motion: Non-stressful movements within pain-free limits.
- Core stabilization exercises: Focus on activating deep abdominal and lumbar multifidus muscles without spinal loading.
- Nerve Glides/Sliders: Gentle exercises to improve nerve mobility and reduce tension, especially beneficial for persistent radicular symptoms.
- Posture Education: Emphasis on ergonomic principles.
- Walking: Progressively increase duration and distance of walking.
- Activity Restriction: No heavy lifting (>5-10 lbs), strenuous exercise, or prolonged sitting/standing.
Intermediate Rehabilitation Phase (Weeks 6-12)
-
Progression of Exercises:
Gradually increase intensity and complexity of exercises.
- Strengthening: Progressive resistance exercises for core, gluteal, and lower extremity musculature.
- Flexibility: Gentle stretching for hamstrings, hip flexors, and pectorals, as appropriate.
- Balance and Proprioception: Incorporate exercises to improve balance and body awareness.
- Cardiovascular Conditioning: Introduce low-impact aerobic activities (e.g., stationary bike, elliptical).
- Activity Modification: May begin light work activities if physically tolerable and approved by the surgeon. Avoid repetitive bending, lifting, or twisting.
Advanced Rehabilitation Phase (Weeks 12+)
- Functional Training: Progress to sport-specific or work-specific activities.
- High-Impact Activities: Gradually introduce higher impact activities, based on fusion status (if applicable) and resolution of symptoms.
- Return to Work/Sport: A structured, graded return to full activity under the guidance of the surgeon and therapist. For fusion patients, radiographic confirmation of solid fusion is often desired before full return to strenuous activities.
- Long-Term Maintenance: Emphasize lifelong adherence to core strengthening, proper body mechanics, and regular exercise to prevent recurrence and maintain spinal health.
Summary of Key Literature / Guidelines
Evidence-based guidelines inform optimal management of disc-related neural pathology, ranging from conservative care to surgical intervention.
-
SPORT Trial (Spine Patient Outcomes Research Trial):
Landmark studies comparing operative and non-operative treatments for lumbar disc herniation, spinal stenosis, and degenerative spondylolisthesis.
- Lumbar Disc Herniation: Demonstrated that patients undergoing surgery for lumbar disc herniation experienced greater improvement in pain and function at 2-year follow-up compared to those treated non-operatively, although a substantial percentage of non-operative patients eventually crossed over to surgery. The long-term (10-year) results showed little difference between the initial treatment groups, highlighting that surgery provides faster relief but long-term outcomes may converge.
- Implications: Supports surgical intervention for carefully selected patients with persistent symptoms after conservative treatment, especially for immediate symptomatic relief.
-
NASS (North American Spine Society) Clinical Guidelines:
Provides comprehensive, evidence-based recommendations for the diagnosis and treatment of various spinal conditions, including lumbar and cervical radiculopathy and myelopathy. These guidelines are crucial for standardizing care and informing decision-making.
- Key Recommendations: Emphasize a trial of conservative care, judicious use of imaging, and clear indications for surgical decompression in cases of progressive neurological deficit, intractable pain, or myelopathy.
- Cochrane Reviews and Meta-analyses: Numerous systematic reviews consolidate evidence regarding the effectiveness of different surgical techniques (e.g., microdiscectomy vs. open discectomy, ACDF vs. disc arthroplasty) and conservative treatments. These often highlight areas where further high-quality research is needed.
-
Emerging Technologies and Concepts:
- Minimally Invasive Spine Surgery (MISS): Techniques such as tubular microdiscectomy, endoscopic spine surgery, and lateral interbody fusions have shown comparable efficacy to open techniques for appropriate indications, often with benefits of reduced blood loss, shorter hospital stays, and faster recovery. The neural impact of these approaches, particularly concerning nerve root manipulation and retraction, requires careful consideration.
- Disc Regeneration: Ongoing research explores biological treatments, such as growth factors, cell-based therapies, and gene therapy, aimed at regenerating disc tissue and potentially preventing or reversing degeneration, which could have profound implications for neural health by maintaining disc height and stability.
- Artificial Disc Replacement (ADR): For specific cervical and lumbar indications, ADR aims to preserve motion at the treated segment, theoretically reducing the risk of adjacent segment disease. Long-term neural outcomes compared to fusion remain an area of active study.
In conclusion, the intricate "vital link" between neural tissue and the intervertebral disc dictates the complex presentation and management strategies for spinal pathologies. A deep understanding of neuroanatomy, neurophysiology, biomechanics, and evidence-based surgical principles is essential for all orthopedic surgeons dedicated to spinal care.
Clinical & Radiographic Imaging