INTRODUCTION TO ULNAR NERVE PATHOLOGY
The ulnar nerve is critical for the fine motor dexterity and intrinsic power of the hand. Injuries or chronic compressive neuropathies of the ulnar nerve present significant reconstructive challenges for the orthopedic surgeon. High ulnar nerve injuries, in particular, are notorious for poor intrinsic muscle recovery due to the prolonged distance regenerating axons must travel, often leading to irreversible motor endplate degradation before reinnervation can occur.
To address this, the paradigm of ulnar nerve reconstruction has shifted toward distal nerve transfers, which bypass the zone of injury and provide a local source of motor axons. Concurrently, the management of chronic compressive neuropathies, such as tardy ulnar nerve palsy and cubital tunnel syndrome, has evolved significantly, with a growing emphasis on evidence-based, minimally invasive, and anatomically respectful surgical interventions.
TARDY ULNAR NERVE PALSY AND CUBITAL TUNNEL SYNDROME
Cubital tunnel syndrome represents the second most common compression neuropathy in the upper extremity. It is characterized by compression or traction of the ulnar nerve as it traverses the medial elbow. Tardy ulnar nerve palsy, a historical term often associated with delayed neuropathy following childhood lateral condyle fractures or cubitus valgus deformities, shares a similar pathophysiological endpoint requiring meticulous evaluation and management.
Pathoanatomy and Biomechanics
The ulnar nerve is vulnerable to compression at several distinct anatomical sites around the elbow:
* The arcade of Struthers (hiatus in the medial intermuscular septum).
* The medial epicondyle (retroepicondylar groove).
* The cubital tunnel proper (beneath the arcuate ligament of Osborne, extending between the humeral and ulnar heads of the flexor carpi ulnaris [FCU]).
* The deep flexor pronator aponeurosis.
Biomechanically, elbow flexion decreases the cross-sectional area of the cubital tunnel by up to 55%, while simultaneously increasing intraneural pressure and placing longitudinal traction on the nerve. Prolonged flexion, therefore, induces transient ischemia, which, if repetitive, leads to intraneural edema, localized demyelination, and eventual axonal loss.
Clinical Evaluation and Classification
The severity of cubital tunnel syndrome dictates the treatment algorithm. The classification system originally proposed by McGowan, and later revised by Dellon, categorizes the dysfunction into three distinct stages:
- Mild Dysfunction (Grade I): Characterized by intermittent paresthesias in the ulnar digits and subjective weakness. There is no measurable intrinsic atrophy or objective motor loss.
- Moderate Dysfunction (Grade II): Presents with intermittent paresthesias accompanied by measurable weakness in pinch or grip strength. Early signs of intrinsic imbalance may be present.
- Severe Dysfunction (Grade III): Defined by persistent, unremitting paresthesias, measurable weakness, and profound intrinsic muscle atrophy (e.g., first dorsal interosseous wasting, Wartenberg's sign, or clawing).
Conservative Management Protocols
Conservative treatment is the first-line approach for mild to moderate cubital tunnel syndrome and should be rigorously attempted for a minimum of 3 months before surgical intervention is considered.
The cornerstone of non-operative management is activity modification and postural correction. Patients must be instructed to avoid prolonged elbow flexion, particularly in the workplace (e.g., resting elbows on desks) and during sleep.
Nighttime splinting is highly effective. Patients are provided with elbow extension splints to maintain the elbow at approximately 30 to 45 degrees of flexion.
Clinical Pearl: The extension splint must not be fitted with the forearm held in forced pronation. Pronation exacerbates symptoms by increasing tension across the flexor-pronator origin and further compressing the nerve. A neutral forearm position is optimal.
If formal splints are poorly tolerated, a simple towel or pillow secured loosely around the anterior elbow can adequately limit extreme flexion during sleep. Svernlov et al. reported a highly successful improvement rate of 89.5% in patients with mild to moderate cubital tunnel syndrome treated conservatively with a combination of elbow extension splinting, patient education, and activity modification.
Surgical Indications and Current Trends
Surgical decompression is recommended for patients who fail a 3-month trial of conservative management, those presenting with severe dysfunction (Grade III), or those with progressive motor deficits. Careful preoperative evaluation, including Electromyography (EMG) and Nerve Conduction Studies (NCS), is mandatory to confirm the diagnosis, localize the lesion, and establish a baseline for postoperative comparison.
The surgical armamentarium for cubital tunnel syndrome includes:
1. Simple (In Situ) Decompression: Open or endoscopic.
2. Medial Epicondylectomy: Minimal or partial.
3. Anterior Transposition: Subcutaneous, intramuscular, or submuscular.
Current Evidence-Based Paradigm:
Historically, anterior transposition was considered the gold standard. However, the current trend strongly favors simple or in situ decompression for primary, uncomplicated cases. Prospective randomized studies have demonstrated that in situ decompression yields clinical outcomes equal to those of anterior transposition, with lower complication rates and faster recovery times.
Surgical Warning: When performing an in situ decompression, the surgeon must take immense care to release all constricting tissues (from the arcade of Struthers down to the deep FCU fascia) without destabilizing the nerve. Avoid aggressive circumferential neurolysis, which devascularizes the nerve and promotes perineural scarring.
Transposition is now generally reserved for specific indications:
* Patients with a subluxating ulnar nerve (either preoperatively or iatrogenically induced during decompression).
* Valgus deformity of the elbow.
* Revision surgery for recurrent symptoms.
* Severe osteophyte formation in the retroepicondylar groove.
Outcomes in the Literature:
* Simple Decompression: Taniguchi et al. reported good-to-excellent results in 82% (14 of 17) of patients treated with simple decompression through a minimal incision. Goldfarb et al. noted that only 7% of 56 patients experienced recurrent symptoms postoperatively.
* Medial Epicondylectomy: Good results range from 45% to 93%. However, persistent medial elbow pain is a known complication, reported in up to 45% of patients at 6 months postoperatively. Amako et al. compared minimal versus partial medial epicondylectomy and found no difference in neurological improvement, leading them to recommend the minimal approach to preserve the medial collateral ligament complex and reduce pain.
* Transposition: Studies comparing subcutaneous and submuscular transpositions have found no significant differences in long-term outcomes. Techniques utilizing a fasciodermal sling (constructed from the antebrachial fascia overlying the flexor-pronator mass) to stabilize the transposed nerve have also shown favorable results.
NERVE TRANSFER FOR ULNAR NERVE RECONSTRUCTION
In cases of high ulnar nerve transection or severe proximal crush injuries, primary repair or grafting often yields dismal intrinsic hand function. The distance from the proximal forearm or arm to the intrinsic muscles of the hand is too great; the motor endplates undergo irreversible fibrosis before the regenerating axons (growing at approximately 1 mm per day) can reach them.
To circumvent this, Mackinnon and Novak (1999) introduced a groundbreaking technique: transferring the distal portion of the anterior interosseous nerve (AIN) directly into the deep motor branch of the ulnar nerve in the distal forearm/wrist. This distal nerve transfer drastically reduces the regenerative distance, allowing for rapid reinnervation of the intrinsic muscles. In 2002, Haase and Chung corroborated the efficacy of this technique, reporting excellent return of intrinsic function.
Rationale and Biomechanics
The AIN is a motor branch of the median nerve. Its terminal branch supplies the pronator quadratus (PQ) muscle. Because forearm pronation is redundantly powered by the pronator teres, sacrificing the terminal AIN to the PQ results in negligible functional deficit.
The deep motor branch of the ulnar nerve supplies the hypothenar muscles, all interossei, the third and fourth lumbricals, and the adductor pollicis. By coapting the AIN to the ulnar motor fascicles in the distal forearm, a high-density, synergistic motor nerve is utilized to restore critical pinch strength and prevent clawing.
🔪 Surgical Technique 62-14: AIN to Ulnar Motor Nerve Transfer
Indications: High ulnar nerve injuries (proximal to the mid-forearm) with no evidence of intrinsic recovery, ideally performed within 6 to 9 months of the initial injury.
Step 1: Positioning and Incision
The patient is positioned supine with the arm extended on a hand table. A tourniquet is applied to the proximal arm.
Make a longitudinal incision over the ulnar neurovascular bundle in the distal third of the forearm, extending distally across the wrist crease into the Guyon canal.
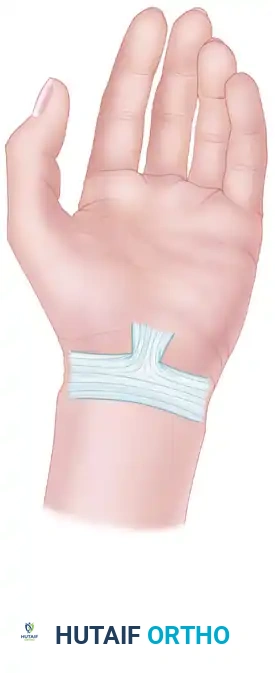
Figure A: Surface anatomy and planned incision over the distal ulnar forearm extending into Guyon's canal.
Step 2: Exposure of the Ulnar Nerve and Guyon's Canal
Deepen the incision through the subcutaneous tissue. Incise the volar carpal ligament to decompress Guyon's canal fully. Identify the ulnar neurovascular bundle. Retract the ulnar artery radially to expose the ulnar nerve.
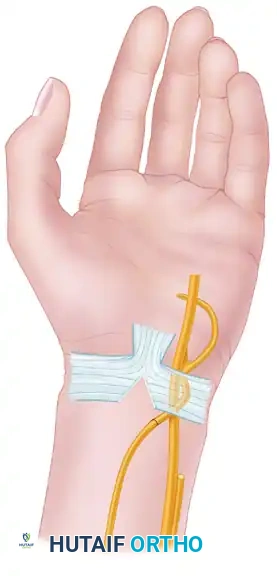
Figure B: Deep surgical exposure revealing the ulnar nerve, ulnar artery, and the surrounding fascial structures.
Step 3: Intraneural Dissection of the Ulnar Nerve
Identify the bifurcation of the ulnar nerve into its superficial sensory branch and deep motor branch within the palm, as it curves around the pisiform and hook of the hamate.
Using loupe or microscopic magnification, carefully perform an intraneural neurolysis, separating the motor fascicles from the sensory fascicles proximally into the distal forearm, up to the level of the proximal border of the pronator quadratus.
Surgical Pearl: The topography of the ulnar nerve in the distal forearm is highly predictable. The motor fascicles are typically located in the ulnar-dorsal quadrant of the nerve. Meticulous interfascicular dissection is required to avoid injuring the sensory component.
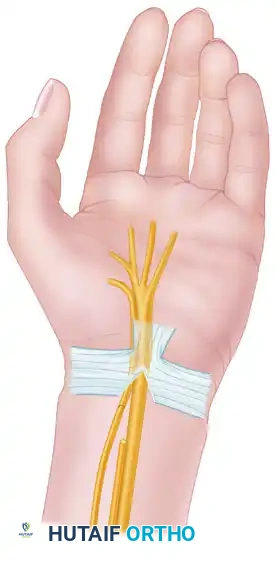
Figure C: Intraneural dissection separating the ulnar motor fascicles from the ulnar sensory nerve.
Step 4: Identification and Harvest of the AIN
Retract the flexor tendons radially to expose the pronator quadratus muscle. Identify the terminal motor branch of the AIN (supplying the pronator quadratus) coursing along the interosseous membrane and entering the undersurface of the muscle.
Dissect the AIN distally as far as possible into the muscle belly to maximize length. Transect the AIN at its terminal branching point within the pronator quadratus.
Step 5: Nerve Coaptation
Return to the neurolyzed ulnar nerve. Transect the isolated ulnar motor fascicles proximally, ensuring enough length is maintained on the distal stump to allow for a tension-free swing toward the AIN.
Bring the distal stump of the ulnar motor branch and the proximal stump of the AIN together.
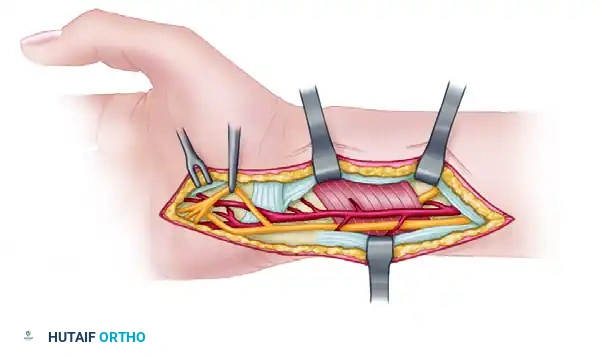
Figure D: Final tension-free coaptation of the terminal AIN to the deep motor branch of the ulnar nerve.
Under operating microscope guidance, perform an epineurial/perineurial repair using 9-0 or 10-0 nylon sutures. Fibrin glue may be used to augment the repair and reduce the number of sutures required, thereby minimizing foreign body reaction and scarring. Deflate the tourniquet, achieve meticulous hemostasis, and close the wound in layers.
Postoperative Care and Rehabilitation
The postoperative protocol for this specific distal nerve transfer deviates from traditional nerve repair paradigms.
Postoperatively, no splinting is involved. The patient is encouraged to begin immediate active and passive range of motion of the wrist and fingers.
Rationale for Immediate Motion:
Because the nerve coaptation is performed in a tension-free manner within the well-vascularized bed of the distal forearm, immobilization is unnecessary. Immediate motion prevents the formation of restrictive perineural adhesions, promotes healthy nerve gliding, and facilitates intrinsic stretching and strengthening exercises.
Motor reeducation is a critical component of the rehabilitation phase. Patients work closely with a specialized hand therapist to perform "donor activation" exercises. They are instructed to actively pronate the forearm (firing the AIN) while simultaneously attempting to abduct and adduct the fingers (ulnar intrinsic function). Over time, cortical plasticity allows the patient to dissociate these movements, resulting in seamless, independent intrinsic hand function.
CONCLUSION
The management of ulnar nerve pathology requires a nuanced understanding of anatomy, biomechanics, and nerve regeneration. For chronic compressive conditions like cubital tunnel syndrome, a conservative-first approach followed by anatomically respectful in situ decompression yields excellent results for the majority of patients. In the setting of devastating high ulnar nerve injuries, the AIN to ulnar motor nerve transfer represents a monumental advancement in peripheral nerve surgery, offering a reliable, biomechanically sound pathway to restore the irreplaceable intrinsic function of the hand.
