Introduction to Metastatic Bone Disease
Metastatic carcinoma represents the most common malignancy treated by orthopaedic surgeons, vastly outnumbering primary bone tumors. To contextualize the epidemiological burden, while approximately 8,000 new primary bone and soft tissue sarcomas are diagnosed annually in the United States, more than 1 million new carcinomas are diagnosed in the same timeframe. Autopsy studies and clinical registries estimate that 50% to 80% of patients with terminal carcinoma possess bone metastases at the time of death.
Historically, the presence of skeletal metastasis was viewed as a terminal event, often leading to a conservative, palliative approach. However, as systemic treatments for primary tumors—including targeted immunotherapies, advanced cytotoxic agents, and precise radiotherapy—continue to improve, patients are experiencing significantly longer survival times following the diagnosis of bone metastases. Consequently, it is imperative that the orthopaedic surgeon abandons any fatalistic attitude toward this patient population. Proper, aggressive orthopaedic care is crucial to minimize intractable pain, maintain skeletal stability, preserve patient independence, and ultimately optimize the overall quality of life.
Clinical Pearl: If a patient has a known history of carcinoma, even in the remote past, a newly discovered bone lesion is statistically most likely to be a metastasis. Furthermore, in any patient older than 40 years of age—even without a known history of malignancy—a newly discovered, aggressive-appearing bone lesion must be treated as metastatic carcinoma or multiple myeloma until proven otherwise.
Diagnostic Evaluation and Staging
The proper workup of a patient with suspected metastases of unknown origin is a critical sequence that must not be truncated. Failure to complete a comprehensive staging workup prior to biopsy can lead to catastrophic errors in patient care, including the inappropriate treatment of a primary bone sarcoma as a metastasis.
The Standardized Workup
The diagnostic algorithm for a suspected metastatic lesion includes:
1. Comprehensive History and Physical Examination: With specific attention to the breast, prostate, and thyroid.
2. Basic Laboratory Panels: Including complete blood count (CBC), comprehensive metabolic panel (CMP) with calcium levels, serum protein electrophoresis (SPEP), urine protein electrophoresis (UPEP), and prostate-specific antigen (PSA) in males.
3. Orthogonal Radiographs: Full-length imaging of the entire involved bone to assess for skip lesions or multiple metastases.
4. Advanced Imaging: A technetium-99m bone scan to identify polyostotic disease, followed by a computed tomography (CT) scan of the chest, abdomen, and pelvis to identify the primary visceral malignancy.
This systematic approach successfully identifies the primary lesion in more than 85% of patients presenting with metastases of unknown origin.
Biopsy Principles
Once the staging workup is complete, a biopsy is indicated.
Surgical Warning: Even if a patient has a known history of carcinoma, a biopsy of the first site of bone disease must be performed to establish a definitive histological relationship between the primary carcinoma and the suspected metastasis.
This biopsy must be executed with the exact same meticulous technique as a biopsy for a suspected primary sarcoma. In rare instances, a presumed metastasis may actually prove to be a primary bone sarcoma.
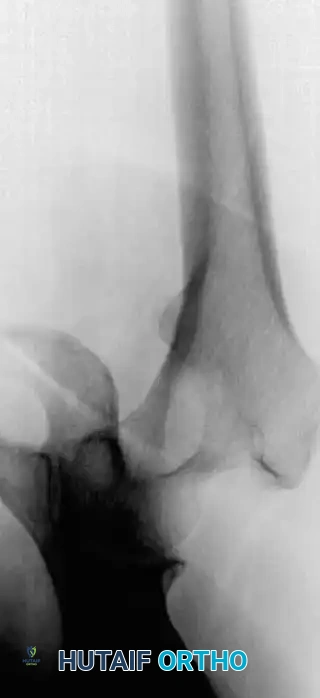

Fig. 22-19: (A) Anteroposterior view of the left hip of a 50-year-old woman with a history of breast cancer, disease-free for 2 years. A lytic lesion is seen in the femoral neck. Although the most likely diagnosis is metastatic carcinoma, a biopsy must be performed. This lesion proved to be a malignant fibrous histiocytoma (primary sarcoma). (B) Anteroposterior view after wide resection and endoprosthetic reconstruction. Prophylactic fixation of this presumed metastasis would have severely compromised her oncologic outcome.
Subsequent bone metastases in the same patient can generally be treated without further biopsy confirmation once the initial metastatic link is established.
Pathophysiology and Primary Tumor Characteristics
The vast majority of carcinomas metastatic to bone originate from the breast and prostate, followed by the lung, kidney, thyroid, and gastrointestinal tract. However, in patients presenting with metastases of unknown origin, the most common primary malignancies are lung or kidney cancer. Breast and prostate cancers are uncommon in this specific cohort because they are typically detected early in their disease course and tend to metastasize to bone relatively late. Conversely, lung and kidney cancers may remain clinically silent until late in the disease process, presenting initially as a skeletal metastasis.
Radiographic Appearance
The radiographic morphology of metastatic carcinoma is highly variable but generally exhibits an aggressive pattern with a wide zone of transition, cortical destruction, and lack of sclerotic margins.
* Blastic Lesions: Typically produced by prostate cancer and breast cancer.
* Lytic Lesions: Classically associated with kidney cancer, thyroid cancer, and gastrointestinal malignancies.
* Mixed Lesions: Often seen in lung cancer and breast cancer.
Diagnostic Pearl: If a metastatic lesion is located distal to the elbow or knee (acrometastasis), lung cancer is the most likely primary source. Additionally, metastatic lung cancer may present with a distinct radiographic appearance resembling a "bite" taken out of the cortex.
Histologically, the microscopic appearance of metastatic carcinoma usually mirrors the primary lesion. In well-differentiated cases, standard hematoxylin and eosin (H&E) staining yields the diagnosis. In poorly differentiated or sarcomatoid variants (e.g., sarcomatoid renal cell carcinoma), immunohistochemistry is required to identify epithelial markers (e.g., cytokeratin).
Indications for Surgical Intervention
The treatment of bone metastases is inherently multimodal, requiring close collaboration with medical and radiation oncologists. While systemic cytotoxic agents, hormone manipulation, and bisphosphonates (which inhibit osteoclast-mediated bone resorption) are managed medically, surgery is strictly indicated for the treatment of impending or actual pathological fractures.
Mirels' Criteria for Impending Fractures
The decision to perform prophylactic fixation of an impending pathological fracture relies on clinical judgment and biomechanical parameters. Mirels devised a highly validated scoring system that evaluates the risk of pathological fracture based on four variables:
Table 22-1: Mirels' Scoring System for Predicting Pathological Fracture
* Site: Upper limb (1 point), Lower limb (2 points), Peritrochanteric (3 points)
* Pain: Mild (1 point), Moderate (2 points), Functional/Severe (3 points)
* Lesion Type: Blastic (1 point), Mixed (2 points), Lytic (3 points)
* Size: < 1/3 of cortex (1 point), 1/3 to 2/3 of cortex (2 points), > 2/3 of cortex (3 points)
According to Mirels' criteria, prophylactic internal fixation should be strongly considered for any patient with a cumulative score of 8 or greater. Other widely accepted parameters for prophylactic fixation include:
* Pain that is refractory to radiation therapy.
* A lesion greater than 2.5 cm in diameter in a weight-bearing bone.
* Destruction of greater than 50% of the diaphyseal cortex.
* An avulsion fracture of the lesser trochanter.
Prophylactic vs. Post-Fracture Fixation
Two absolute principles govern the surgical management of metastatic bone disease:
1. Prophylactic internal fixation of an impending fracture is technically easier, yields better biomechanical constructs, and preserves local anatomy better than the fixation of an actual pathological fracture.
2. Patient morbidity, blood loss, and postoperative recovery times are significantly decreased with prophylactic fixation compared to post-fracture intervention.
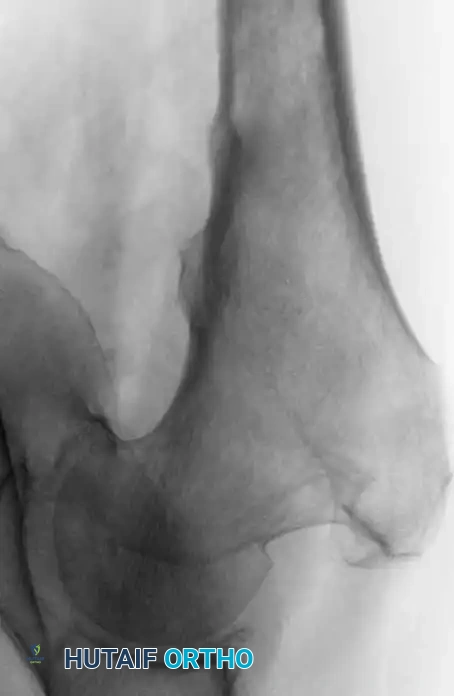
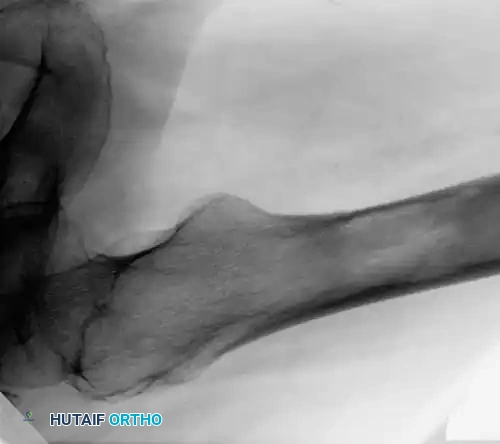
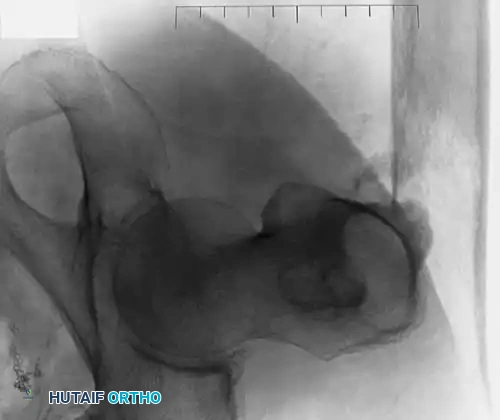
Fig. 22-24: (A, B) Anteroposterior and lateral radiographs of the proximal femur in an 82-year-old man with metastatic kidney cancer revealing multiple lytic lesions. Prophylactic fixation was canceled due to medical instability. (C) Radiograph several weeks later showing a completed pathological fracture. Surgery is now exponentially more difficult, and the patient has suffered severe morbidity.
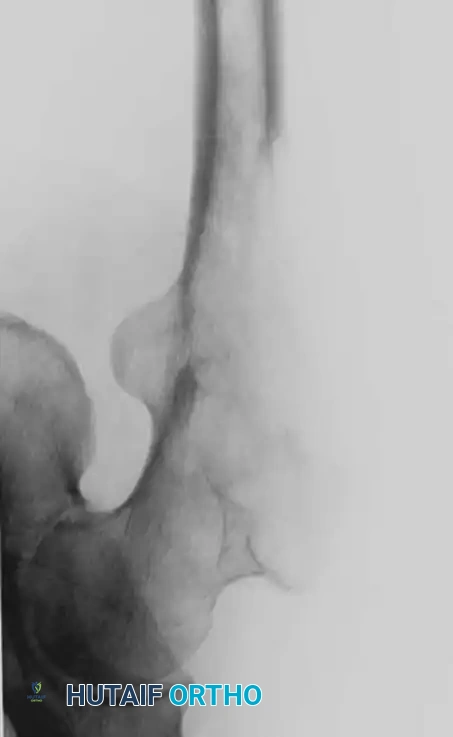
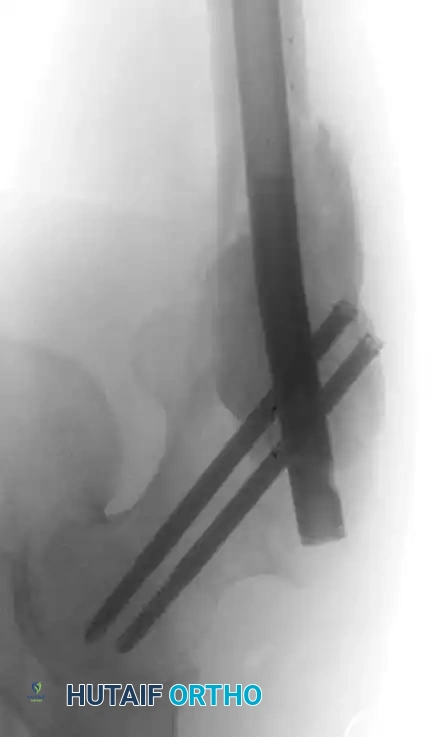
Fig. 22-20: (A) Impending pathological femoral fracture from metastatic squamous cell carcinoma. The femur fractured while the patient rolled in bed the night before scheduled prophylactic surgery. (B) Postoperative radiograph after fixation with a reconstruction nail and polymethylmethacrylate (PMMA) bone cement.
Surgical Principles and Biomechanics
The unpredictability of patient survival makes surgical decision-making challenging. Patients with metastatic lung cancer may survive 6 months to 5 years, while those with breast, prostate, or kidney cancer commonly live many years post-diagnosis.
Therefore, the surgical construct must meet two competing demands:
1. Immediate Stability: The fixation must be rigid enough to allow immediate full weight-bearing. A patient with a limited life expectancy must not be subjected to a prolonged period of restricted weight-bearing or complex rehabilitation.
2. Long-term Durability: The reconstruction must be durable enough to outlast the patient, potentially surviving for many years if the patient responds well to systemic therapy.
Core Operative Techniques
- Tumor Debulking: The tumor should be aggressively curetted and debulked prior to fixation to reduce local tumor burden and create space for structural augmentation.
- Cement Augmentation: The resulting cavitary defect must be filled with polymethylmethacrylate (PMMA) bone cement. PMMA provides immediate compressive strength and enhances screw purchase in compromised bone.
- Full Bone Protection: The entire bone should be protected with cephalomedullary or intramedullary fixation. Short segment plates are prone to failure as disease progresses elsewhere in the same bone.
Step-by-Step Surgical Approaches
1. Intramedullary Nailing and Cementation of Diaphyseal Lesions
For diaphyseal lesions of the femur, tibia, or humerus, locked intramedullary nailing is the gold standard.
Surgical Steps:
1. Positioning: Supine or lateral decubitus on a fracture table, depending on the bone and surgeon preference.
2. Approach: Standard entry portals (e.g., piriformis fossa or greater trochanter for the femur).
3. Preparation: The canal is sequentially reamed. Reamings should be sent for pathology if a biopsy has not been previously obtained.
4. Debulking: A small cortical window may be made over the lesion to thoroughly curette the tumor.
5. Cementation: PMMA is injected into the defect in a doughy state.
6. Implantation: The intramedullary nail is passed through the curing cement to interlock the construct. Proximal and distal locking screws are placed to provide rotational and axial stability.
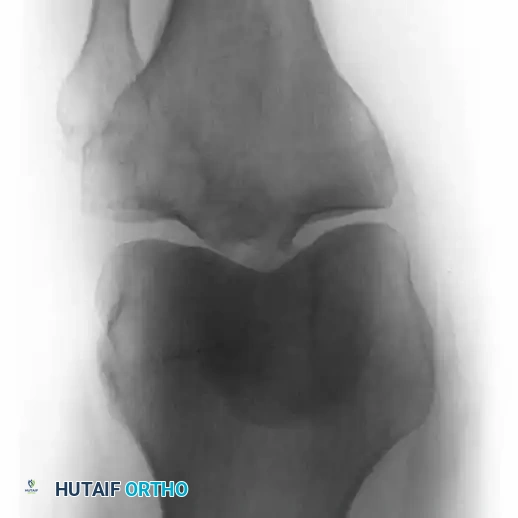
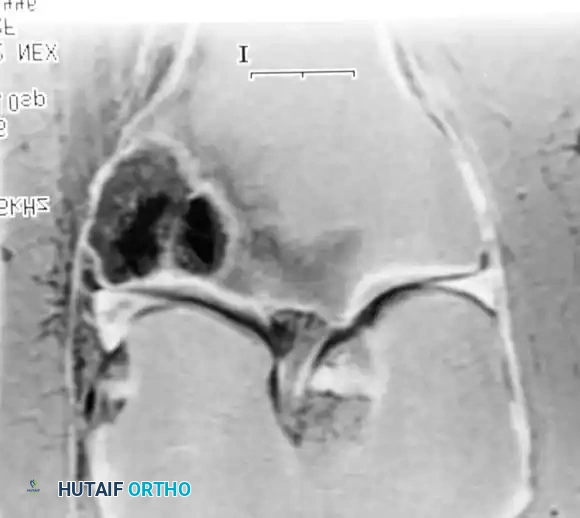
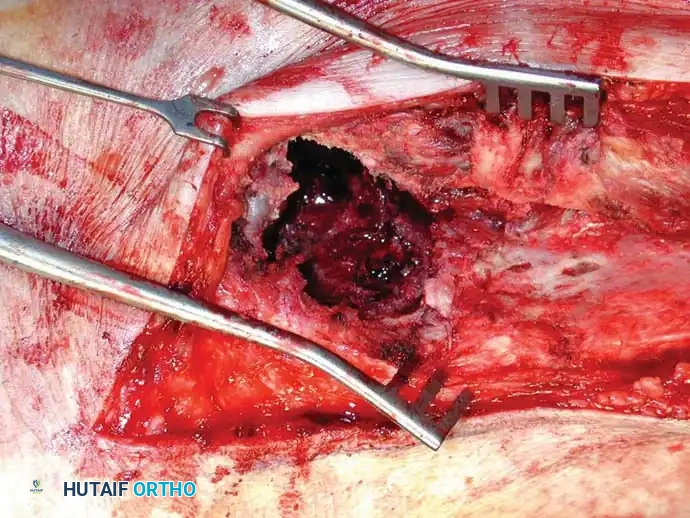
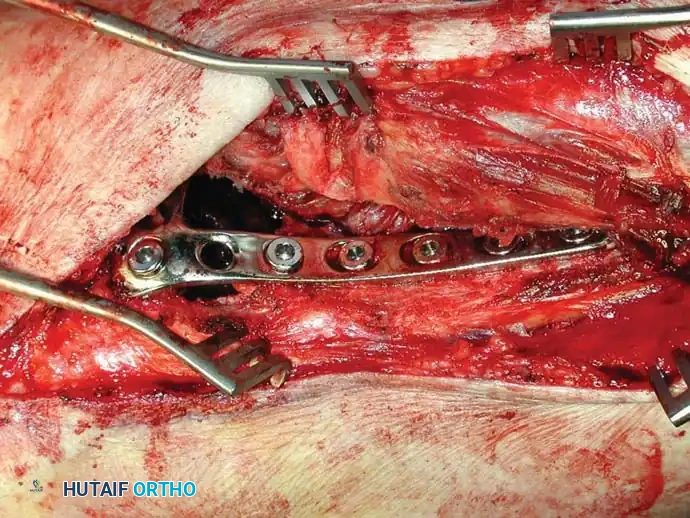
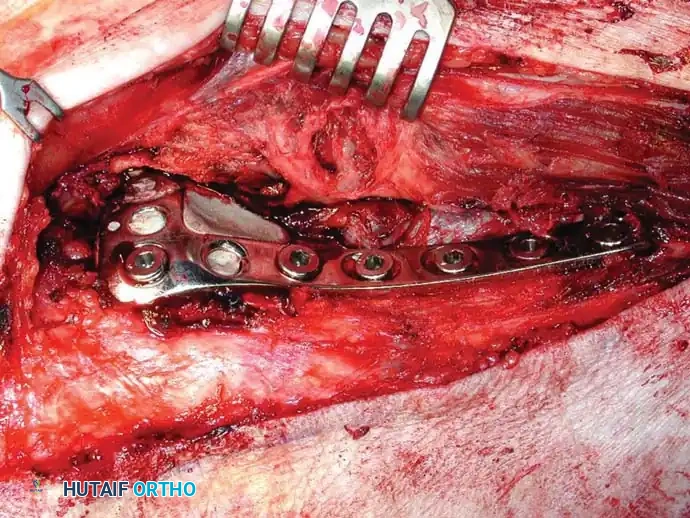
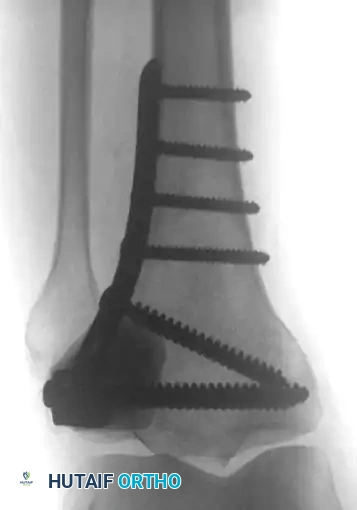
Fig. 22-25: Intraoperative sequence demonstrating extended curettage of a metastatic lesion, preparation of the cavitary defect, placement of a structural locking plate, and augmentation with PMMA bone cement to ensure immediate construct stability.
2. Endoprosthetic Reconstruction
If intramedullary nailing combined with cement cannot provide the stability required for immediate full weight-bearing (e.g., massive cortical destruction, peri-articular lesions), wide resection and endoprosthetic reconstruction are indicated.
Lesions of the femoral neck or extensive destruction of the proximal femur should be treated with hemiarthroplasty or total hip arthroplasty (THA) using a calcar-replacement or proximal femoral replacement prosthesis.
Surgical Warning: Arthroplasty components in the oncologic setting must be fixed with PMMA cement. Biologic (uncemented) ingrowth relies on viable host bone, which is often compromised by the tumor and will be further impaired by postoperative radiation therapy.
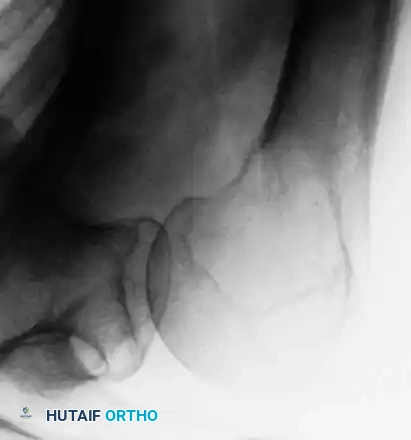
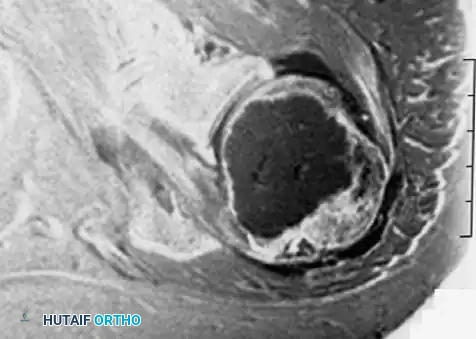
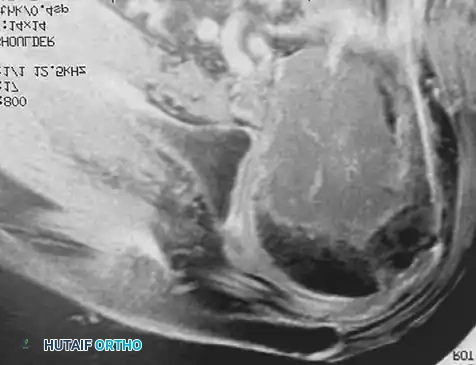
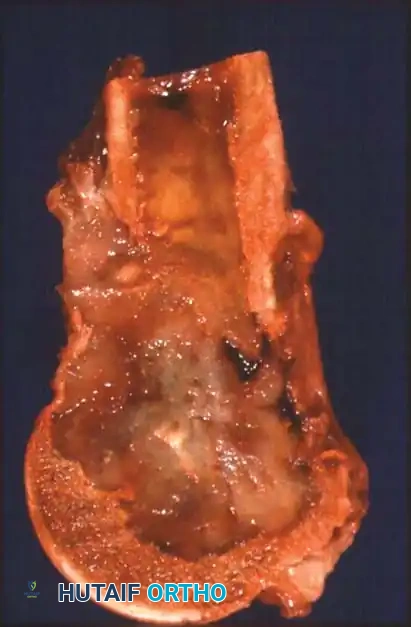
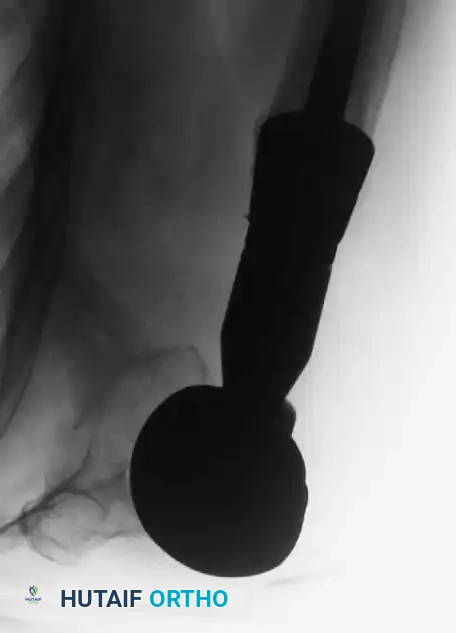
Fig. 22-21: (A) Osteonecrosis of the femoral head following radiation therapy for metastatic breast cancer. (B) Treatment with a cemented total hip arthroplasty. Because the host bone had been irradiated, both the femoral and acetabular components were cemented to ensure immediate and durable fixation.
For distal femoral lesions with extensive joint involvement, a distal femoral replacement (megaprosthesis) is utilized.
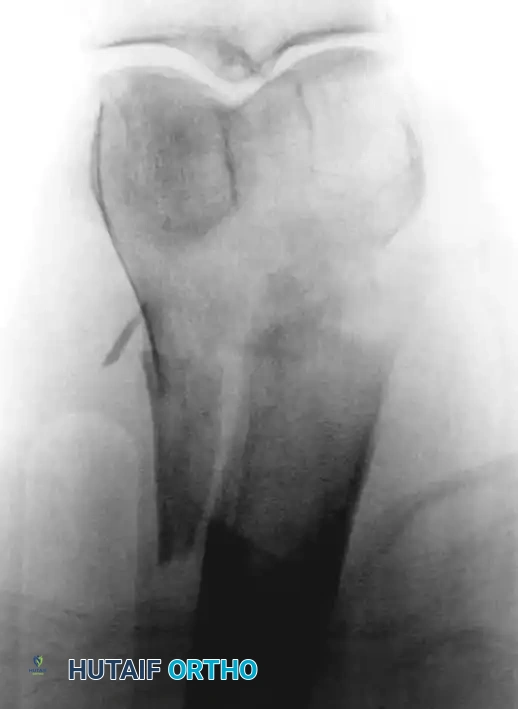
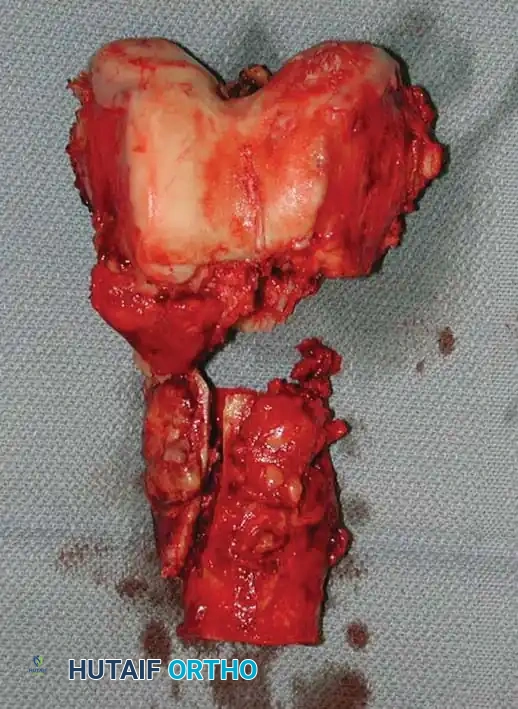
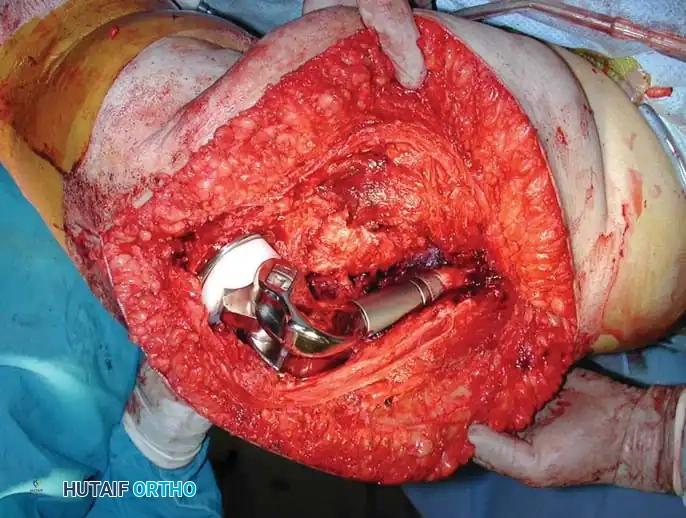
Fig. 22-26: (A) Pathological fracture of the right distal femur secondary to hepatobiliary carcinoma. Due to extensive bone destruction, endoprosthetic reconstruction was chosen. (B) Resected bone fragments showing massive tumor infiltration. (C) Distal femoral replacement prosthesis in situ, allowing immediate full weight-bearing.
3. Acetabular and Pelvic Reconstruction
Metastatic destruction of the acetabulum presents a complex biomechanical challenge. The Harrington classification is often used to guide reconstruction. For significant cavitary defects with medial wall or column compromise, the lesion is curetted, and the structural integrity of the pelvis is rebuilt using threaded Steinmann pins or an acetabular reinforcement cage, heavily augmented with PMMA.
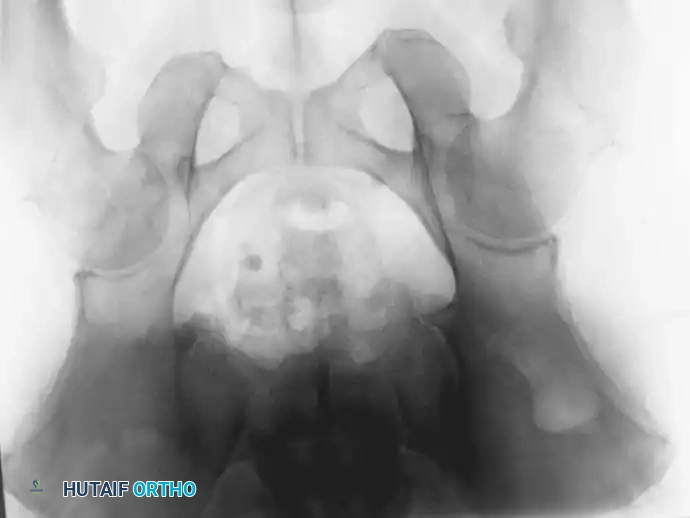
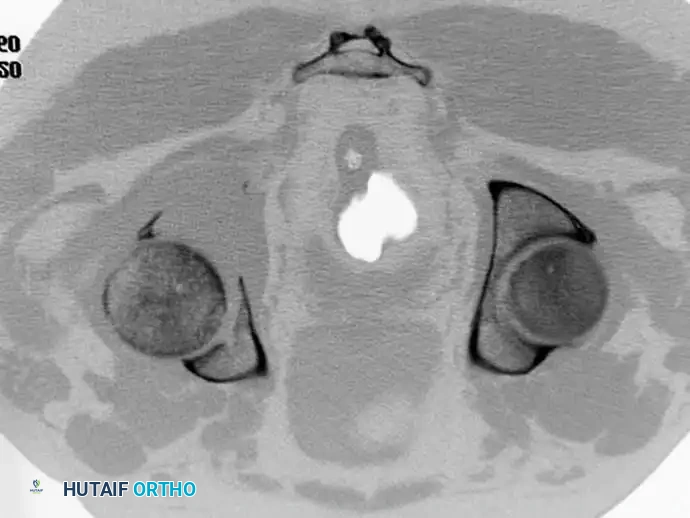
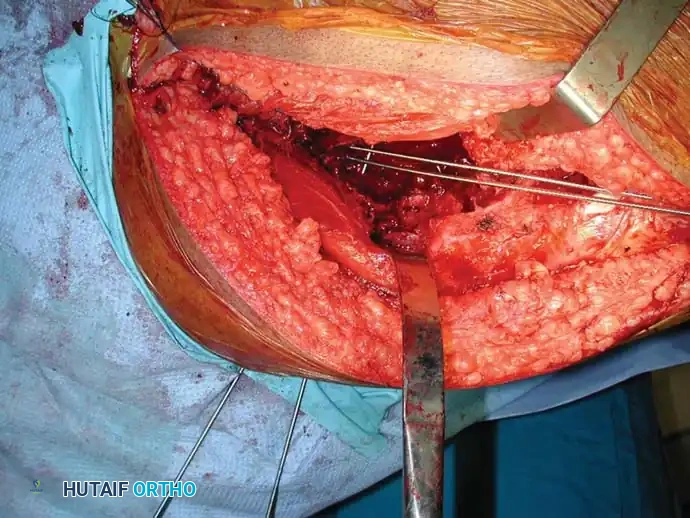
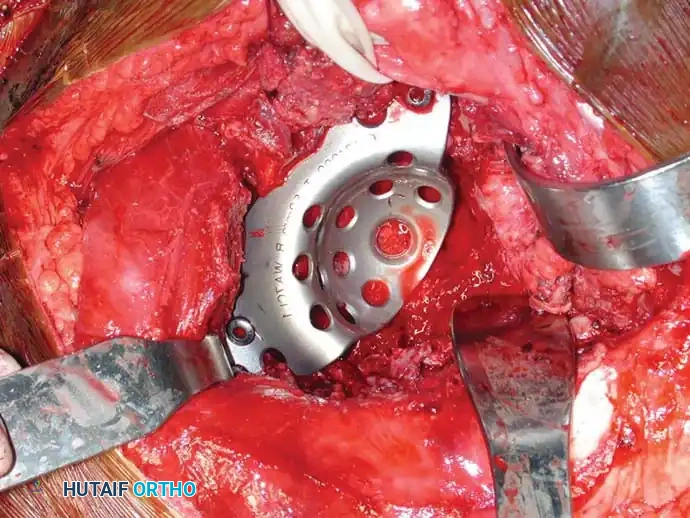
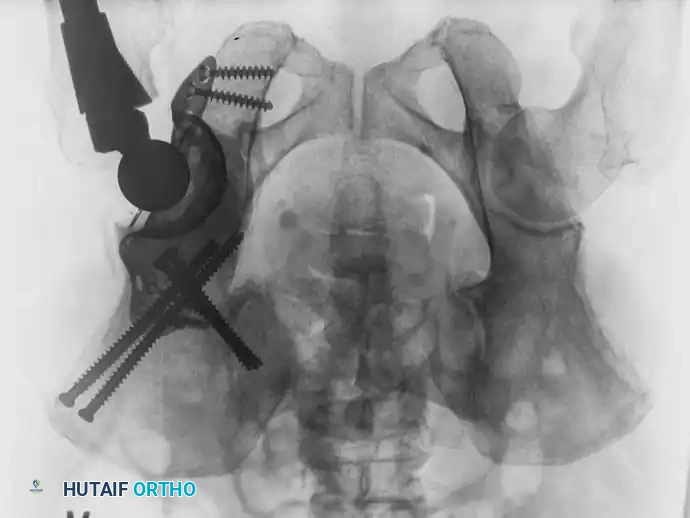
Fig. 22-23: (A, B) Radiograph and CT of a 66-year-old man with metastatic lung cancer destroying the right acetabular posterior column and wall. Preoperative embolization was performed to minimize blood loss. (C) Intraoperative view after curettage. Guidewires were placed from the defect to the posterior ilium and anterior iliac crest. Cannulated screws were placed over the wires to support the cement mantle. (D) An acetabular cage was placed, and the defect was filled with PMMA as the cup was cemented. (E) Postoperative radiograph demonstrating a stable construct.
4. Curettage and Cementation of Distal Extremity Lesions
For lesions distal to the knee or elbow (e.g., distal tibia, ankle), where intramedullary nailing or massive endoprostheses are less feasible, extended curettage followed by defect packing with PMMA is highly effective. This technique provides immediate structural support and excellent pain relief.
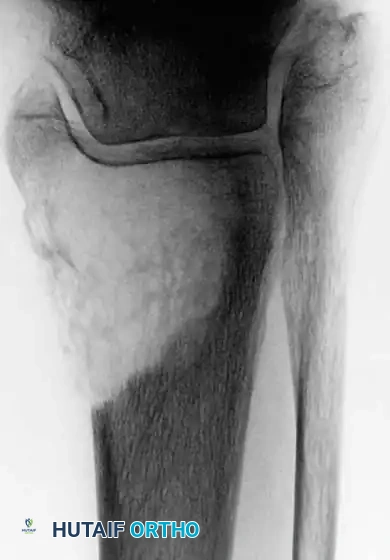
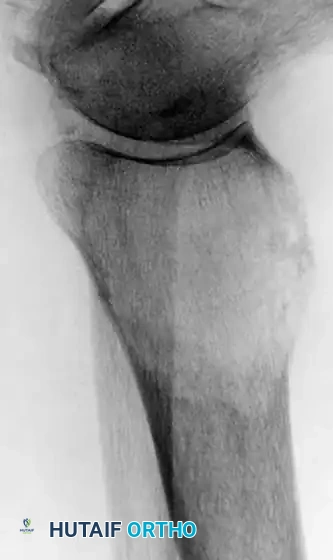
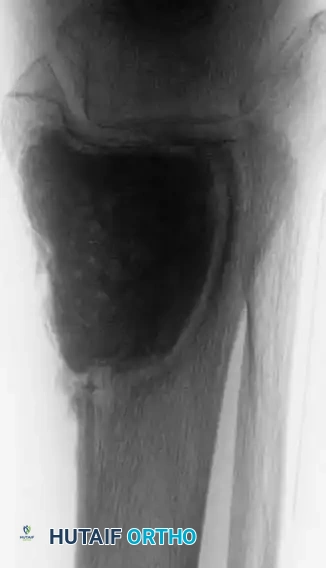

Fig. 22-27: (A, B) Anteroposterior and lateral radiographs of the left ankle of a 78-year-old woman with metastatic kidney cancer. (C, D) Postoperative views after extended curettage and packing of the defect with methacrylate. The patient resumed immediate full weight-bearing with complete pain relief.
Postoperative Protocols and Multidisciplinary Care
Surgical intervention is only one pillar of the multimodal treatment of metastatic carcinoma. Postoperative care must be meticulously coordinated with the oncology team.
Radiation Therapy
Most symptomatic bone metastases are highly responsive to radiation therapy, which helps achieve local
📚 Medical References
- metastatic carcinoma to the shoulder girdle region: indications, preoperative evaluation, surgical technique, and results, J Surg Oncol 77:105, 2001.
- Wutschert R, Bounameaux H: Determination of amputation level in ischemic limbs: reappraisal of the measurements of TcPo 2 , Diabetes Care 20:1315, 1997.
- Wyss CR, Harrington RM, Burgess EM, et al: Transcutaneous oxygen tension as a predictor of success after an amputation, J Bone Joint Surg 70A:203, 1988.
- Yowler CJ, Mozingo DW, Ryan JB, et al: Factors contributing to delayed extremity amputation in burn patients, J Trauma 45:522, 1998.
