IDIOPATHIC TRANSIENT OSTEOPOROSIS
Idiopathic transient osteoporosis (ITO) is a rare, self-limiting clinical entity characterized by the acute or subacute onset of joint pain, localized osteopenia, and bone marrow edema on magnetic resonance imaging (MRI). While the exact etiology remains elusive, it is widely considered part of the bone marrow edema syndrome (BMES) spectrum.
Demographics and Clinical Presentation
Idiopathic transient osteoporosis of the hip occurs most frequently in middle-aged men (typically in their fourth or fifth decades) and in women during the third trimester of pregnancy or the immediate postpartum period. Patients typically present with an insidious onset of increasing groin or anterior thigh pain, accompanied by an antalgic gait and progressive local muscle wasting. Range of motion is often preserved but painful at the extremes. Notably, the symptoms are bilateral in approximately one-third of patients, either occurring simultaneously or sequentially in a migratory pattern.
Diagnostic Imaging and Staging
The diagnosis of ITO relies heavily on a high index of clinical suspicion correlated with advanced imaging, as early plain radiographs are notoriously unremarkable.
- Plain Radiography: Demineralization and loss of the subchondral trabecular pattern may not become apparent on plain radiographs for up to 6 weeks after the onset of symptoms. Joint space narrowing is characteristically absent, differentiating it from inflammatory or degenerative arthritides.
- Radionuclide Bone Scan: Technetium-99m bone scintigraphy will demonstrate homogeneously increased radiotracer uptake in the affected femoral head and neck, often preceding radiographically visible osteopenia.
- Magnetic Resonance Imaging (MRI): MRI is the gold standard and is highly sensitive for early detection. However, it is imperative to recognize that other aggressive entities—such as osteonecrosis (avascular necrosis), osteomyelitis, and occult neoplasms—can mimic ITO.
Grimm et al. elegantly described three distinct phases of transient osteoporosis of the hip as visualized on MRI:
1. Diffuse Phase: Characterized by a widespread, ill-defined pattern of "bone marrow edema" (decreased signal on T1-weighted images and increased signal on T2-weighted/STIR sequences) encompassing the femoral head and extending into the femoral neck.
2. Focal Phase: Occurring 2 to 3 months after symptom onset. If the patient has been strictly non-weight-bearing, the marrow lesions retract and their signal intensity normalizes. However, if weight-bearing continues, a focal lesion persists primarily in the superior weight-bearing zone of the femoral head—the area of greatest biomechanical strain.
3. Residual Phase: Beginning 2 to 3 months after the focal stage, this phase marks clinical and radiographic resolution. The femoral neck marrow signal normalizes to that of fatty marrow, though occasionally, a thin horizontal signal (representing a healing subchondral stress response) may be seen just below the subchondral plate.
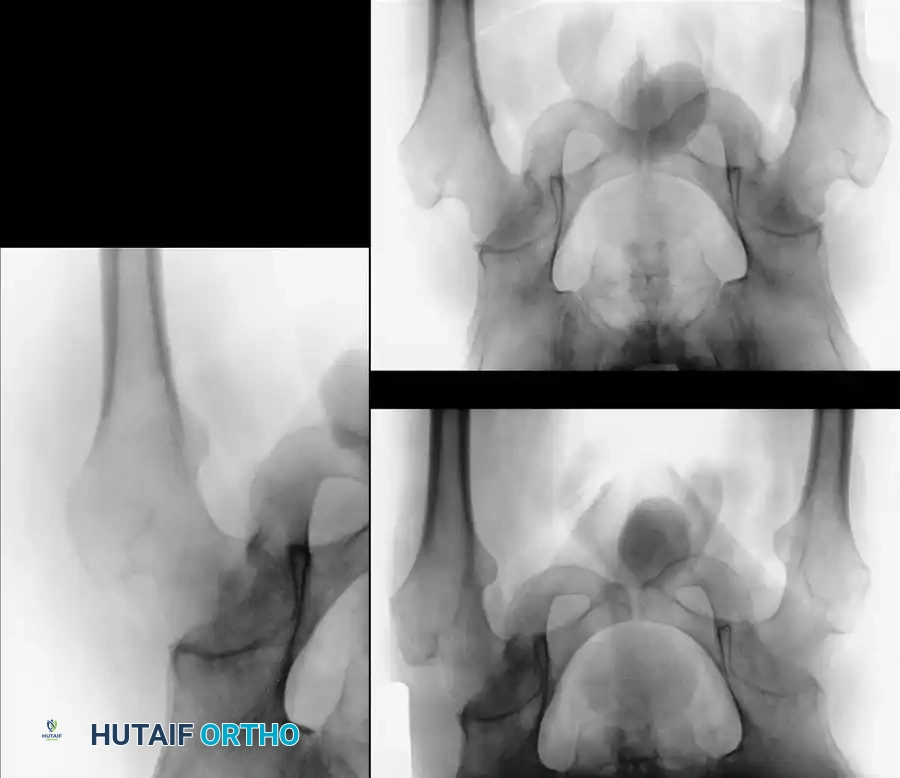
Clinical Pearl: The presence of a subchondral fracture line or a focal, well-demarcated anterosuperior lesion with a "double-line sign" on MRI is pathognomonic for osteonecrosis, not transient osteoporosis. Differentiating these two entities is the most critical step in the diagnostic algorithm.
Management Protocols
Because transient osteoporosis resolves predictably with conservative measures over 6 to 12 months, surgical intervention is rarely, if ever, indicated.
Historically, noting that the initial phase of ITO suggested severe bone marrow edema with elevated intraosseous pressure, some authors (such as Hofmann et al.) performed core decompressions, reporting immediate pain relief and rapid MRI normalization. However, contemporary orthopedic consensus strongly advises against surgical intervention.
Conservative Management Protocol:
* Protected Weight-Bearing: Immediate transition to crutches or a walker with toe-touch weight-bearing to prevent subchondral insufficiency fractures.
* Pharmacotherapy: Analgesics, nonsteroidal anti-inflammatory drugs (NSAIDs), and occasionally bisphosphonates or calcitonin to modulate bone turnover and manage pain.
* Surveillance: Serial clinical examinations and radiographs every 4 to 6 weeks until clinical resolution is achieved.
Surgical Warning: It is paramount to differentiate ITO from early-stage osteonecrosis. Performing an early core decompression mistakenly for transient osteoporosis exposes the patient to unnecessary surgical risks (e.g., iatrogenic fracture, infection) for a condition that is inherently self-limiting.
Transient Osteoporosis of the Foot and Ankle
While the hip is the most common site, transient osteoporosis can affect the ankle and foot. Gallant, Fisher, and Sziklas described this phenomenon predominantly in postmenopausal women. The clinical course mirrors that of the hip: it is benign, self-limiting, and responds excellently to conservative treatment (immobilization in a controlled ankle motion [CAM] boot and protected weight-bearing). This entity must be carefully differentiated from Complex Regional Pain Syndrome (CRPS/Reflex Sympathetic Dystrophy), which typically presents with more profound autonomic dysfunction, allodynia, and trophic skin changes.
NEUROPATHIC ARTHROPATHY (CHARCOT JOINT)
Neuropathic arthropathy, commonly referred to as a Charcot joint, is a progressive, devastating, and often painless degenerative arthropathy that develops most frequently in weight-bearing joints secondary to an underlying sensory neuropathy.
Etiology and Pathogenesis
The causes of neuropathic arthropathy are manifold. Today, the predominant global cause is diabetes mellitus. However, a thorough diagnostic workup must consider other etiologies, including:
* Leprosy and Yaws
* Congenital insensitivity to pain
* Spinal dysraphism (Spina bifida, myelomeningocele)
* Syringomyelia (classically affecting the upper extremity)
* Peripheral neuropathy secondary to alcoholism, avitaminosis, or amyloidosis
* Spinal cord or peripheral nerve injury
* Post-renal transplant arthropathy and repeated intraarticular steroid injections
Neurosyphilis (Tabes Dorsalis): Syphilis, caused by the spirochete Treponema pallidum, remains an infrequent but resurgent cause of neuropathic arthropathy. Due to inadequate treatment of primary syphilis or incidental antibiotic exposure, many patients lack classic signs. A high index of clinical suspicion and appropriate serological testing (VDRL, RPR, FTA-ABS) are mandatory.
Pathophysiology:
Two primary theories explain the pathogenesis of Charcot arthropathy, which likely act synergistically:
1. Neurotraumatic Theory: Destruction of afferent proprioceptive and nociceptive fibers leads to a loss of protective sensation. The joint is subjected to repetitive, unrecognized microtrauma, leading to ligamentous laxity, articular cartilage destruction, and periarticular fractures.
2. Neurovascular Theory: Autonomic neuropathy leads to a loss of sympathetic tone, resulting in hyperdynamic blood flow (arteriovenous shunting). This localized hyperemia stimulates osteoclastic bone resorption, leading to profound osteopenia and structural collapse.
On histological examination of the synovium from trophic joints, shards of bone and cartilage (osteochondral fragments) are seen deeply embedded within the hyperplastic synovium—a finding that is highly diagnostic of neuropathic arthropathy.
Clinical Pearl: Johnson postulated that the destruction caused by neuropathic arthropathy can often be minimized by sufficient protection after minor fractures, sprains, or effusions. These injuries incite a hyperemic inflammatory process that must be allowed to subside completely before weight-bearing is permitted. Radiographs must demonstrate bone repair rather than active resorption before loading the joint.
Management of the Charcot Foot
Even the most minor foot or ankle injury in a diabetic patient must be evaluated with extreme caution and observed repeatedly until all signs of inflammation (erythema, calor, edema) have resolved.
Nonoperative Management:
When the diagnosis of a Charcot foot is established, the cornerstone of treatment is offloading. Morgan et al. popularized the use of a total-contact orthosis, such as the Charcot Restraint Orthotic Walker (CROW). This custom-molded orthosis effectively controls limb edema, distributes plantar pressures, returns the patient to ambulatory status, and prevents significant progression of midfoot collapse (rocker-bottom deformity).
Ulcer and Infection Management:
Aggressive management of ulcerations over bony prominences (e.g., metatarsophalangeal joints, collapsed cuboid) is critical.
* Superficial ulcers are treated with minimal debridement of the crater edges and application of a total contact cast (TCC) or a well-padded short leg plaster cast.
* Extensive ulcerations, deep space abscesses, or osteomyelitis require aggressive surgical debridement. This may involve single or multiple ray resections, transmetatarsal amputations, or hyperbaric oxygen therapy.
* With meticulous debridement, appropriate antibiotic therapy, and vascular optimization, many diabetic feet that previously would have been amputated can now be salvaged.
Surgical Reconstruction of the Midfoot/Hindfoot:
Patients with trophic changes in the midfoot and hindfoot are infrequently treated surgically during the acute (Eichenholtz Stage I) phase, as aggressive debridement of hyperemic, necrotic bone only exacerbates instability.
Surgery is reserved for the coalescent or reconstructive phases (Eichenholtz Stages II/III) when marked deformity precludes brace wear and causes recurrent ulceration.
* Indications: Rigid, non-plantigrade foot, recurrent ulceration failing conservative care, severe instability.
* Technique: Midfoot or hindfoot corrective osteotomy (e.g., biplanar wedge resections) and extended arthrodesis ("superconstructs").
* Principles: Use of rigid internal fixation (beaming techniques with large-diameter solid screws or specialized Charcot plates), autogenous bone grafting, and prolonged postoperative cast immobilization (often 6 to 12 months).
Management of the Neuropathic Ankle
The management of the neuropathic ankle is predominantly nonoperative. However, severe, unbraceable deformity or impending soft-tissue compromise may justify an attempt at ankle arthrodesis as a limb-salvage procedure prior to considering a below-knee amputation.
Surgical Techniques for Ankle Arthrodesis:
1. External Fixation: Techniques utilizing external compression (e.g., Charnley or Calandruccio frames, or modern Ilizarov/Taylor Spatial Frames) augmented anteriorly by a sliding tibial graft have historically been popular, allowing for compression without placing hardware in potentially infected bone.
2. Intramedullary Nailing: The modern standard for neuropathic ankle arthrodesis is the retrograde tibiotalocalcaneal (TTC) intramedullary nail. Inserted retrograde through the calcaneus, across the subtalar and ankle joints, and into the tibial diaphysis, this device provides rigid, load-sharing biomechanical stability.
3. Talectomy: If the talus has completely disintegrated (frequently seen in pediatric patients with Charcot joints or severe adult neuroarthropathy), excision of the remaining necrotic talus followed by a primary tibiocalcaneal fusion is the treatment of choice.
Management of the Neuropathic Knee
If a neuropathic knee is painless and stable, bracing (e.g., a custom knee-ankle-foot orthosis [KAFO]) is the treatment of choice. However, many patients develop disabling instability or pain, necessitating operative consideration.
The Total Knee Arthroplasty (TKA) Controversy:
The use of TKA in neuropathic arthropathy remains highly controversial. The loss of proprioception leads to eccentric loading, rapid polyethylene wear, and massive osteolysis.
While some authors (e.g., Soudry et al., Parvizi et al.) have reported moderate success using highly constrained, hinged, or posterior-stabilized prostheses with custom augments, the complication rates remain unacceptably high. Kim, Kim, and Oh reported that at a 5-year follow-up, only 53% of TKAs in Charcot joints were satisfactory, with frequent catastrophic failures including aseptic loosening, periprosthetic fracture, and complete ligamentous disruption.
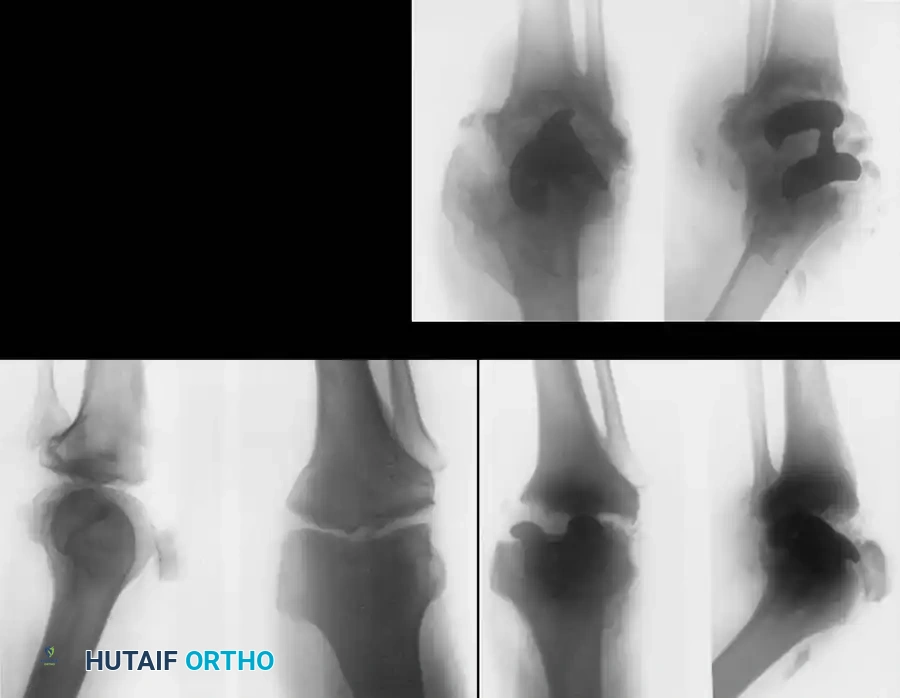
Surgical Warning: Total joint arthroplasty in a neuropathic knee is fraught with peril. The lack of protective neuromuscular reflexes inevitably leads to premature implant failure, massive bone loss, and a high likelihood of eventual amputation.
Knee Arthrodesis:
Arthrodesis is universally considered the surgical treatment of choice for the painful, unstable neuropathic knee. Achieving a solid fusion is notoriously difficult due to poor bone stock and impaired healing.
Strict adherence to the following surgical principles is mandatory:
1. Complete Debridement: Meticulous excision of all hypertrophic synovium, necrotic bone, and fibrotic tissue.
2. Careful Carpentry: Precise preparation of apposing bone surfaces (often utilizing cylindrical reaming or flat cuts) to maximize cancellous bone contact.
3. Strong Internal Fixation: Utilization of a long, rigid intramedullary nail spanning from the proximal femur to the distal tibia, or dual orthogonal plating.
4. Adequate External Support: Prolonged postoperative immobilization, often requiring a spica cast or long-leg cast for 6 to 12 months.
Management of the Neuropathic Hip
Neuropathic arthropathy of the hip presents a profound reconstructive challenge.
Fracture Management:
Fresh femoral neck fractures in neuropathic patients rarely heal after standard internal fixation (e.g., cannulated screws or dynamic hip screws). The true neuropathic nature of the fracture is often unrecognized until the fixation fails, the joint disintegrates, and the hardware cuts out. If internal fixation is attempted, the joint must be strictly immobilized in a hip spica cast postoperatively.
Arthroplasty and Arthrodesis:
* Total Hip Arthroplasty (THA): THA in a neuropathic hip is almost universally contraindicated. The literature is replete with reports of recurrent, intractable dislocations and rapid aseptic loosening.
* Arthrodesis: Achieving a successful arthrodesis of a neuropathic hip is exceptionally rare. Key and Burman historically noted the near impossibility of this procedure.
* Salvage Procedures: When the femoral head and neck have been completely destroyed, a Schanz osteotomy (a valgus-producing proximal femoral osteotomy) may be considered. This procedure medializes the mechanical axis and improves the resting tension of the abductor musculature, providing a stable, albeit stiff, fulcrum for ambulation. Ultimately, surgery of any kind in the neuropathic hip is indicated only for severe, intractable disability.
Neuropathic Arthropathy of the Spine (Charcot Spine)
Since Jean-Martin Charcot’s original description of spinal lesions in patients with tabes dorsalis, spinal neuropathic arthropathy has been documented across a spectrum of neurological deficits. Today, it is most frequently seen in patients with spinal cord injury (paraplegia), spinal dysraphism, diabetic neuropathy, and following extensive spinal fusions or laminectomies.
The Charcot spine is characterized by progressive, destructive discovertebral changes, massive osteophyte formation, and severe instability, often leading to a "ball-and-socket" pseudoarthrosis. This can result in progressive kyphotic deformity and, critically, a loss of sitting balance or further neurological compromise.
Surgical management is highly complex and involves extensive anterior and posterior circumferential fusion (360-degree constructs) utilizing robust instrumentation and copious autogenous bone grafting. The goal is to restore spinal alignment, decompress neural elements, and achieve rigid stabilization to prevent the relentless progression of the neuropathic destruction.
📚 Medical References
- Neuropathic arthropathy as a possible cause of failure of a whole joint allograft: a case report, Clin Orthop Relat Res 234:124, 1988.
- Trovik CS: Local recurrence of soft tissue sarcoma: a Scandinavian Sarcoma Group project, Acta Orthop Scand 72(suppl 300):1, 2001.
- Tsuchiya H, Tomita K, Mori Y, et al: Caffeine-assisted chemotherapy and minimized tumor excision for nonmetastatic osteosarcoma, Anticancer Res 18:657, 1998.
- Tsuchiya H, Tomita K, Mori Y, et al: Marginal excision for osteosarcoma with caffeine-assisted chemotherapy, Clin Orthop Relat Res 358:27, 1999.
- Turcotte RE, Kurt AM, Sim FH, et al: Chondroblastoma, Hum Pathol 24:944, 1993.
- Uchida A, Araki N, Shinto Y, et al: The use of calcium hydroxyapatite ceramic in bone tumour surgery, J Bone Joint Surg 74B:298, 1990.
- Unni KK: Dahlin’s bone tumors: general aspects and data on 11,087 cases, 5th ed, Philadelphia, 1996, Lippincott-Raven. Usui M, Murakami T, Naito T, et al: Some problems in wrist reconstruction after tumor resection with vascularized fi bularhead graft, J Reconstr Microsurg 12:81, 1996.
- Uyttendaele D, De Schryver A, Claessens H, et al: Limb conservation in primary bone tumors by resection extracorporeal irradiation and reimplantation, J Bone Joint Surg 70B:348, 1988.
- Van der Bijl AE, Taminiau AHM, Hermans J, et al: Accuracy of the Jamshidi Trocar Biopsy in the diagnosis of bone tumors, Clin Orthop Relat Res 334:233, 1997.
- Van Steensel MAM, Homminga GN, Buma P, et al: Optimization of cryopreservative procedures for human articular cartilage chondrocytes, Arch Orthop Trauma Surg 113:318, 1994.
- Vander Griend RA, Enneking WF: Radiologic imaging techniques in the diagnosis and treatment of osteogenic sarcoma, Semin Orthop 3:59, 1988.
- Vander Griend RA, Funderburk CH: The treatment of giant-cell tumors of the distal part of the radius, J Bone Joint Surg 75A:899, 1993.
- VanderWilde RS, Wold LE, McLeod RA, et al: Mayo Clinic tumor rounds: eosinophilic granuloma, Orthopedics 13:1301, 1990.
- Velchik MG, Heyman S, Makler PT Jr, et al: Bone scintigraphy: differentiating benign cortical irregularity of the distal femur from malignancy, J Nucl Med 25:72, 1984.
- Vicas E, Beauregard G, McKay Y: Malignant giant cell tumour of the distal femur treated by excision, allografting and ligamentous reconstruction: an 18-year follow-up, Can J Surg 40:459, 1997.
- Volpe CM, Pell M, Doerr RJ, et al: Radical scapulectomy with limb salvage for shoulder girdle soft tissue sarcoma, Surg Oncol 5:43, 1996.
- Waisman M, Kligman M, Roffman M: Posterior approach for radical excision of sacral chordoma, Int Orthop 21:181, 1997.
- Waldman BJ, Zerhouni EA, Frassica FJ: Case reports: recurrence of giant cell tumor of bone: the role of MRI in diagnosis, Orthopedics 20:67, 1997.
- Wang JW, Shih CH: Allograft transplantation in aggressive or malignant bone tumors, Clin Orthop Relat Res 297:203, 1993.
- Ward WG: Orthopaedic oncology for the nononcologist orthopaedist: introduction and common errors to avoid, Instr Course Lect 48:577, 1999.
- Ward WG, Kilpatrick S: Fine needle aspiration biopsy of primary bone tumors, Clin Orthop Relat Res 373:80, 2000.
- Ward WG, Spang J, Howe D: Metastatic disease of the femur: surgical management, Orthop Clin North Am 31:633, 2000.
- Ward WG, Spang J, Howe D, et al: Femoral recon nails for metastatic disease: indications, technique, and results, Am J Orthop 29:34, 2000.
- Ward WG, Yang RS, Eckardt JJ: Endoprosthetic bone reconstruction following malignant tumor resection in skeletally immature patients, Orthop Clin North Am 27:493, 1996.
- Watts HG: Introduction to resection of musculoskeletal sarcomas, Clin Orthop Relat Res 153:31, 1980.
- Weber KL, Lin PP, Yasko AW: Complex segmental elbow reconstruction after tumor resection, Clin Orthop Relat Res 415:31, 2003.
- Weir LM, Vanbergeyk AB, Masri BA, et al: Local excision without radiation for high-grade soft-tissue sarcoma of the extremity and superfi cial trunk, Sarcoma 4:113, 2000.
- Weis LD: The success of limb-salvage surgery in the adolescent patient with osteogenic sarcoma, Adolesc Med 10:451, 1999.
- Weis LD, Huber DF: Limb salvage after scapulohumeral tumor resection utilizing the Tikhoff-Linberg procedure, Surg Rounds Orthop 5:43, 1989.
- Widhe B, Widhe T: Initial symptoms and clinical features in osteosarcoma and Ewing sarcoma, J Bone Joint Surg 82A:667, 2000.
- Wilkins RM, Soubeiran A: The Phenix expandable prosthesis: early American experience, Clin Orthop Relat Res 382:51, 2001.
- Willen H: Fine needle aspiration in the diagnosis of bone tumors, Acta Orthop Scand 273:47, 1997.
- Winderen M, Stenwig AE, Solheim OP, et al: Dynamic bone scintigraphy for evaluation of tumor response after preoperative chemotherapy: a retrospective study of osteosarcoma and Ewing’s sarcoma patients, Acta Orthop Scand 285(suppl):11, 1999.
- Winkelmann WW: Hip rotationplasty for malignant tumors of the proximal part of the femur, J Bone Joint Surg 68A:362, 1986.
- Winkelmann WW: Rotationplasty in the local treatment of osteosarcoma, Semin Orthop 3:40, 1988.
- Winkelmann WW: Type-B-IIIa hip rotationplasty: an alternative operation for the treatment of malignant tumors of the femur in early childhood, J Bone Joint Surg 82A:814, 2000.
- Wittig J, Bickels J, Wodajo F, et al: Utilitarian shoulder approach for malignant tumor resection, Orthopedics 25:479, 2002.
- Wojciech W, Izbicki T, Rychlowska M, et al: Malignant humeral bone tumors in children: excision and reconstruction with the use of rotated clavicle, J Surg Oncol 62:183, 1996.
- Wold LE, McLeod RA, Sim FH, et al: Atlas of orthopedic pathology, Philadelphia, 1990, Saunders. Wong CC, Pho RWH, Rauff A, et al: Surgical treatment of primary tumours of innominate bone, Ann Acad Med 17:132, 1988.
- Wuisman P, Enneking WF: Prognosis for patients who have osteosarcoma with skip metastasis, J Bone Joint Surg 72A:60, 1990.
- Wuisman P, Härle A, Matthiass HH, et al: Two-stage therapy in the treatment of sacral tumors, Arch Orthop Trauma Surg 108:255, 1989.
- Wunder J, Leitch K, Griffi n A, et al: Comparison of two methods of reconstruction for primary malignant tumors at the knee: a sequential cohort study, J Surg Oncol 77:89, 2001.
- Wunder JS, Paulian G, Huvos AG, et al: The histological response to chemotherapy as a predictor of the oncological outcome of operative treatment of Ewing sarcoma, J Bone Joint Surg 80A:1020, 1998.
- Wurtz LD, Peabody TD, Simon MA: Delay in the diagnosis and treatment of primary bone sarcoma of the pelvis, J Bone Joint Surg 81A:317, 1999.
- Xu WP, Song XW, Yue SY, et al: Primary sacral tumors and their surgical treatment: a report of 87 cases, Chin Med J 103:879, 1990.
- Xunyuan D, Minsin J, Yuanpei X: Radical resection of the shoulder girdle for a malignant tumor: four case reports, Orthopedics 12:1017, 1989.
- Yajima H, Tamai S, Mizumoto S, et al: Vascularized fi bula graft for reconstruction after resection of aggressive benign and malignant bone tumors, Microsurgery 13:227, 1992.
- Yasko AW, Fanning CV, Ayala AG, et al: Percutaneous techniques for the diagnosis and treatment of localized Langerhanscell histiocytosis (eosinophilic granuloma of bone), J Bone Joint Surg 80A:219, 1998.
- Yip KMH, Leung PC, Kumta SM: Giant cell tumor of bone, Clin Orthop Relat Res 323:60, 1996.
- Yonemoto T, Tatezaki S, Ishii T, et al: Prognosis of osteosarcoma with pulmonary metastases at initial presentation is not dismal, Clin Orthop Relat Res 349:194, 1998.
- Yun YH, Kim NH, Han DY, et al: An investigation of bone necrosis and healing after cryosurgery, phenol cautery or packing with bone cement of defects in the dog femur, Int Orthop 17:176, 1993.
- Zatsepin ST: Conservative operations for pelvic bone tumours, Int Orthop 4:259, 1981.
- Zehr RJ: Treatment options for orthopaedic oncologic entities. Instr Course Lect 48:591, 1999.
- Zunino JH, Johnston JO: Early results of lower limb surgery for osteogenic sarcoma of bone, Orthopedics 21:47, 1998.
- Zunino JH, Johnston JO: Prognostic value of histologic tumor necrosis assessment in osteogenic sarcoma of bone, Am J Orthop 29:369, 2000.
