Introduction to Orthopaedic Microsurgery
Microsurgery has fundamentally transformed the landscape of orthopaedic reconstruction, expanding the boundaries of limb salvage, nerve repair, and composite tissue transplantation. The application of microvascular techniques is now indispensable in the surgical management of complex hand injuries, replantation of amputated parts, and the transfer of vascularized bone, muscle, and cutaneous flaps to both the upper and lower extremities.
The successful execution of microsurgery demands more than just a theoretical understanding of anatomy; it requires the absolute mastery of specialized instrumentation, optical magnification, and meticulous tissue handling. This comprehensive guide delineates the foundational principles, surgical techniques, and postoperative protocols essential for the modern orthopaedic microsurgeon.
Optical Magnification and Ergonomics
Microsurgery encompasses surgical procedures performed on structures so diminutive that magnification via an operating microscope is an absolute prerequisite. While standard surgical magnifying loupes (up to 5× magnification) are adequate for macroscopic dissection and the identification of larger neurovascular bundles, they are insufficient for true microvascular anastomosis.
The Operating Microscope
An operating microscope providing magnification ranging from 16× to 40× is essential when manipulating structures less than 2 mm in diameter.
* Dissection and Exposure: Magnification of 6× to 10× is typically utilized for the initial dissection and mobilization of small nerves and vessels.
* Microsurgical Repair: Magnification of 16× to 25× is the standard for the actual anastomosis of vessels and the coaptation of nerves.
* High-Power Inspection: Magnification up to 40× may be employed to inspect the intimal integrity of a completed anastomosis.
For procedures necessitating an assistant who must actively participate in the microsurgical field, a double binocular (diploscope) microscope is mandatory. Triploscopes are also available for secondary assistants or observers. Modern microscopes are equipped with electrical foot controls to seamlessly adjust focus and magnification without breaking the sterile field or interrupting the surgeon's manual workflow.
Surgeon Ergonomics and Tremor Control
Regardless of baseline proficiency in macroscopic hand surgery, the acquisition of microsurgical skill is a distinct and demanding process. Extraneous movements are exponentially amplified under the microscope.
SURGICAL WARNING: Factors that induce fatigue or exacerbate physiologic tremor must be systematically eliminated. Surgeons should avoid caffeine consumption immediately prior to microsurgery, ensure adequate rest, and maintain a comfortable, neutral posture.
Bracing the elbows and wrists on a stable platform is critical. The surgeon must develop the discipline to maintain constant visual contact with the operating field through the microscope, relying on proprioceptive awareness to locate unseen hands and instruments.
Microsurgical Instrumentation and Suture Materials
A simplified, highly organized approach to instrumentation is preferred to minimize clutter and maximize efficiency.
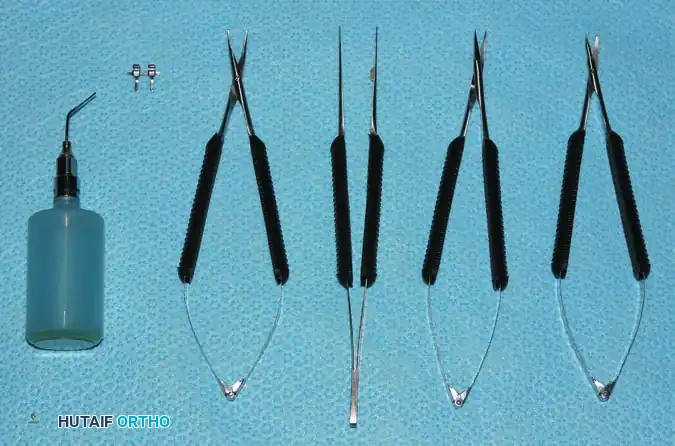
Essential Instruments
The basic microsurgical tray should include:
* Jeweler’s Forceps: Two or three straight and curved jeweler’s forceps are required for delicate tissue manipulation. Modified jeweler’s forceps are also utilized as bipolar coagulation forceps for the precise, low-energy coagulation of small side branches.
* Microscissors: Spring-loaded microscissors (adventitial and straight) are essential for vessel preparation and nerve trimming.
* Microvascular Clamps: Clamps must be carefully selected based on closing pressure. A closing pressure of less than 30 g/mm² is imperative for small vessels to achieve hemostasis without inducing crush injury to the delicate vascular intima.
* Accessory Tools: Micro-irrigating cannulas (using heparinized saline) and dilating probes are critical for vessel preparation and clearing the lumen of micro-thrombi.
Suture Materials
Microvascular and microneural repairs require ultrafine, non-absorbable monofilament sutures.
* Suture Diameters: Ranging from 18 to 35 µm.
* Needle Diameters: Atraumatic needles ranging from 50 to 139 µm.
* Standard Usage: Nylon sutures designated as 9-0, 10-0, 11-0, and 12-0 are commercially available. A 9-0 or 10-0 suture is typically used for larger vessels (e.g., radial/ulnar arteries), while 11-0 or 12-0 is reserved for digital vessels and nerve fascicles.
Training and Skill Acquisition
Surgeons should not expect to master microsurgery intuitively. The acquisition of these techniques requires rigorous, dedicated practice in an animal laboratory setting before clinical application.
CLINICAL PEARL: Initial training requires approximately 6 to 8 hours of daily practice in the laboratory for 2 to 3 weeks, typically utilizing a rat femoral artery/vein model. Thereafter, regular clinical or laboratory practice is essential to maintain proficiency.
Because clinical microsurgical procedures (such as replantations or free flaps) frequently require many hours, the efficiency of the surgeon and the operative team is a prime consideration in minimizing ischemia time and optimizing outcomes.
Microsurgical Treatment of Nerve Injuries
The goal of peripheral nerve repair is to achieve tension-free, anatomically precise coaptation of nerve fascicles to maximize axonal regeneration and functional recovery.
Primary Neurorrhaphy
Primary repair is indicated for sharp, clean nerve transections without segmental loss. The epineurium is meticulously approximated using 8-0 or 9-0 nylon sutures. Under microscopic magnification (16× to 25×), the surgeon must ensure that the fascicular topography is correctly aligned, utilizing surface landmarks such as longitudinal epineurial vessels to prevent rotational malalignment.
Interfascicular Grafting
When a nerve injury involves a crush component, avulsion, or delayed presentation resulting in a gap that cannot be closed without tension, interfascicular nerve grafting is mandatory.
* Tension is the enemy of nerve regeneration. Even mild tension causes ischemia at the repair site, leading to excessive scarring and blocked axonal sprouting.
* Technique: The neuroma is resected back to healthy fascicles. Autologous nerve grafts (most commonly the sural nerve or medial antebrachial cutaneous nerve) are harvested and reversed (to prevent axonal escape down side branches). The grafts are interposed between the corresponding fascicular groups using 10-0 or 11-0 nylon sutures.
Replantation Principles
Replantation of amputated parts represents one of the most dramatic applications of microsurgery. Success depends on careful patient selection, meticulous part preservation, and a highly coordinated surgical team.
Indications and Contraindications
Patient selection is paramount. Not every amputated part should be replanted.
Indications:
* Thumb amputations (at any level).
* Multiple digit amputations.
* Amputations in children (due to superior regenerative capacity and neuroplasticity).
* Single digit amputations distal to the flexor digitorum superficialis (FDS) insertion (Zone I), where functional recovery is highly predictable.
* Hand, wrist, or forearm amputations (macro-replantation).
Contraindications:
* Absolute: Severe crush or avulsion injuries with extensive segmental vessel damage; prolonged warm ischemia time (>12 hours for digits, >6 hours for major limb amputations containing muscle); life-threatening concomitant injuries.
* Relative: Single digit amputations in Zone II (due to poor functional outcomes from tendon adhesions); extreme age; severe preexisting medical comorbidities; severe psychiatric illness or inability to comply with rigorous postoperative rehabilitation.
Management of Ischemia Time
- Warm Ischemia: Anoxic time at room temperature. Muscle undergoes irreversible necrosis after 6 hours of warm ischemia. Digits (containing no muscle belly) can tolerate up to 12 hours.
- Cold Ischemia: The amputated part should be wrapped in saline-moistened gauze, placed in a sealed plastic bag, and then placed on an ice-water slurry (never directly on ice, which causes frostbite). Proper cooling extends the viable ischemia time to 24 hours for digits.
Order of Repair in Replantation
A standardized sequence of repair minimizes ischemia time and provides a stable platform for microvascular anastomosis.
- Bone Identification and Shortening: Adequate bone shortening is the most critical step to ensure tension-free vascular and neural repairs. Rigid fixation is achieved with K-wires, intraosseous wiring, or mini-plates.
- Extensor Tendon Repair: Provides dorsal stability.
- Flexor Tendon Repair: Performed before vascular repair to avoid disrupting delicate anastomoses later.
- Arterial Anastomosis: Re-establishes inflow. (In cases of prolonged ischemia, temporary shunting or arterial repair may precede tendon repair).
- Nerve Repair: Coaptation of digital nerves.
- Venous Anastomosis: Re-establishes outflow. A ratio of two veins repaired for every one artery is ideal to prevent venous congestion.
- Skin Closure: Must be loose and tension-free. Fasciotomies or skin grafting may be required.
Single-Stage Tissue Transfer (Free Flaps)
Free tissue transfer involves the detachment of a composite block of tissue (skin, muscle, bone, or a combination) on its dominant vascular pedicle and its transplantation to a distant recipient site via microvascular anastomosis.
Indications and Advantages
Free flaps are indicated for complex defects where local or regional flaps are inadequate, unavailable, or would result in unacceptable donor site morbidity.
* Advantages: Provides well-vascularized tissue to irradiated, infected, or poorly perfused beds; allows for single-stage reconstruction; facilitates early mobilization; and permits the transfer of specialized tissues (e.g., functioning muscle, vascularized bone).
Selection of Free Flaps
The choice of flap is dictated by the requirements of the recipient site (coverage, dead space obliteration, functional restoration, structural support).
Cutaneous and Fasciocutaneous Flaps
- Anterolateral Thigh (ALT) Flap: A highly versatile perforator flap based on the descending branch of the lateral circumflex femoral artery. It provides a large skin paddle, can be thinned, and has a long, robust pedicle.
- Lateral Arm Flap: Based on the posterior radial collateral artery. Excellent for moderate-sized defects of the hand and upper extremity, providing thin, pliable skin.
- Scapular and Parascapular Flaps: Based on the circumflex scapular artery. Provides a large, hairless skin paddle with a consistent pedicle, though it requires intraoperative patient repositioning.
- Free Groin Flap: Based on the superficial circumflex iliac artery. Historically significant but less commonly used today due to a short pedicle and variable anatomy.
Muscle and Musculocutaneous Flaps
- Latissimus Dorsi Transfer: Based on the thoracodorsal artery. The workhorse for massive defects requiring significant dead space obliteration (e.g., large lower extremity trauma). Can be harvested with or without a skin paddle.
- Rectus Abdominis Transfer: Based on the deep inferior epigastric artery. Provides excellent bulk and a long pedicle.
- Gracilis Muscle Transfer: Based on the medial circumflex femoral artery. Frequently utilized for functioning neuromuscular transfers (e.g., facial reanimation, restoration of finger flexion in brachial plexus injuries) by anastomosing both the vascular pedicle and the obturator nerve to a recipient motor nerve.
Free Vascularized Bone Transplants
- Free Vascularized Fibula Transfer: Based on the peroneal artery. The gold standard for reconstructing segmental bone defects greater than 6 cm (e.g., post-tumor resection, infected nonunions). It provides dense cortical bone capable of significant hypertrophy.
- Medial Femoral Condyle Flap: Based on the descending genicular artery. Ideal for smaller, recalcitrant nonunions (e.g., scaphoid nonunion) requiring highly osteogenic, vascularized cancellous bone.
Composite Transfers from the Foot
- Great Toe Wraparound Flap: Utilized for thumb reconstruction. Provides a customized skin/nail envelope while preserving the skeletal integrity of the donor great toe.
- Second Toe Transplantation: Ideal for replacing amputated fingers or for thumb reconstruction when joint transfer is required. Based on the dorsalis pedis/first dorsal metatarsal artery system.
Postoperative Care and Monitoring
The success of microvascular surgery extends well into the postoperative period. The primary cause of flap or replant failure is vascular thrombosis, which typically occurs within the first 48 to 72 hours.
General Postoperative Care
- Environment: The patient's room should be kept warm to prevent vasospasm.
- Positioning: The reconstructed extremity should be elevated to promote venous drainage but not so high as to compromise arterial inflow.
- Hydration and Hemodynamics: Maintain euvolemia and normotension. Avoid vasoconstrictive medications.
- Anticoagulation: Protocols vary by institution but often include aspirin, low-molecular-weight dextran, or subcutaneous heparin. Leeches (Hirudo medicinalis) may be employed for isolated venous congestion.
Monitoring Techniques
Continuous and meticulous monitoring is mandatory.
* Clinical Assessment: The most reliable indicator. Assess color, capillary refill (normal is 1-2 seconds), tissue turgor, and temperature.
* Arterial Insufficiency: Pale, cool, empty capillary refill.
* Venous Congestion: Blue/purple, swollen, rapid (<1 second) capillary refill, dark bleeding on pinprick.
* Objective Monitoring: Surface Doppler ultrasonography, implantable venous Doppler probes, and continuous surface temperature monitoring (a drop of >2°C compared to a control digit suggests vascular compromise) are critical adjuncts.
SURGICAL WARNING: If vascular compromise is identified, conservative measures (loosening dressings, adjusting position, hydration) should be attempted immediately. If improvement is not seen within 30 minutes, emergent operative re-exploration is mandatory. Time is tissue.
Conclusion
Orthopaedic microsurgery represents the pinnacle of technical precision in surgical reconstruction. Whether performing a complex digital replantation, a functioning free muscle transfer, or a vascularized bone graft, the surgeon must adhere strictly to the principles of meticulous tissue handling, tension-free anastomosis, and rigorous postoperative monitoring. Through dedicated laboratory training and a profound understanding of microvascular biomechanics, the orthopaedic surgeon can achieve remarkable functional salvage in limbs that would otherwise be lost.
