INTRODUCTION TO AUTOLOGOUS BONE GRAFTING AND THE RIA SYSTEM
The management of critical-sized bone defects, atrophic nonunions, and complex arthrodeses frequently demands the utilization of high-quality bone graft material. While the iliac crest bone graft (ICBG) has historically been considered the gold standard due to its rich osteogenic, osteoinductive, and osteoconductive properties, it is associated with significant donor-site morbidity, including chronic pain, infection, and neurovascular injury.
The advent of the Reamer-Irrigator-Aspirator (RIA) system has revolutionized the harvest of autologous bone graft. By utilizing the intramedullary canals of long bones (femur or tibia), the RIA system allows for the extraction of massive volumes (often exceeding 40 to 90 mL) of morselized bone graft. This intramedullary reamings-derived graft is highly vascularized and exceptionally rich in mesenchymal stem cells (MSCs), osteoprogenitor cells, and osteoinductive growth factors, matching or exceeding the biological profile of ICBG while dramatically reducing donor-site morbidity.
This comprehensive academic guide details the preoperative planning, precise surgical technique, and postoperative management for RIA bone graft harvesting, alongside an in-depth analysis of modern bone graft substitutes, orthobiologics, and biophysical stimulation modalities.
HARVEST OF FEMORAL OR TIBIAL BONE GRAFT WITH THE RIA INSTRUMENTATION
Preoperative Planning and Templating
Meticulous preoperative planning is mandatory to prevent catastrophic iatrogenic complications, such as cortical perforation or thermal necrosis.
- Radiographic Assessment: Obtain full-length, orthogonal anteroposterior (AP) and lateral radiographs of the donor bone (femur or tibia).
- Diameter Selection: Select the proper tube length and assembly for the bone to be reamed. Confirm the reaming diameter with diaphyseal radiographic measurement.
Surgical Warning: For bone harvesting, select a reamer head no larger than 1.5 mm than the measured isthmus diameter. Over-reaming significantly compromises the torsional and bending strength of the diaphysis, increasing the risk of postoperative pathological fracture.
Patient Positioning and Anesthesia
- Anesthesia: General anesthesia or regional neuraxial blockade with profound muscle relaxation is required.
- Positioning: Position the patient as for a standard intramedullary nailing procedure.
- Femur: Supine on a fracture table with traction, or lateral decubitus on a radiolucent flat table, depending on surgeon preference and concurrent procedures.
- Tibia: Supine on a radiolucent table with the knee flexed over a radiolucent triangle to allow hyperflexion for the starting trajectory.
Operative Technique: Step-by-Step
1. Accessing the Medullary Canal
Gain access to the bone utilizing the standard starting points for intramedullary nailing (e.g., piriformis fossa or greater trochanter for the femur; high anterior infrapatellar approach for the tibia).
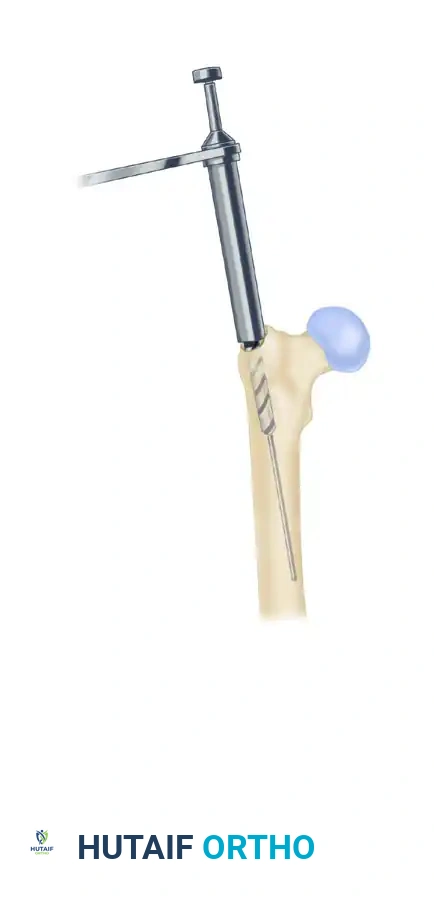
Open the proximal cortex with an awl or cannulated opening reamer.
2. Guidewire Placement
Insert a ball-tipped guidewire (reaming wire) down the medullary canal to the level of the distal physeal scar. Confirm central placement in both the AP and lateral planes using image intensification. Eccentric wire placement increases the risk of cortical blowout.
3. RIA Assembly and Preparation
- Assemble the RIA device strictly according to the manufacturer’s directions.
- Attach the drive shaft to the RIA and cover the connection with the locking clip.
- Attach the drive shaft seal to the proximal end of the drive shaft to maintain the closed suction environment.
- Attach the drive unit (reamer driver).
- Fluid Management: Connect the irrigation tubing to the smaller port marked “I.” Keep this clamped closed until irrigation begins. Connect the aspiration (suction) tubing to the larger port.
- Filter Attachment: Ensure the specialized graft filter canister is securely attached in-line with the aspiration tubing to collect the bone aspirate. Connect the distal end of the aspiration tube to the wall suction.
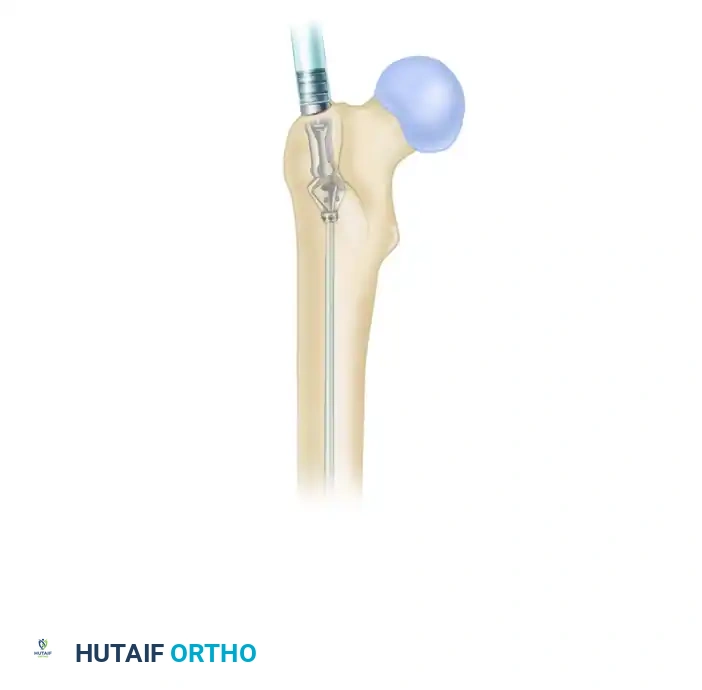
4. Insertion and Active Reaming
Slide the assembled RIA over the guidewire.
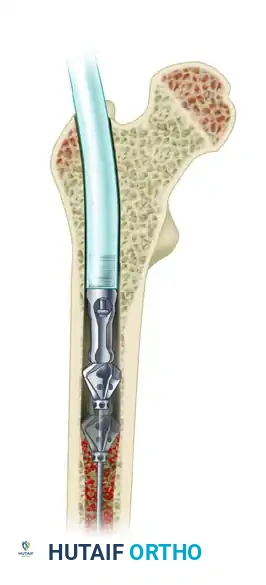
Before advancing into the bone, start the irrigation and aspiration to confirm proper functioning.
Clinical Pearl: Never ream when there is no irrigation or aspiration. The continuous flow of fluid is critical to cool the reamer head, prevent thermal osteonecrosis, and transport the morselized graft into the filter.
Insert the reamer into the bone and confirm its trajectory with fluoroscopy.
As the reamer is advanced under power, a continuous flow of bone and marrow should be visible in the aspiration tube.
5. The "Advance and Retract" Technique
To optimize graft yield and prevent intramedullary pressure spikes (which can lead to fat embolism):
- Ream forward slowly for 20 to 30 mm.
- Retract the reamer 50 to 80 mm to allow the irrigation fluid to fill the newly created space and flush the debris into the aspiration ports.
- Repeat this slow advancement until cortical resistance is felt at the isthmus.
- Repeat the retraction maneuver with reinsertion until the desired end point is reached on image intensification.
- If reaming becomes difficult or the device bogs down, the reamer can be briefly reversed to clear the flutes.
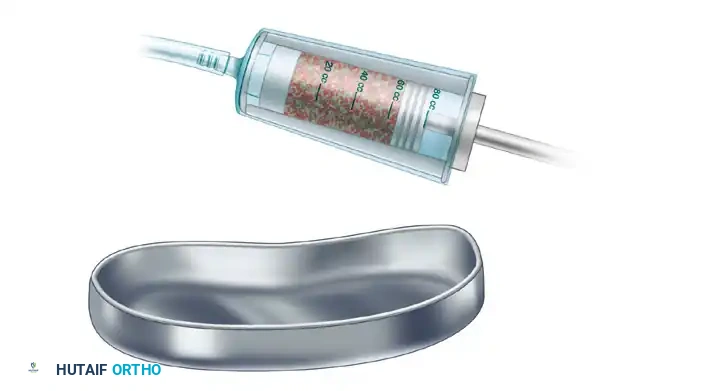
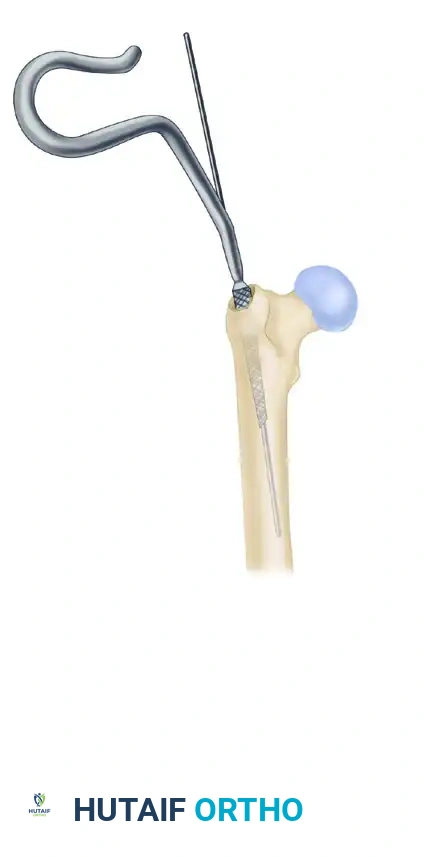
6. Graft Harvesting and Extraction
Once sufficient graft is obtained, stop irrigating after removing the RIA completely from the medullary canal. Turn off the suction or clamp the suction tubing to prevent drying out the graft.
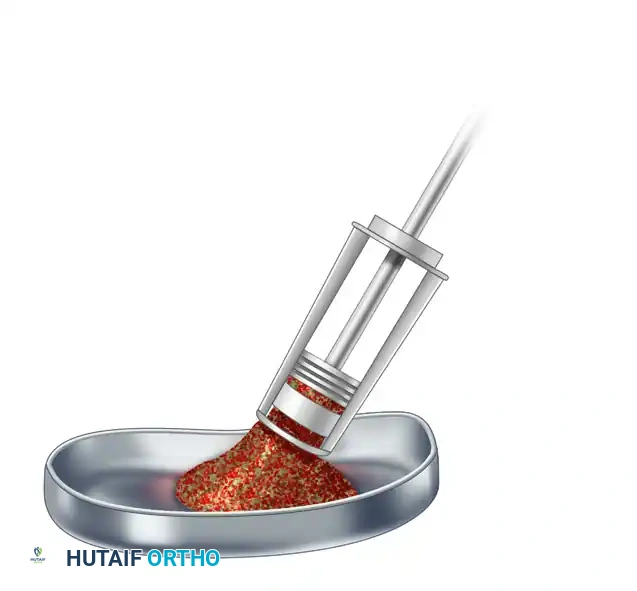
- Hold the graft filter vertically.
- Compress the graft with the provided plunger to squeeze out excess fluid and fat; record the volume measurement.
- With the plunger inserted, invert the filter and remove the inner filter from the outer canister.
- Hold the inner filter over a sterile basin and push out the bone graft.
Postoperative Care and Donor Site Management
Before concluding the procedure, carefully check the donor bone under fluoroscopy for any areas of eccentric thinning, weakness, or iatrogenic reamer perforation. Close the surgical wound in layers as you would after a standard intramedullary nailing.
Rehabilitation Protocol:
Although some cadaveric biomechanical studies have suggested that RIA harvesting (when adhering to the 1.5mm rule) does not dramatically diminish the mechanical properties of the femur and may not strictly require postoperative weight-bearing restrictions, a conservative approach is highly recommended.
- Weight-Bearing: We prefer to protect the donor bone by instructing the patient to use crutches and maintain partial or touch-down weight-bearing status.
- Progression: Advance weight-bearing only when endosteal healing and cortical hypertrophy are confirmed on follow-up radiographs (typically 6 to 12 weeks postoperatively).
BONE GRAFT SUBSTITUTES AND ORTHOBIOLOGICS
When autologous graft is insufficient or contraindicated, bone graft substitutes are employed. These materials are evaluated based on three fundamental biological properties:
1. Osteoconduction: Providing a physical scaffold for new bone growth.
2. Osteoinduction: Stimulating primitive mesenchymal cells to differentiate into bone-forming osteoblasts.
3. Osteogenesis: Providing live, bone-forming cells (osteoblasts and progenitor cells).
Allografts and Demineralized Bone Matrix (DBM)
Allografts (freeze-dried bone, DBM) provide an osteoconductive scaffold and, in the case of DBM, mild osteoinductive properties due to retained growth factors.
Surgical Warning: DBM is contraindicated in patients with severe vascular or neurological disease, fever, uncontrolled diabetes, severe degenerative bone disease, pregnancy, hypercalcemia, renal compromise, Pott disease, or active osteomyelitis/sepsis at the surgical site.
Complications of Allografts:
Transmission of disease from the donor is a rare but documented risk. Other complications include variable osteoinductive strength and deep infection. Even with rigorous donor screening and advanced sterilization methods (gamma irradiation, chemical processing), complete eradication of viral and bacterial agents cannot be guaranteed. Large structural allografts carry the highest risk of disease transmission, with documented cases of bacterial infections and Hepatitis B/C. DBM, due to its extensive processing, is significantly less likely to transmit infection.
Growth Factor–Based Bone Graft Substitutes
Marshall Urist first discovered Bone Morphogenetic Protein (BMP) in 1965, recognizing its profound ability to induce enchondral bone formation. BMPs are part of a massive group of cytokines within the transforming growth factor-beta (TGFβ) superfamily.
Most proteins in the TGFβ family regulate and modulate soft tissues, but specific BMPs are highly osteoinductive. Presently, only two recombinant proteins are FDA-approved for human use:
- rhBMP-2: FDA-approved for use in anterior lumbar interbody fusion (ALIF) with a titanium cage, and open tibial shaft fractures.
- rhBMP-7 (OP-1): Limited to use under Humanitarian Device Exemptions (HDE) for revision spinal fusions and recalcitrant long-bone nonunions.
Carrier Systems:
BMP-2 and BMP-7 are water-soluble and will rapidly diffuse away from the surgical site without a carrier. They are supplied with, or added to, an absorbable carrier (typically bovine or human type 1 collagen). Choosing a carrier that also possesses osteoconductive properties magnifies the inductive process. Care must be taken to avoid compressing the collagen sponge excessively, which extrudes the BMP liquid.
Other promising proteins currently under investigation for bone formation include Platelet-Derived Growth Factor (PDGF) and Vascular Endothelial Growth Factor (VEGF), which drive critical angiogenesis during callus formation.
Cell-Based, Ceramic, and Polymer Substitutes
Cell-Based Grafts:
These utilize autologous cells to seed a scaffold. The most frequently used is Bone Marrow Aspirate Concentrate (BMAC). Future applications may heavily feature adult somatic stem cells, dermal stem cells, and fetal cord blood cells.
Ceramic-Based Substitutes:
Ceramics provide excellent osteoconduction without the risk of disease transmission. Options include:
- Calcium sulfate
- Calcium phosphate (tricalcium phosphate, synthetic hydroxyapatite)
- Bioactive glass (silicate-based glass that bonds intimately with host tissue)
These products are highly osteointegrative but are inherently brittle. They are not recommended for load-bearing applications unless modified or used within protective cages. They serve as excellent void fillers or carriers for DBM and BMPs.
Polymer-Based Substitutes:
Polymers can be natural or synthetic, and biodegradable (polyglycolic acid) or nonbiodegradable. Composites of polymers and ceramics can occasionally be used in load-bearing areas, though rapid resorption limits the structural utility of biodegradable variants.
Miscellaneous Substitutes:
- Coralline Hydroxyapatite: An early bone substitute that resorbs very slowly. Strong in compression but weak in shear, limiting spinal applications.
- Chitosan and Sponge Skeletons: Emerging materials requiring close proximity to host bone to achieve effective conduction.
BIOPHYSICAL STIMULATION: ELECTRICAL AND ULTRASOUND
When biological and mechanical environments require augmentation, biophysical stimulation can alter the cellular microenvironment to favor osteogenesis.
Electromagnetic Stimulation (PEMF)
Pulsed Electromagnetic Field (PEMF) stimulation has been utilized since the 1970s for the treatment of delayed unions and nonunions, boasting success rates of 64% to 85%.
- Mechanism: While the exact mechanotransduction pathway is complex, in vitro exposure of osteoblasts to electromagnetic fields stimulates the secretion of critical growth factors, including BMP-2, BMP-4, TGFβ, and IGF-2.
- Efficacy: Double-blind prospective studies confirm positive effects on healing femoral and tibial osteotomies, though it has not been proven universally effective for fresh, uncomplicated fractures.
Low-Intensity Pulsed Ultrasound (LIPUS)
LIPUS provides mechanical acoustic energy that translates into biochemical signals at the fracture site.
- Mechanism: Low-intensity ultrasound increases the incorporation of calcium ions in cartilage and bone cell cultures. It upregulates the expression of genes involved in healing, including IGF and TGFβ.
- Clinical Evidence: In murine models, LIPUS accelerates soft callus formation and endochondral ossification. Animal studies show a 1.5x acceleration in fresh fracture healing. Clinical trials demonstrate an approximately 40% shorter healing time in tibial and radial fractures.
- Indications: LIPUS is highly recommended to enhance fracture healing in high-risk populations, including smokers, diabetics, patients with vascular insufficiency or osteoporosis, and those on inhibitory medications (corticosteroids, NSAIDs, calcium channel blockers).
INDICATIONS FOR SURGICAL FRACTURE FIXATION
The decision to proceed with operative intervention versus conservative management is dictated by fracture morphology, patient physiology, and biomechanical demands.
Absolute and High-Priority Indications
- Displaced Intra-articular Fractures: Requiring anatomic reduction to prevent post-traumatic osteoarthritis.
- Unstable Fractures: Where an appropriate trial of conservative management (casting/bracing) has failed to maintain acceptable alignment.
- Major Avulsion Fractures: Associated with the disruption of critical musculotendinous units or ligaments (e.g., patellar tendon avulsions, displaced greater tuberosity fractures) that yield poor results non-operatively.
- Displaced Pathological Fractures: In patients who are not imminently terminal, to provide pain relief and restore mobility.
- Fractures with Known Poor Conservative Outcomes: Including displaced femoral neck fractures, Galeazzi fracture-dislocations, and Monteggia fracture-dislocations.
- Displaced Physeal Injuries: Specifically Salter-Harris types III and IV, which cross the epiphyseal plate and carry a high propensity for premature growth arrest if not anatomically reduced.
- Impending or Actual Compartment Syndrome: Fractures requiring emergent fasciotomies mandate concurrent skeletal stabilization to protect the soft tissues.
- Nonunions and Malunions: Especially those that are malreduced, where previous conservative or surgical treatments have failed.
Relative Indications (Moderate Probability of Improved Function)
- Polytrauma Patients: Unstable spinal injuries, long bone fractures, and unstable pelvic fractures require early total care or damage control orthopedics to facilitate nursing, pulmonary toilet, and mobilization.
- Delayed Unions: After an appropriate but unsuccessful trial of conservative management.
- Impending Pathological Fractures: Prophylactic stabilization based on Mirels' criteria.
- Unstable Open Fractures: Requiring rigid fixation to protect soft tissue envelopes and facilitate repeated debridements.
By mastering the RIA harvest technique, understanding the nuanced application of orthobiologics, and adhering to strict biomechanical principles for fracture fixation, the orthopedic surgeon can reliably navigate complex reconstructive challenges and optimize patient outcomes.
