Comprehensive Introduction and Patho-Epidemiology
Metacarpal shaft and neck fractures represent a substantial proportion of orthopedic hand trauma, accounting for approximately 30% to 40% of all hand fractures. These injuries exhibit a bimodal distribution, predominantly affecting young, active males secondary to high-energy trauma, sports-related injuries, or interpersonal violence, and older individuals due to low-energy falls on osteoporotic bone. The fifth metacarpal is overwhelmingly the most frequently injured, often manifesting as the classic "boxer's fracture" at the metacarpal neck, though shaft fractures across all rays are routinely encountered in high-volume trauma centers. Understanding the epidemiological patterns is paramount, as the mechanism of injury directly informs the soft tissue envelope's status, the fracture pattern's energy, and the subsequent surgical decision-making process.
The pathophysiology of metacarpal displacement is dictated by the intricate balance of the hand's intrinsic and extrinsic musculature. The interosseous muscles, originating on the metacarpal shafts, exert a strong volar and proximal deforming force. When a fracture occurs, this muscular pull, combined with the extrinsic flexor forces, predictably results in apex-dorsal angulation and longitudinal shortening. Furthermore, the asymmetric pull can induce rotational malalignment. Rotational deformity is the most functionally devastating consequence of a mismanaged metacarpal fracture; a mere 5 degrees of malrotation at the metacarpal base can amplify to 1.5 centimeters of digital overlap at the fingertips, severely compromising the functional cascade of composite digital flexion and grip strength.
Historically, the surgical management of displaced metacarpal fractures relied heavily on open reduction and internal fixation (ORIF) utilizing dynamic compression plates, mini-fragment plates, and lag screws. While ORIF provides absolute stability and permits primary bone healing, the literature unequivocally demonstrates a high morbidity profile associated with this approach in the hand. Plate and screw fixation carries a complication rate approaching 42%, primarily driven by extensive periosteal stripping, extensor tendon adhesions, hardware prominence, and profound postoperative joint stiffness. The dorsal surgical approach required for plating inevitably violates the delicate gliding layers of the extensor apparatus, often leading to a protracted and painful rehabilitation course with suboptimal functional recovery.
Consequently, the paradigm of metacarpal fracture management has profoundly shifted toward minimally invasive techniques, culminating in intramedullary (IM) pinning emerging as the contemporary gold standard for transverse, short oblique, and neck fractures. Pioneered by Foucher as "bouquet osteosynthesis" and subsequently refined, IM pinning leverages the principles of relative stability and secondary bone healing via callus formation. This technique provides robust three-point biomechanical stability while meticulously preserving the fracture hematoma and the critical periosteal blood supply. By minimizing soft tissue disruption, IM pinning drastically reduces the incidence of tendon adhesions, facilitates immediate postoperative mobilization, and optimizes the restoration of the hand's functional cascade, representing a critical advancement in operative hand trauma.
Detailed Surgical Anatomy and Biomechanics
A profound mastery of metacarpal osteology is the foundation of successful intramedullary fixation. The metacarpal bones are tubular structures exhibiting a natural dorsal convexity and a volar concavity. The medullary canal is not a uniform cylinder; it features a distinct diaphyseal isthmus that flares proximally at the metaphyseal base and distally at the metacarpal head. The intramedullary pinning technique explicitly capitalizes on this anatomical bow and the varying canal diameters. By introducing a pre-bent Kirschner wire (K-wire) through the proximal base and advancing it into the distal metaphysis, the wire must navigate the isthmus and engage the endosteal cortex, creating a dynamic, tensioned construct that resists bending and torsional forces.
The vascular anatomy of the metacarpals dictates the necessity of minimally invasive approaches. The primary blood supply is derived from the nutrient artery, which enters the volar aspect of the proximal third of the shaft, supplemented by a rich periosteal network fed by the dorsal metacarpal arteries. Open plating techniques inherently devitalize this periosteal plexus through extensive subperiosteal elevation. Intramedullary pinning, conversely, is an endosteal tissue-sparing procedure. It preserves the vital periosteal sleeve and the fracture hematoma—a rich milieu of osteoprogenitor cells, cytokines, and growth factors essential for robust endochondral ossification and rapid clinical union.
Neurologically and tendinously, the dorsal aspect of the hand presents a dense and vulnerable surgical landscape. The dorsal sensory branches of the ulnar nerve (DSBUN) and the superficial branch of the radial nerve (SBRN) arborize extensively over the proximal metacarpal bases. Iatrogenic injury to these nerves during portal creation can result in debilitating neuromas or complex regional pain syndrome (CRPS). Furthermore, the extensor digitorum communis (EDC) tendons, extensor indicis proprius (EIP), and extensor digiti minimi (EDM) glide directly over the metacarpal periosteum, stabilized by the sagittal bands at the metacarpophalangeal (MCP) joints. The surgical entry portals for IM pinning must be meticulously planned to navigate between these tendinous and neural structures, utilizing blunt dissection to establish a safe trajectory into the osseous base.
Biomechanically, intramedullary pinning operates on the principle of three-point elastic fixation. When a pre-bent K-wire is inserted into the canal, the apex of the bend contacts the endosteal surface of the dorsal cortex, while the proximal and distal ends of the wire exert opposing forces against the volar cortex. This internal tension stabilizes the fracture against the apex-dorsal deforming forces of the intrinsic muscles. Rotational stability, while historically considered a weakness of single-wire IM fixation, is achieved through a combination of maximizing canal fill (using the largest diameter wire the isthmus will accommodate), securing the wire tip firmly into the dense subchondral bone of the metacarpal head, and the interdigitation of the fracture fragments once anatomically reduced. In highly unstable patterns, multiple smaller wires (bouquet technique) can be utilized to enhance rotational and angular control.
Exhaustive Indications and Contraindications
The decision to proceed with operative intervention for metacarpal fractures requires a nuanced synthesis of radiographic parameters, clinical deformity, and patient-specific functional demands. The overarching goal is the restoration of the hand's transverse and longitudinal arches, ensuring a normal digital cascade without scissoring or rotational overlap. While many metacarpal fractures are amenable to non-operative management with functional bracing or intrinsic-plus cast immobilization, specific thresholds of displacement mandate surgical correction to prevent long-term biomechanical dysfunction and grip strength deficits.
Surgical indications are strictly defined by the anatomic location and the degree of deformity. For metacarpal neck fractures, the acceptable degree of apex-dorsal angulation increases from the radial to the ulnar side of the hand, reflecting the compensatory mobility of the carpometacarpal (CMC) joints. Angulation exceeding 10–15 degrees in the index and long fingers, 30–40 degrees in the ring finger, or 40–50 degrees in the small finger constitutes a clear indication for IM pinning. For metacarpal shaft fractures, tolerances are drastically lower; shortening greater than 2 to 3 millimeters or any clinically detectable rotational malalignment dictates surgical intervention. Furthermore, multiple concurrent metacarpal fractures inherently destabilize the structural arch of the hand, making them prime candidates for IM fixation to restore global hand architecture.
Despite its versatility, intramedullary pinning is not universally applicable and carries absolute and relative contraindications. Intra-articular fractures extending into the MCP or CMC joints with step-off or incongruity cannot be managed solely with IM wires; these require anatomic open reduction and absolute stability via interfragmentary lag screws to prevent post-traumatic arthrosis. Severe comminution of the metacarpal shaft represents another critical contraindication. IM pinning relies on cortical contact for length maintenance and rotational stability; in highly comminuted patterns, the lack of cortical buttressing will lead to inevitable secondary shortening and telescoping over the wire, necessitating bridging plates or external fixation constructs instead.
| Clinical Parameter | Indications for IM Pinning | Contraindications for IM Pinning |
|---|---|---|
| Fracture Location | Metacarpal neck, transverse shaft, short oblique shaft | Intra-articular extension (MCP or CMC joint) |
| Angulation (Neck) | >15° (Index/Long), >40° (Ring), >50° (Small) | Minimal angulation amenable to closed reduction |
| Shortening (Shaft) | > 2-3 mm | Severe segmental bone loss |
| Rotational Deformity | Any clinical malrotation (scissoring/overlap) | None |
| Fracture Pattern | Simple 2-part, minimal comminution | Severe butterfly comminution (lack of cortical contact) |
| Soft Tissue Envelope | Closed injuries, Grade I/II open fractures | Active deep space infection, severe crush requiring flaps |
Special consideration must be given to the physiological demands and compliance of the patient. Open fractures, particularly those requiring serial debridement or complex soft tissue coverage, benefit greatly from the low-profile nature of IM implants, which do not interfere with wound healing or flap adherence unlike bulky dorsal plates. However, in patients with active localized infections, such as those presenting late with untreated human bite wounds ("fight bites"), the introduction of hardware into the medullary canal is absolutely contraindicated until the infection is definitively eradicated through serial arthrotomies and targeted antimicrobial therapy.
Pre-Operative Planning, Templating, and Patient Positioning
Meticulous preoperative planning is the linchpin of successful intramedullary fixation, beginning with a rigorous clinical and radiographic assessment. The clinical examination must focus on the digital cascade during composite active flexion; any deviation of the fingertips from pointing toward the scaphoid tubercle indicates rotational malalignment. Radiographic evaluation requires true anteroposterior (AP), lateral, and oblique views of the specific injured digit, rather than generic hand films, to accurately quantify shortening and apex-dorsal angulation. The true lateral view is particularly critical for assessing the degree of volar comminution, which dictates the inherent stability of the fracture post-reduction.
Digital templating is highly recommended to determine the appropriate Kirschner wire diameter and length. The surgeon must evaluate the diameter of the medullary isthmus on the AP radiograph. The goal is to select a wire that provides maximum canal fill without causing iatrogenic cortical blowout during insertion. Typically, a 1.6 mm (0.062-inch) or 2.0 mm (0.078-inch) K-wire is optimal for the metacarpal shafts of adult males, while smaller 1.2 mm or 1.6 mm wires may be required for females or smaller digits. Planning the entry point trajectory is also crucial; the angle of approach must be shallow enough to allow the wire to glance off the endosteal cortex rather than penetrating the volar shaft.
Anesthetic considerations have evolved significantly, with Wide Awake Local Anesthesia No Tourniquet (WALANT) emerging as a transformative approach for metacarpal fixation. Utilizing a mixture of lidocaine and epinephrine provides excellent hemostasis and profound local anesthesia without the need for a tourniquet or sedation. The paramount advantage of WALANT is the ability to assess active, voluntary digital motion intraoperatively. Once the wire is placed, the surgeon can ask the patient to actively flex and extend the digits, allowing for real-time, dynamic confirmation that the rotational alignment is perfect and that the extensor mechanism is unhindered by the hardware. Alternatively, regional supraclavicular or axillary blocks provide excellent anesthesia and postoperative analgesia if WALANT is not utilized.
Patient positioning and operating room setup must be optimized for seamless fluoroscopic imaging. The patient is positioned supine with the affected extremity extended on a radiolucent hand table. If WALANT is not used, a well-padded upper arm tourniquet is applied. The mini C-arm fluoroscopy unit should be brought in parallel to the hand table, entering from the distal or lateral aspect. The monitor must be positioned directly across from the surgeon to ensure ergonomic visualization. The hand should be draped free to allow unrestricted manipulation of the digits for reduction maneuvers and cascade assessment. All necessary equipment, including heavy wire cutters, T-handle chucks, sharp awls, and a variety of pre-cut K-wires, must be readily available on the sterile field before the initial incision.
Step-by-Step Surgical Approach and Fixation Technique
Localization and Incision
The procedure initiates with precise fluoroscopic localization to minimize the surgical footprint. Utilizing the mini C-arm in the AP plane, a radiopaque instrument is used to identify the exact center of the metacarpal base, both proximally and dorsally. This location is marked with a surgical skin marker. Accuracy at this stage dictates the success of the entire trajectory; an eccentric entry point will force the wire against the lateral cortices, increasing the risk of iatrogenic fracture and malalignment.
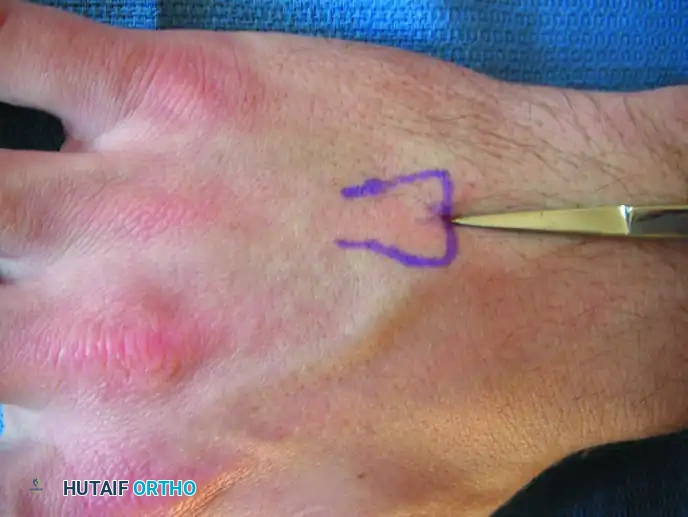
Following localization, a 0.5-cm to 1.0-cm longitudinal incision is made beginning 1.0 to 1.5 cm proximal to the fluoroscopic mark, extending distally. This proximal extension is a critical technical nuance; it allows the surgeon to lower their hand and approach the metacarpal base at a very shallow angle (typically 15 to 20 degrees relative to the shaft). A steep approach angle will drive the wire directly into the volar cortex, whereas a shallow angle facilitates smooth gliding down the medullary canal.
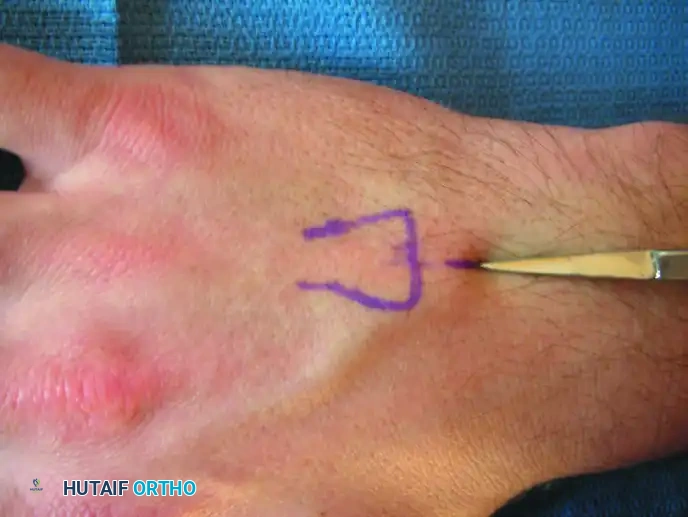
Soft Tissue Dissection and Portal Creation
Once the skin is incised, meticulous blunt dissection is mandatory. Using a small curved hemostat, the surgeon spreads longitudinally through the subcutaneous tissues. This blunt technique gently sweeps the delicate dorsal sensory branches of the ulnar or radial nerves out of the operative field. The extensor tendons are identified and retracted laterally or medially depending on the specific ray being addressed. Sharp dissection in this zone is strictly prohibited due to the high risk of catastrophic nerve or tendon laceration.
Upon visualizing the bare dorsal cortex of the metacarpal base, an entry portal must be created. A sharp awl, a pair of sharp iris scissors used as a trocar, or a power drill with a small burr can be utilized. The portal must be perfectly aligned with the longitudinal axis of the metacarpal shaft in both the AP and lateral planes. The opening should be slightly larger than the selected K-wire to prevent binding during insertion and to allow for minor trajectory adjustments.
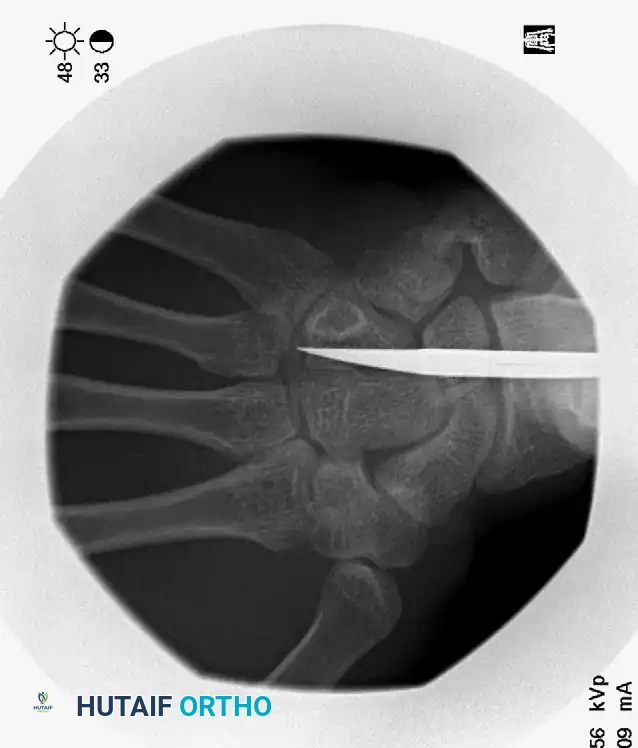
Wire Preparation
Proper preparation of the Kirschner wire is arguably the most critical step in preventing intra-articular complications. Standard K-wires feature sharp trocar or diamond tips designed to cut through bone. If advanced down the medullary canal, these sharp tips can easily pierce the subchondral bone and articular cartilage of the metacarpal head, resulting in devastating joint penetration. To mitigate this, the surgeon must use heavy wire cutters to amputate the sharp tip, creating a blunt, flat end.
After blunting the tip, the distal 5 to 10 millimeters of the wire are gently curved to an angle of approximately 15 to 20 degrees. This pre-bend serves a dual purpose. First, it allows the blunt tip to bounce off the endosteal cortices rather than penetrating them. Second, it transforms the wire into a steerable instrument; by rotating the proximal end of the wire, the surgeon can direct the curved tip to navigate past the fracture site, bypass areas of comminution, and find the distal medullary canal.
Fracture Reduction and Wire Advancement
Prior to wire passage, the fracture must be anatomically reduced. For metacarpal neck fractures, the Jahss maneuver is the gold standard: the MCP joint is flexed to 90 degrees to tighten the collateral ligaments, locking the proximal phalanx to the metacarpal head. The surgeon then applies a dorsally directed force through the proximal phalanx while providing volar counter-pressure on the metacarpal shaft, effectively correcting the apex-dorsal angulation.
The preoperative deformity must be rigorously analyzed to understand the required reduction vectors.
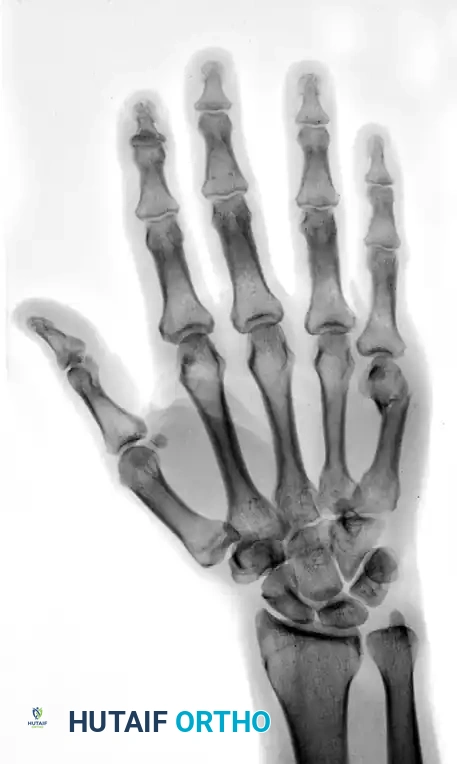
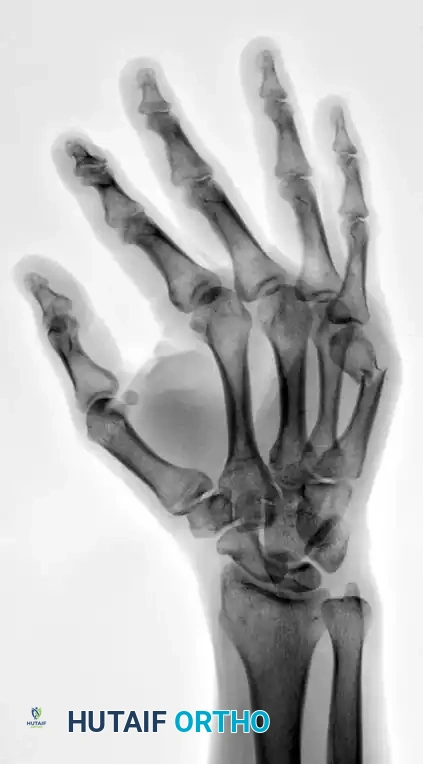
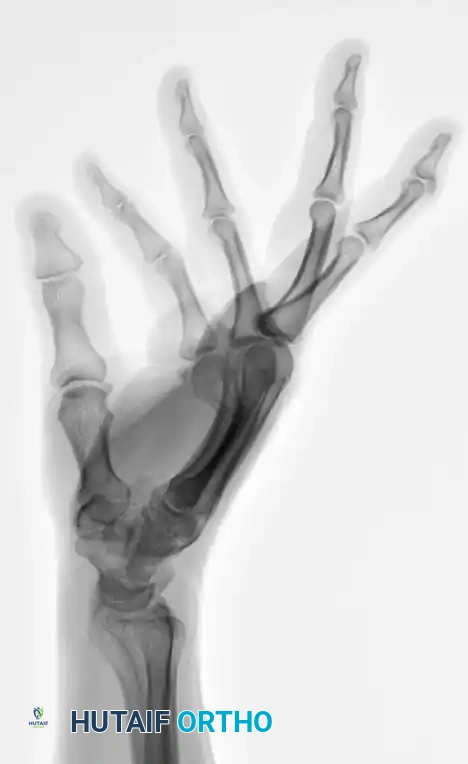
The unbent proximal end of the prepared K-wire is mounted into a manual T-handle chuck. Manual insertion is vastly superior to power driving, as it provides the surgeon with exquisite tactile feedback, allowing them to feel the wire traversing the isthmus and engaging the endosteum. The wire is advanced down the canal to the fracture site. Reduction is confirmed fluoroscopically, and the wire is steered across the fracture into the distal fragment. Live fluoroscopy is utilized to ensure the wire remains contained within the medullary canal. The blunt tip is advanced until it impacts the dense subchondral bone of the metacarpal head, seating firmly just proximal to the articular cartilage.
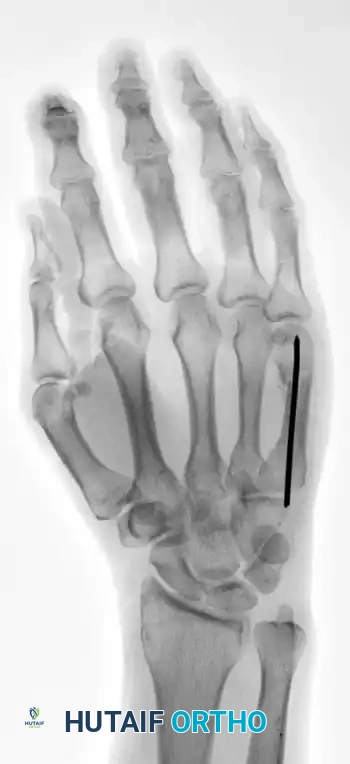
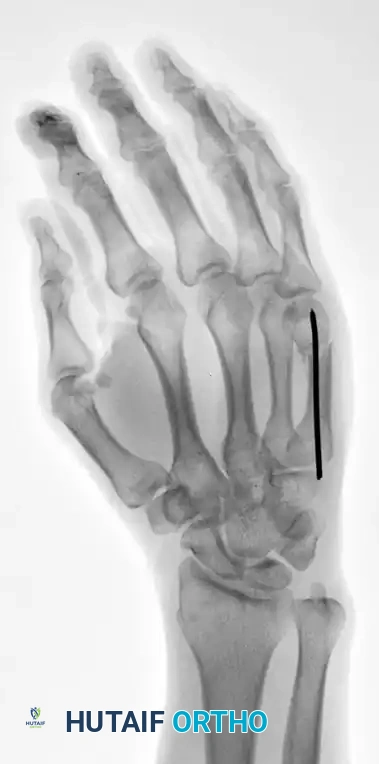
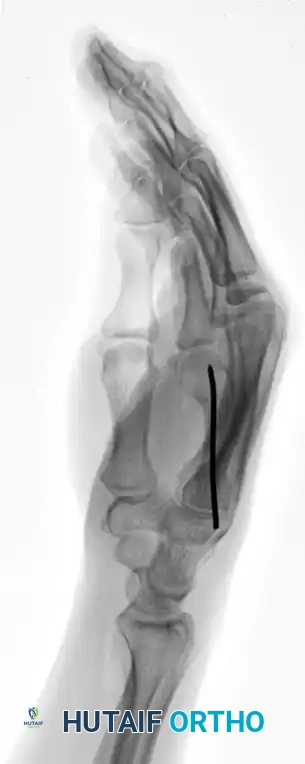
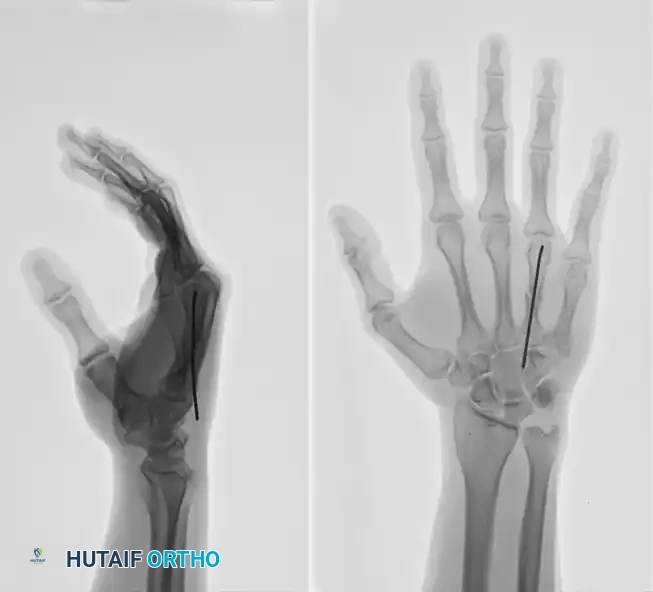
This technique demonstrates equal efficacy for mid-shaft fractures, restoring length and alignment through the same biomechanical principles, as seen in the management of the fourth metacarpal fracture below:
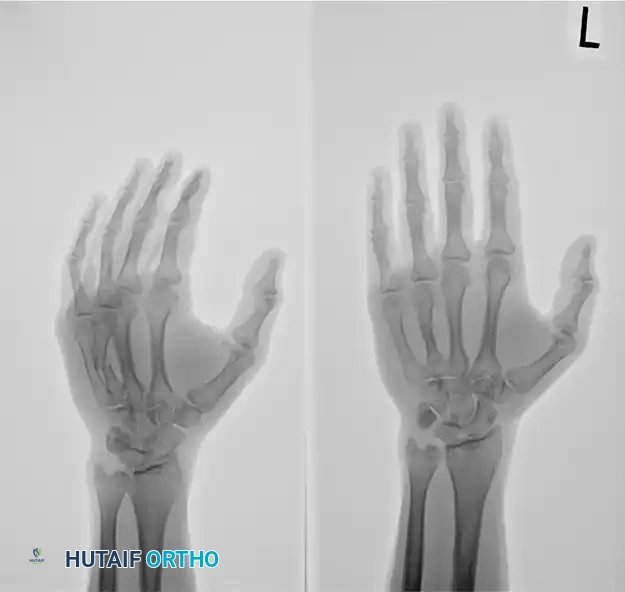
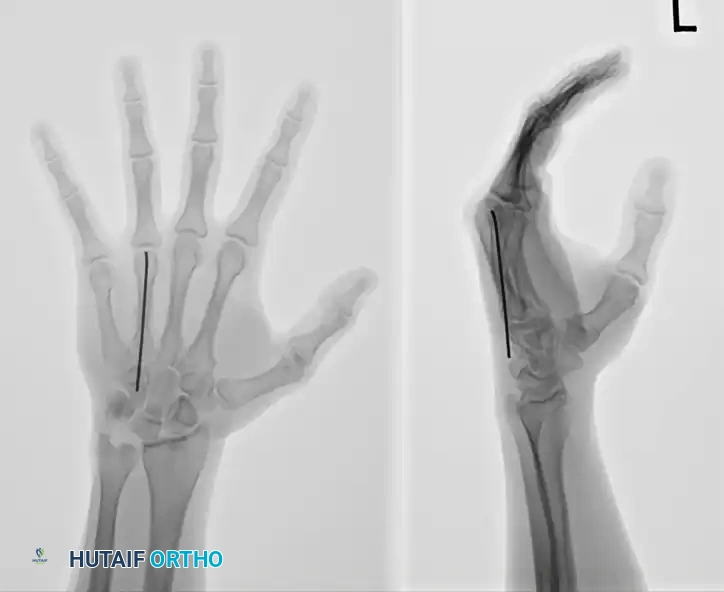
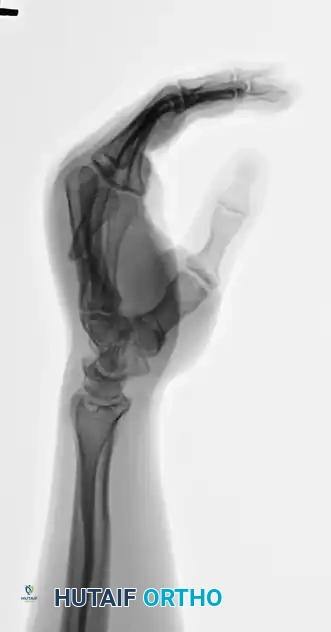
Limited Open Reduction
In a subset of cases, closed reduction is unachievable. This is typically due to the interposition of soft tissues—such as periosteum, intrinsic muscle fascia, or even extensor tendons—into the fracture site. Alternatively, the proximal fragment may have buttonholed through the surrounding musculature. If the wire cannot be passed across the fracture after two or three deliberate attempts, the surgeon must abandon closed efforts. Repeated forceful probing will inevitably lead to iatrogenic comminution, cortical blowout, and further devascularization of the fracture fragments.
When closed reduction fails, a limited open reduction is mandated. A dorsal longitudinal incision is made directly over the fracture site. The extensor mechanism is gently retracted, and the fracture hematoma is evacuated. The interposed soft tissues are carefully extracted from the fracture gap using fine forceps. Under direct vision, the fracture is anatomically reduced using reduction forceps or dental picks.
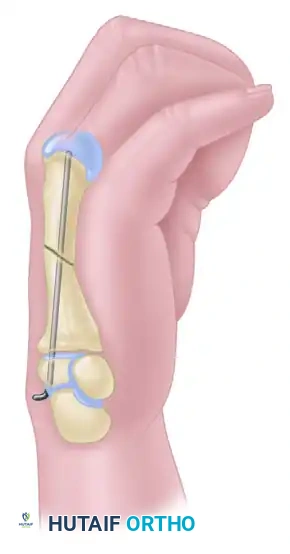
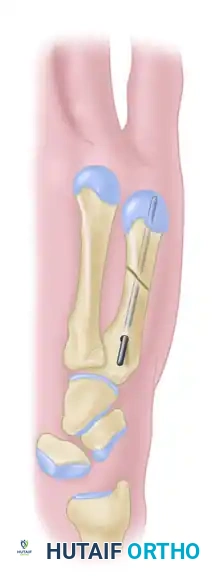
Once the anatomical reduction is visualized and held securely, the intramedullary wire is passed from the proximal fragment, across the visually reduced fracture site, and into the distal fragment. This limited open approach ensures perfect alignment without the extensive periosteal stripping associated with traditional plating.
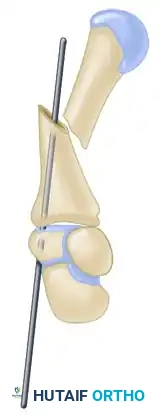
Final Seating and Closure
Following anatomical reduction and confirmation that the distal wire tip is securely anchored in the subchondral bone, the proximal end of the wire requires meticulous management. The wire must be cut flush with the proximal cortex of the metacarpal base. Utilizing a tamp, the cut end of the K-wire is gently impacted slightly deeper, ensuring it is completely buried beneath the dorsal cortex or, at minimum, deep to the periosteum and subcutaneous tissue.
Leaving the proximal wire proud is a severe technical error. The continuous gliding of the extensor tendons over a sharp, prominent wire end will invariably cause debilitating tenosynovitis, profound pain during active motion, and potentially catastrophic extensor tendon rupture. Once the wire is buried, the wound is irrigated, and the skin is closed with simple interrupted non-absorbable sutures (e.g., 4-0 Nylon). A sterile, non-adherent dressing is applied, followed by a well-padded intrinsic-plus splint to support the hand during the immediate postoperative inflammatory phase.
Complications, Incidence Rates, and Salvage Management
While intramedullary pinning significantly reduces the morbidity associated with open plating, it is not without inherent risks. The overall complication rate for IM pinning of metacarpal fractures ranges from 10% to 15%, a stark improvement over the 40% historical rate for ORIF, but still requiring vigilant postoperative management. The majority of complications are related to hardware irritation, loss of reduction, or superficial infections, most of which can be mitigated through precise surgical technique and rigorous adherence to postoperative protocols.
Hardware-related complications are the most frequently encountered. Proximal wire migration or backing out can occur if the wire does not achieve sufficient purchase in the distal subchondral bone or if the fracture remains excessively mobile. A prominent proximal wire will rapidly induce extensor tendon irritation. If wire backing out is identified radiographically and is symptomatic, the hardware must be removed once clinical union is achieved (typically at 4 to 6 weeks). In cases of impending tendon rupture, premature wire removal may be necessary, potentially requiring conversion to a functional brace if the fracture is not yet fully consolidated.
Loss of reduction, particularly rotational malalignment, represents a significant functional failure. This typically occurs in fractures with unrecognized comminution or when an undersized wire is utilized, failing to provide adequate canal fill and rotational stability. If a rotational deformity is identified in the early postoperative period (within 1-2 weeks), immediate return to the operating room for revision fixation is indicated. Delayed presentations of malunion require complex corrective osteotomies. Joint stiffness, while less common than with ORIF, can still occur, particularly if the MCP joints are immobilized in extension rather than the safe intrinsic-plus position (70-90 degrees of flexion).
| Complication
