NONOPERATIVE MANAGEMENT OF THE SPASTIC UPPER EXTREMITY
The management of the spastic upper extremity, most commonly seen in the context of cerebral palsy (CP) or post-traumatic brain injury, requires a highly nuanced, multidisciplinary approach. The primary goals of intervention are to improve the patient's functional capacity, facilitate activities of daily living (ADLs), enhance hygiene, and optimize cosmetic appearance.
Historically, the paradigm of care relied heavily on prolonged nonoperative modalities. However, modern orthopaedic consensus recognizes that while nonoperative management is foundational, it serves primarily to delay the onset of fixed myostatic contractures, optimize the patient for eventual surgical reconstruction, and maximize the efficacy of postoperative rehabilitation.
Orthotic Management and Splinting
Traditionally, early and aggressive splinting was universally employed to prevent the development of fixed contractures in spastic muscles and joints. The biomechanical rationale was to provide a prolonged, low-load stretch to the hypertonic flexor-pronator muscle groups, thereby maintaining sarcomere length and preventing capsular contracture.
However, contemporary pediatric orthopaedic practice has largely evolved away from continuous splinting protocols. Many surgeons have now abandoned rigid, full-time splinting methods for several evidence-based reasons:
1. Rarity of Early Fixed Contractures: True fixed myostatic contractures rarely occur in very young children. The deformities observed are predominantly dynamic, driven by upper motor neuron hypertonia rather than structural shortening.
2. Physiological Relaxation During Sleep: During sleep, particularly in the rapid eye movement (REM) phase, the upper extremity often becomes completely relaxed and supple due to decreased reticulospinal tract activity. This physiological relaxation effectively obviates the need for rigid night splinting.
3. Patient Compliance and Functional Inhibition: Daytime splinting is inherently cumbersome. In an active child, a rigid orthosis restricts exploratory play, inhibits sensory feedback, and is frequently rejected. Furthermore, immobilizing a limb can exacerbate "learned nonuse," a phenomenon where the child neurologically ignores the affected extremity.
Clinical Pearl: If splinting is deemed necessary—typically to prevent skin maceration in severe flexion deformities or as a postoperative adjunct—it must be meticulously fabricated. A well-formed splint must be devoid of pressure points to prevent ischemic ulceration. It should hold the wrist in as much extension as tolerated (ideally 20° to 30°), with the metacarpophalangeal (MCP) and interphalangeal (IP) joints in almost complete extension, and the thumb positioned out of the palm in palmar abduction.
Recent advancements have highlighted the utility of dynamic, functional orthoses. A functional orthosis that provides dynamic wrist extension and thumb abduction, while allowing active digital flexion, has been empirically found to improve measured hand function and encourage spontaneous use of the extremity.
Hand Therapy and Rehabilitation
Hand therapy is a cornerstone of both nonoperative management and postoperative recovery. It is critical to understand the physiological limitations of therapy: it is rarely successful in training a child to consciously relax spastic muscles. The upper motor neuron lesion precludes normal reciprocal inhibition.
Instead, the true value of hand therapy lies in:
* Strengthening Antagonists: Targeted exercises to strengthen weakened, nonspastic antagonist muscles (e.g., the extensor compartment).
* Reflex Control: Utilizing neurodevelopmental techniques to control exaggerated reflexes and abnormal synergistic movement patterns.
* Preoperative Evaluation: Serial assessments by a specialized hand therapist are invaluable in evaluating patients for surgical procedures, determining the degree of voluntary motor control, and assessing cognitive readiness for postoperative rehabilitation.
* Family Support: Providing critical support and education to the patient and family in managing the chronic nature of the disorder.
Neuromodulation: Electrical Stimulation
Neuromuscular electrical stimulation (NMES) aimed at strengthening nonspastic but weak extensor compartment muscles has a defined, albeit controversial, role in nonoperative management. The physiological goal is to recruit Type II muscle fibers in the weakened extensors to counteract the dominant spastic flexors.
Previous literature has yielded conflicting results regarding its long-term efficacy. While functional improvement has been reported with the combined use of electrical stimulation and dynamic splinting, the primary limitation is sustainability. Because the underlying neurological deficit remains unchanged, a lifelong application of the NMES program is necessary to maintain any gained strength or functional improvement. Consequently, it is often utilized as a temporary adjunct or a preoperative conditioning tool rather than a definitive solution.
Chemodenervation: Botulinum Toxin Type A
Interest in the use of botulinum toxin type A (BoNT-A) in the treatment of the spastic upper extremity has increased exponentially. BoNT-A acts by cleaving the SNAP-25 protein within the SNARE complex, thereby inhibiting the presynaptic release of acetylcholine at the neuromuscular junction and producing a reversible, dose-dependent chemical denervation.
Decreasing spasticity in the dominant flexor-pronator groups helps to improve the control of movement patterns through a combination of:
1. Pharmacological Lengthening: Allowing the spastic muscle groups to stretch.
2. Postural Improvement: Correcting the resting posture of the wrist and digits.
3. Antagonist Strengthening: Providing a "window of opportunity" for therapy to strengthen the antagonistic extensor muscles without fighting severe hypertonia.
Several studies have demonstrated highly promising short-term results, particularly in improving hygiene, easing splint application, and enhancing specific ADLs. However, the results are inherently temporary, typically lasting 6 to 9 months as the nerve terminals sprout new endings and re-establish neuromuscular transmission.
Surgical Warning: BoNT-A is highly effective for dynamic spasticity but is completely ineffective for fixed myostatic contractures. It is frequently used as a diagnostic tool to differentiate between the two prior to surgical intervention.
Long-term follow-up studies are ongoing to determine if repeated injections lead to sustained functional improvement or if patients develop neutralizing antibodies (resistance) or allergic complications. The most common reasons for the failure of BoNT-A therapy include:
* Unrecognized fixed joint contractures.
* Absence of selective motor control in the antagonist muscles.
* Profound sensory impairment (astereognosis).
* Deep-seated learned nonuse.
TRANSITION TO OPERATIVE MANAGEMENT: SURGICAL RECONSTRUCTION
When nonoperative modalities have been exhausted, or when fixed contractures and severe dynamic deformities impede function and hygiene, surgical reconstruction is indicated. The classic presentation of the spastic upper extremity includes a severe flexion and pronation contracture of the wrist, a thumb-in-palm deformity, and profoundly weak wrist extension.
Preoperative Clinical Assessment
Careful preoperative evaluation is paramount. The surgeon must differentiate between dynamic spasticity and fixed contractures, assess voluntary motor control, and evaluate the patient's sensibility.
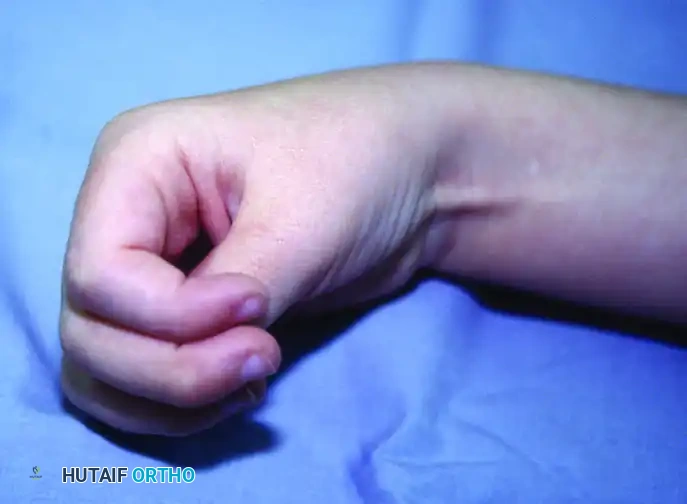
FIGURE 72-3 A: Preoperative clinical photograph demonstrating a severe flexion and pronation contracture of the wrist and forearm. The flexor carpi ulnaris (FCU) and pronator teres are typically the primary deforming forces.
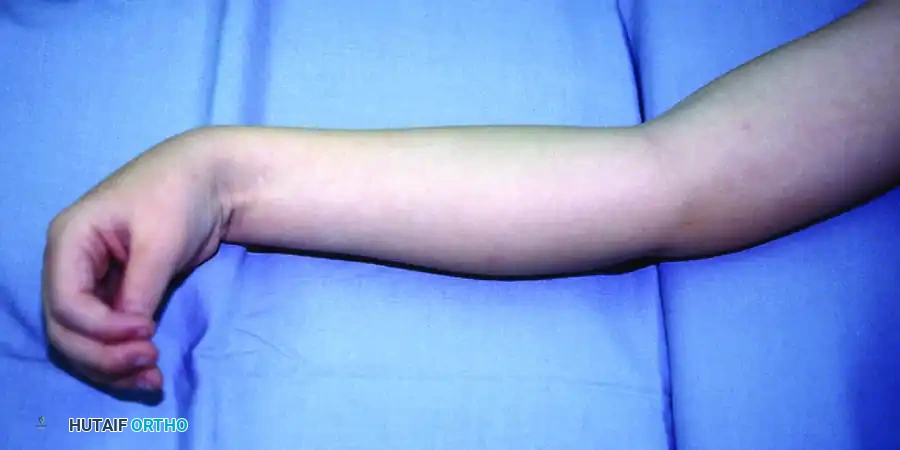
FIGURE 72-3 B: Preoperative presentation of a severe thumb-in-palm deformity. This is often driven by spasticity of the adductor pollicis, flexor pollicis longus (FPL), and the intrinsic thenar musculature, combined with weakness of the abductor pollicis longus (APL) and extensor pollicis brevis/longus (EPB/EPL).
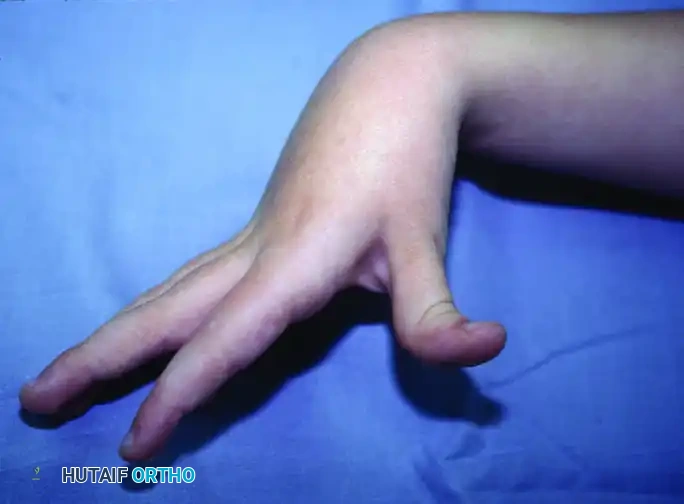
FIGURE 72-3 C: Demonstration of weak wrist extension. The patient is unable to actively extend the wrist past neutral due to the overpowering spasticity of the volar flexors and the inherent weakness of the extensor carpi radialis longus and brevis (ECRL/ECRB).
Step-by-Step Surgical Technique
The surgical reconstruction of the spastic hand is highly individualized but frequently involves a combination of fractional flexor lengthening, tendon transfers, and intrinsic releases. The procedure detailed below addresses the deformities shown in the preoperative figures.
1. Fractional Lengthening of the Volar Flexors
To address the flexion contracture without sacrificing grip strength entirely, fractional (aponeurotic) lengthening is preferred over Z-lengthening or complete tenotomy.
* Approach: A longitudinal volar incision is made over the distal forearm.
* Technique: The musculotendinous junctions of the flexor digitorum superficialis (FDS), flexor digitorum profundus (FDP), and flexor carpi radialis (FCR) are identified. Transverse incisions are made through the tendinous aponeurosis while meticulously preserving the underlying muscle fibers.
* Correction: The wrist and fingers are passively extended, allowing the muscle fibers to slide and lengthen the musculotendinous unit by 1 to 2 centimeters.
2. Flexor Carpi Ulnaris (FCU) to Extensor Carpi Radialis Longus (ECRL) Transfer
The FCU is often the strongest spastic muscle and the primary deforming force causing wrist flexion and ulnar deviation. Transferring it to the ECRL removes this deforming force and repurposes it to provide robust wrist extension (the Green transfer).
* Harvest: The FCU is detached from its insertion on the pisiform. It is mobilized proximally to the proximal third of the forearm. The ulnar nerve and artery, which lie immediately deep and radial to the FCU, must be carefully protected.
* Routing: The FCU tendon is routed subcutaneously around the ulnar border of the forearm to the dorsal aspect of the wrist.
* Insertion: The ECRL tendon is identified at the base of the second metacarpal. A Pulvertaft weave is utilized to interlace the FCU into the ECRL.
* Tensioning: The transfer is tensioned with the wrist held in approximately 30° to 45° of extension and the forearm in neutral rotation.
Surgical Pitfall: Over-tensioning the transfer can lead to a fixed extension contracture, while under-tensioning will result in a failure to achieve active wrist extension. The resting cascade of the hand must be carefully observed during tensioning.
3. Thumb-in-Palm Reconstruction
Correcting the thumb-in-palm deformity is critical for restoring pinch kinematics and allowing the hand to open to grasp objects.
* Intrinsic Release: Through a palmar incision parallel to the thenar crease, the origin of the adductor pollicis is released from the third metacarpal. If necessary, the deep head of the flexor pollicis brevis (FPB) and the first dorsal interosseous are also released.
* Extensor Augmentation: To actively pull the thumb out of the palm, the brachioradialis (BR) or palmaris longus (PL) can be transferred to the abductor pollicis longus (APL) or extensor pollicis longus (EPL). Alternatively, the EPL can be rerouted out of Lister's tubercle to provide a more direct abduction vector.
Postoperative Protocol and Outcomes
Postoperatively, the upper extremity is immobilized in a long-arm cast with the elbow at 90° of flexion, the forearm in supination, the wrist in 30° to 40° of extension, and the thumb in wide palmar abduction.
Immobilization is maintained for 4 weeks to allow for secure tendon healing at the transfer sites. Following cast removal, a custom thermoplastic splint is fabricated, and an intensive, structured hand therapy program is initiated. Therapy focuses on cortical re-education, teaching the patient to fire the transferred FCU to achieve wrist extension.
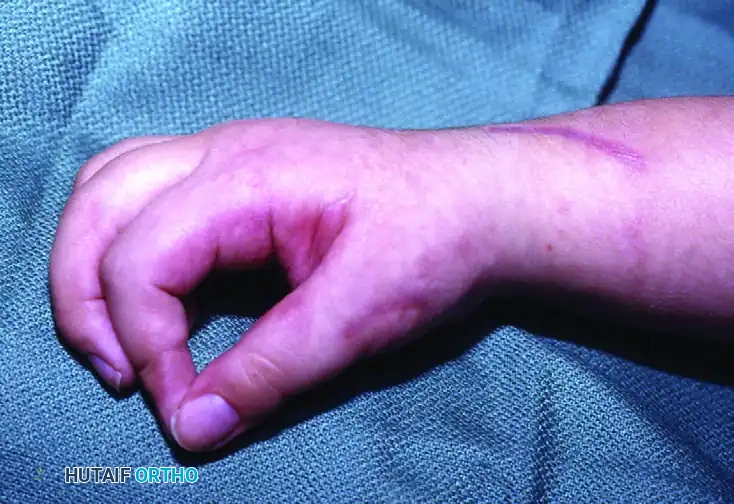
FIGURE 72-3 D: Postoperative clinical result following fractional lengthening of the flexors, FCU-to-ECRL tendon transfer, and thumb-in-palm reconstruction. The patient now demonstrates active wrist extension, a corrected resting posture, and the ability to perform functional pinch kinematics. The dorsal surgical scar from the tendon transfer routing is visible.
The ultimate success of operative management relies heavily on meticulous surgical execution, appropriate patient selection (ensuring adequate cognitive function and voluntary motor control), and rigorous postoperative rehabilitation. When executed correctly, these reconstructive procedures profoundly enhance the functional independence and quality of life for patients with a spastic upper extremity.
