Introduction to Tendon Injuries of the Hand
The management of flexor and extensor tendon injuries remains one of the most challenging and technically demanding areas of hand surgery. The primary goal of tendon repair is to restore the delicate balance between tensile strength and unhindered excursion. Since the foundational work of Bunnell in the early 20th century, the field has evolved significantly through the contributions of Boyes, Stark, Verdan, Kessler, Kleinert, Gelberman, Manske, Hunter, Lister, and Strickland.
Modern operative orthopaedics emphasizes a profound understanding of tendon anatomy, biomechanics, nutrition, and the biology of healing. Current practice dictates that successful outcomes rely not only on meticulous surgical execution but also on highly structured, evidence-based postoperative rehabilitation protocols designed to stimulate intrinsic healing while preventing restrictive adhesions.
Anatomy and Biomechanics of Flexor Tendons
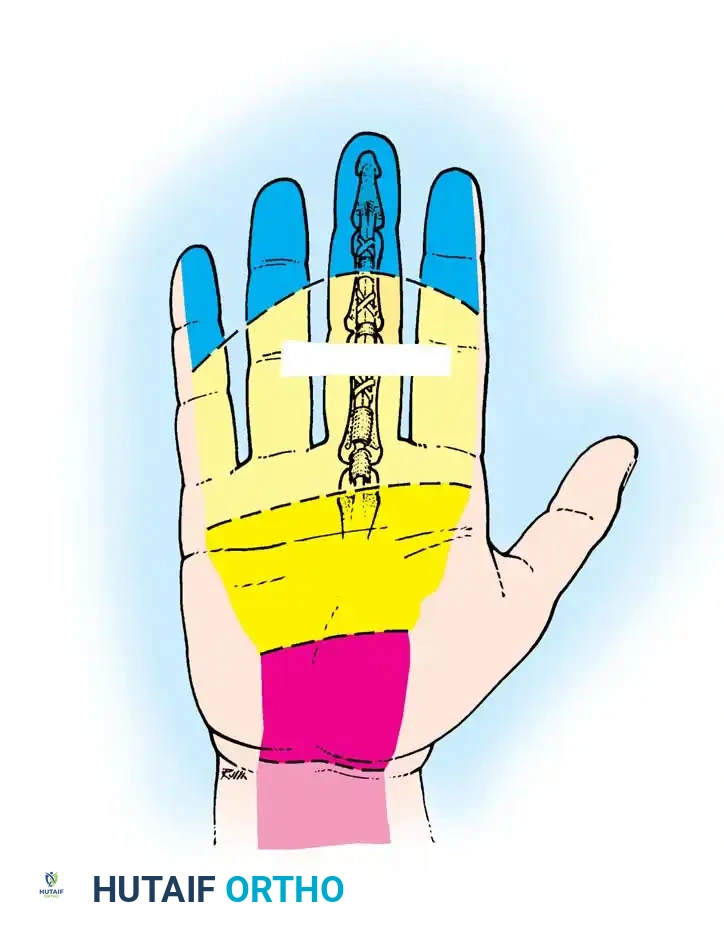
A comprehensive understanding of the flexor tendon anatomy in the forearm, wrist, and hand is a prerequisite for any surgeon undertaking these repairs. The flexor digitorum profundus (FDP) and flexor digitorum superficialis (FDS) operate within a highly specialized fibro-osseous canal, stabilized by a complex retinacular pulley system.
Tendon Nutrition and Vascularity
Tendon nutrition is derived from two distinct, yet complementary, sources:
1. Synovial Diffusion: Synovial fluid produced within the tenosynovial sheath is pumped into the tendon through the mechanism of cyclic loading and excursion.
2. Vascular Perfusion: Blood supply is provided through longitudinal vessels in the paratenon, intraosseous vessels at the tendon insertion, and the highly specialized vincular circulation.
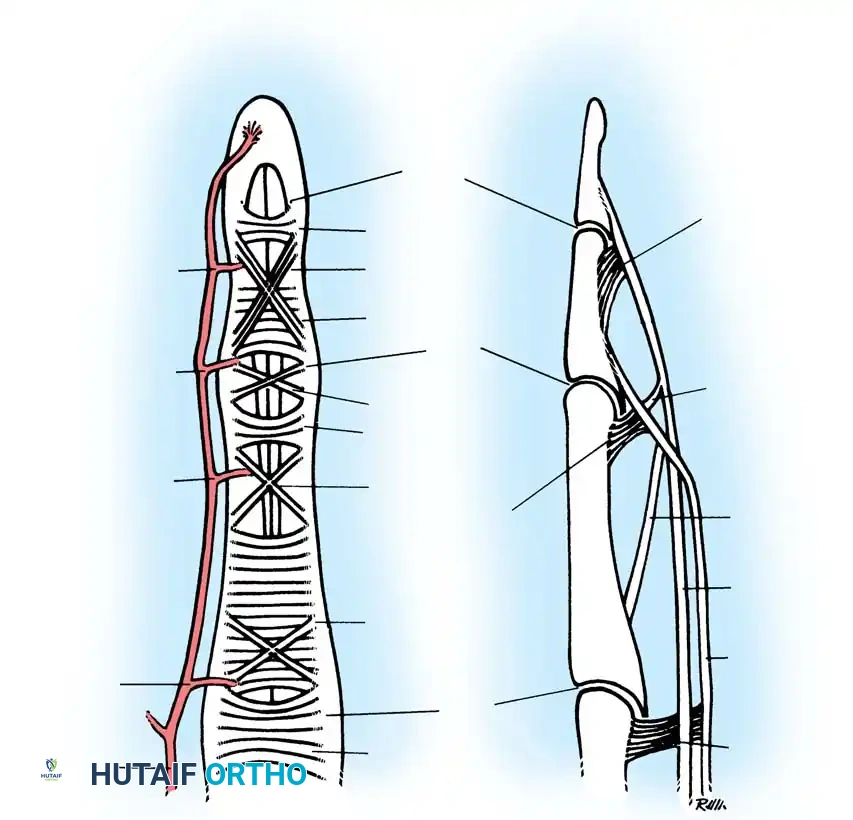
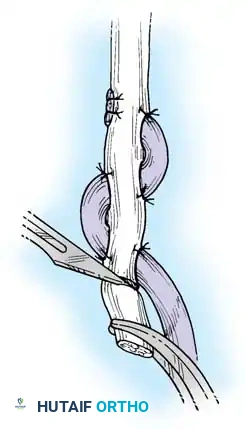
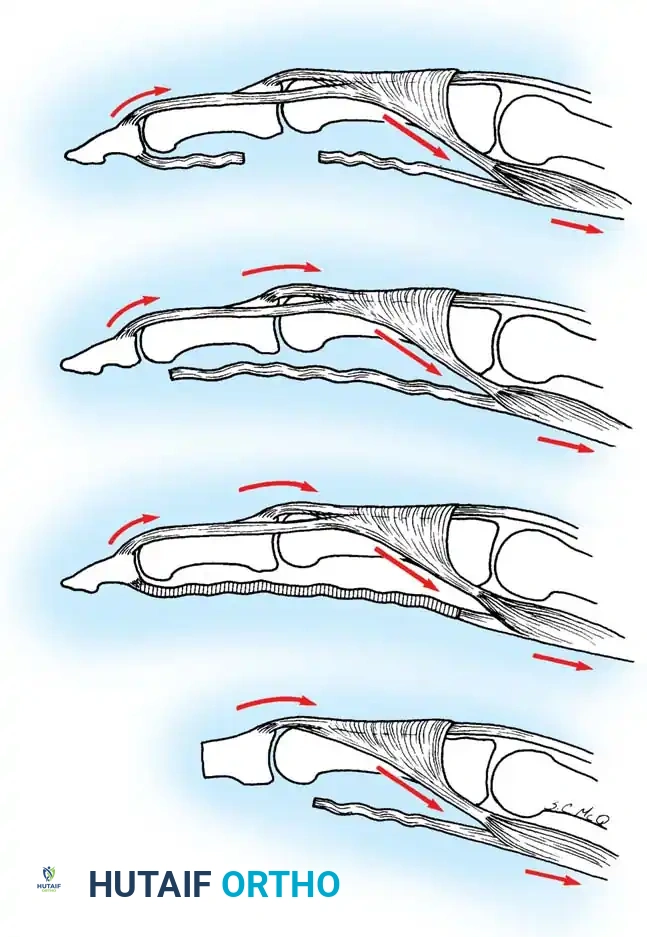
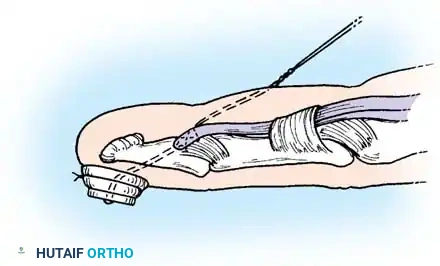
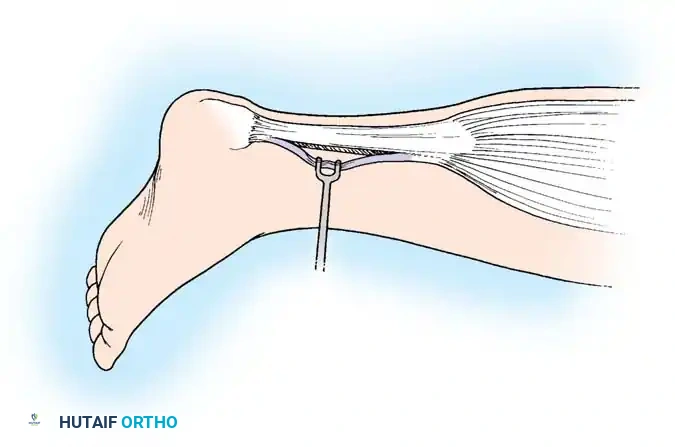
Fig. 63-1: Vascular supply to flexor tendons is provided by four transverse communicating branches of the digital arteries, feeding the vinculum breve and longum of both the superficialis and profundus tendons.
Clinical Pearl: There are distinct zones of relative ischemia within the flexor tendons that are highly susceptible to rupture and poor healing. The FDS has an ischemic zone beneath the A2 pulley, while the FDP has two ischemic zones—one beneath the A2 pulley and another beneath the A4 pulley. Surgical handling in these zones must be exceptionally atraumatic.
The Biology of Tendon Healing
Tendon healing occurs via a combination of extrinsic and intrinsic mechanisms, progressing through three overlapping phases:
* Inflammatory Phase (48 to 72 hours): Characterized by cellular infiltration, edema, and the release of chemotactic factors. The repair relies entirely on the strength of the suture material during this phase.
* Fibroblastic Phase (5 days to 4 weeks): Fibroblasts synthesize disorganized type III collagen. The tendon begins to gain intrinsic strength, but the repair site remains vulnerable to gap formation.
* Remodeling Phase (4 weeks to ~3.5 months): Collagen aligns longitudinally along lines of stress, transitioning to type I collagen, significantly increasing tensile strength.
The extrinsic mechanism involves the invasion of peripheral fibroblasts and inflammatory cells, which is the primary driver of scar tissue and adhesion formation. The intrinsic mechanism relies on the proliferation of tenocytes residing within the tendon itself.
Surgical Warning: Tendon injury combined with trauma to the synovial sheath and prolonged immobilization inevitably leads to extensive, motion-limiting adhesions. Experimental models (Tanaka et al.) demonstrate that controlled, cyclic tension applied to healing tendons preferentially stimulates the intrinsic healing response over the extrinsic response, forming the biological rationale for early active motion (EAM) protocols.
Clinical Evaluation and Examination
Evaluation of a patient with a suspected tendon injury begins with a standard trauma assessment, ruling out associated fractures, dislocations, and neurovascular compromise. Careful examination of the digital nerves and arteries must precede any functional tendon testing, as local anesthesia will obscure sensory findings.
Resting Posture and Passive Maneuvers
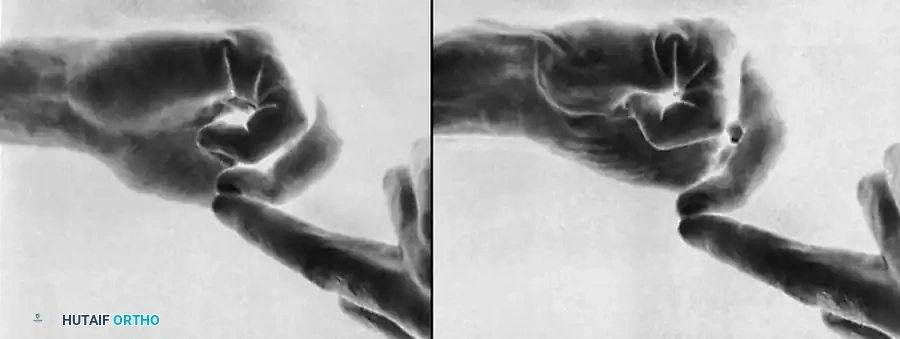
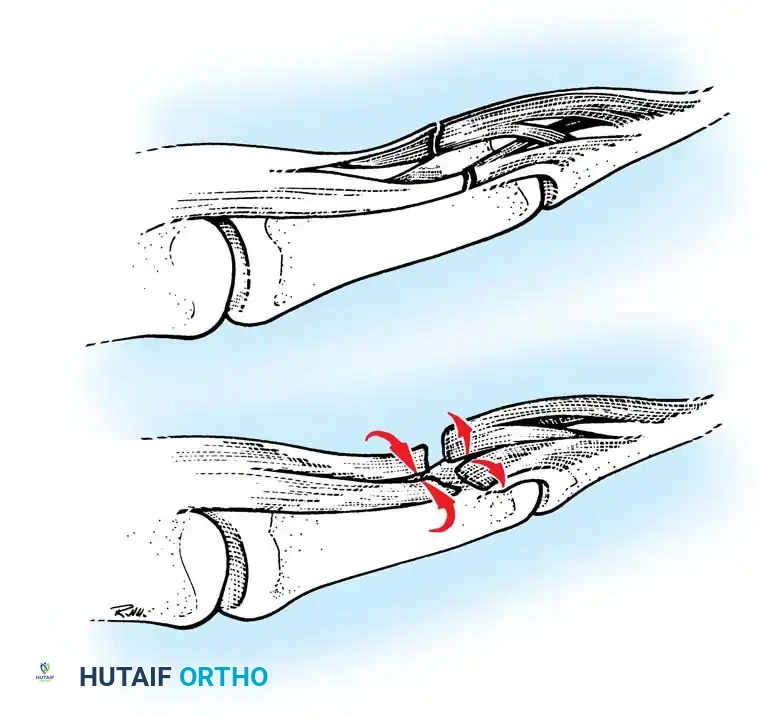
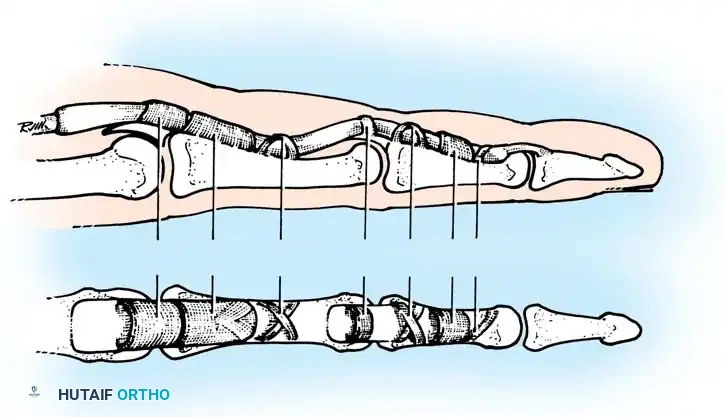
Even in the absence of gross deformity or active patient cooperation, the resting posture of the hand provides critical diagnostic clues. The normal resting cascade of the fingers exhibits increasing flexion from the index to the small finger.
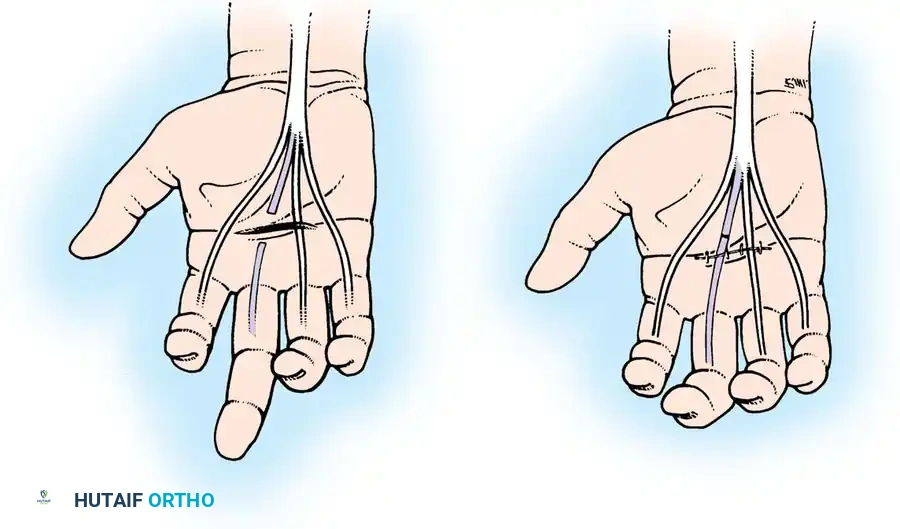
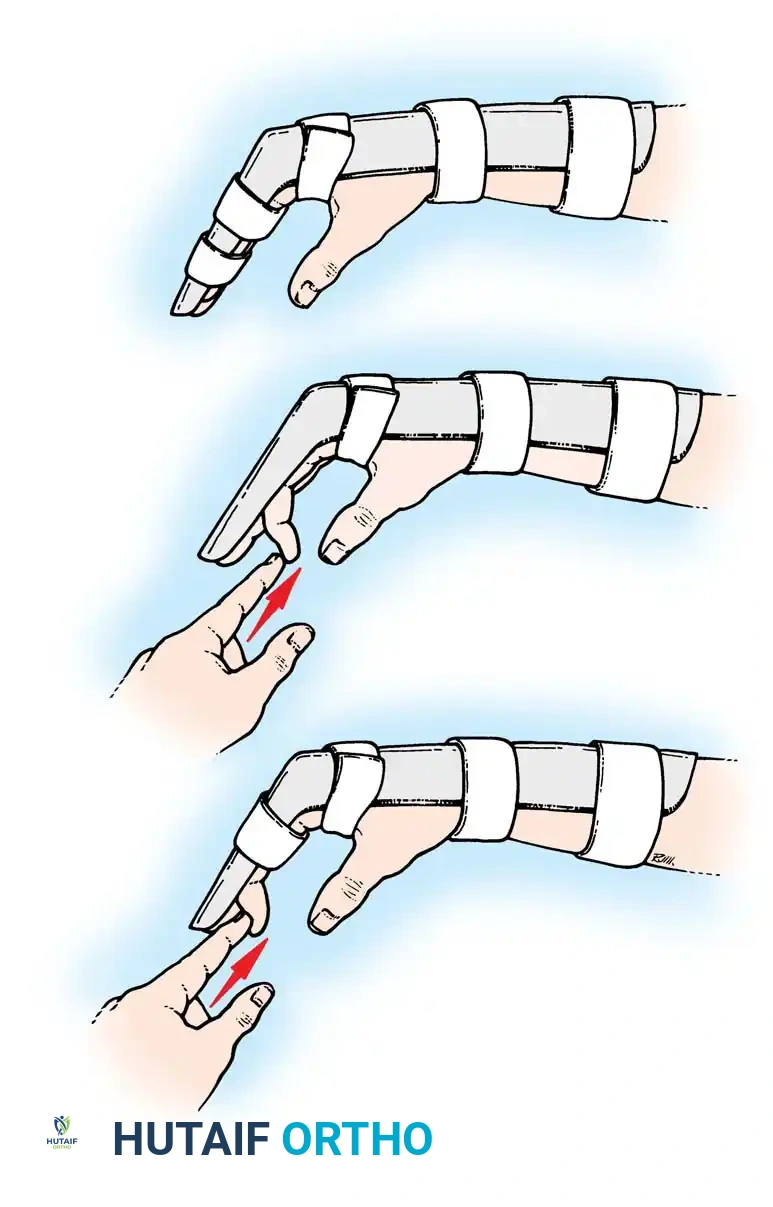
Fig. 63-2: (A) Loss of the normal resting cascade. If a finger remains extended when the hand is at rest, the flexor tendons have been severed. (B) Restoration of the normal flexed posture after profundus and sublimis repair.
Passive maneuvers are highly reliable, especially in pediatric or uncooperative patients:
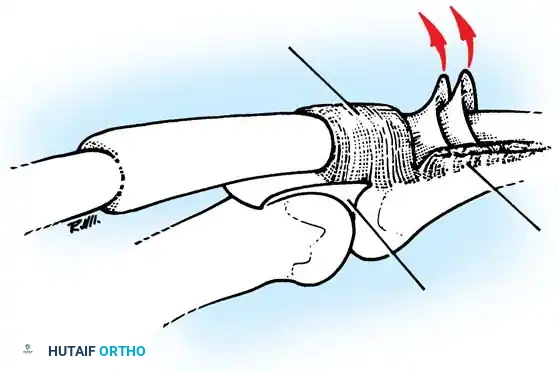
* Tenodesis Effect: Passive extension of the wrist normally produces flexion of the fingers. If the flexor tendons are severed, the affected digit will remain extended.
* Forearm Compression: Gentle compression of the volar forearm muscle mass will cause concomitant flexion of intact digits; an injured digit will fail to flex, indicating a discontinuity in the musculotendinous unit.
Active Functional Testing
When the patient is cooperative, specific active movements are evaluated. The examiner must stabilize adjacent joints to isolate the function of the FDP and FDS.
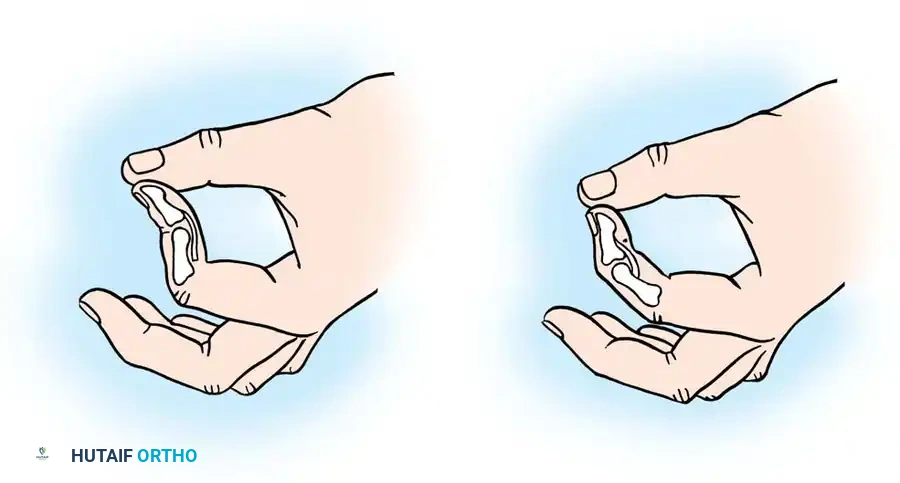
Testing the Flexor Digitorum Profundus (FDP):
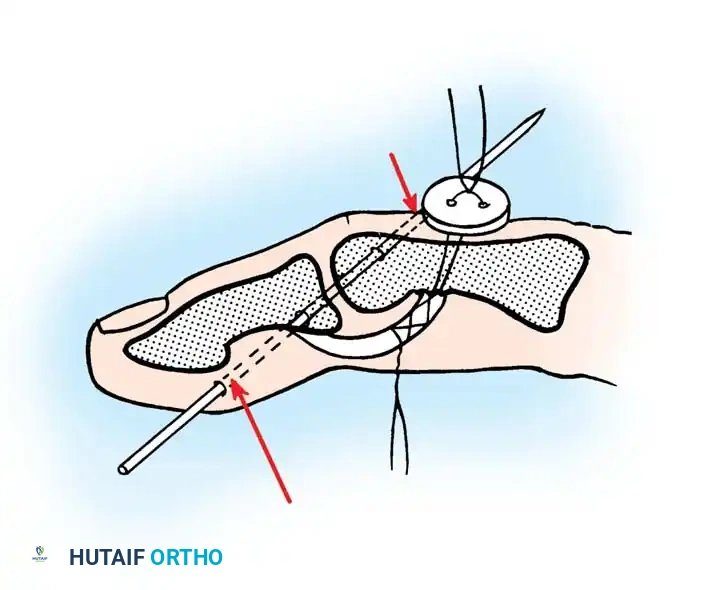
With the proximal interphalangeal (PIP) joint firmly stabilized by the examiner, the patient is asked to flex the distal interphalangeal (DIP) joint. Inability to do so indicates an FDP laceration.
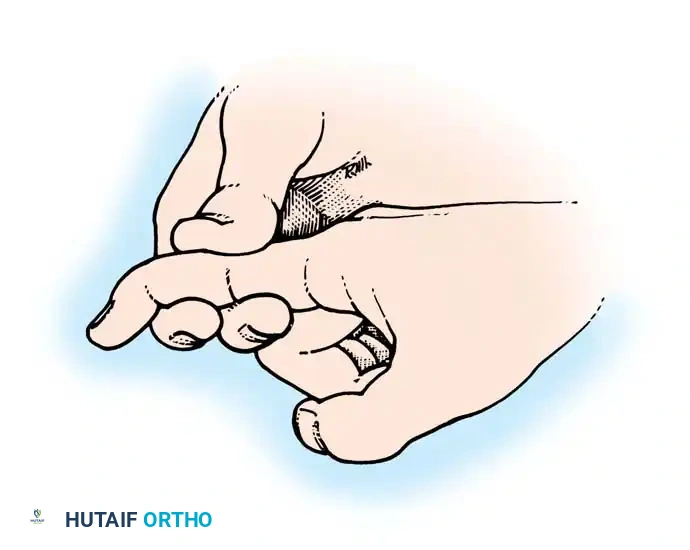
Fig. 63-3: Isolation and testing of the flexor digitorum profundus.
Testing the Flexor Digitorum Superficialis (FDS):
The FDS is tested by holding all adjacent fingers in maximal passive extension. This maneuver anchors the common muscle belly of the FDP, effectively neutralizing its ability to flex the PIP joint of the test digit. The patient is then asked to flex the test finger. If the PIP joint flexes, the FDS is intact.
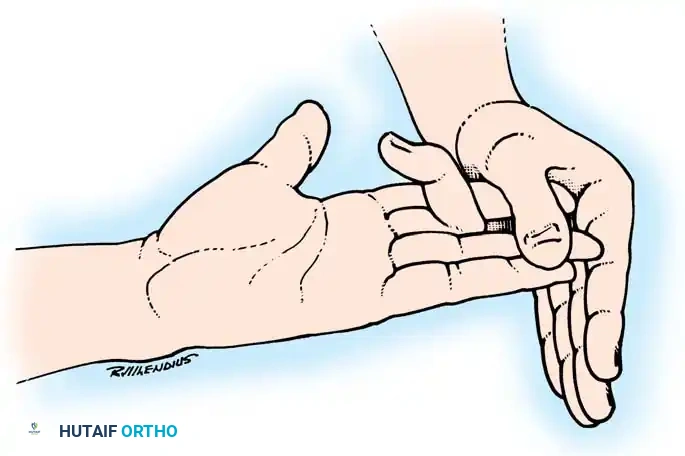
Fig. 63-4: Isolation and testing of the flexor digitorum superficialis.
Pitfall: The index finger FDP often has an independent muscle belly. To accurately test the index FDS, utilize Lister’s paper pinch test. The patient pinches a sheet of paper between the thumb and index finger. An intact FDS allows PIP flexion with DIP hyperextension. If the FDS is severed, the DIP will hyperflex to maintain the pinch.
Surgical Principles of Tendon Repair
The ideal tendon repair provides sufficient tensile strength to withstand the forces of early active motion while minimizing bulk to allow unhindered gliding through the pulley system.
Suture Material and Biomechanics
The choice of suture material significantly impacts the fatigue strength and gap resistance of the repair.
* Braided Synthetic Sutures: Braided polyester (e.g., Ticron, Mersilene) is the gold standard for core sutures. It provides excellent handling, superior knot security, and high resistance to gap formation.
* Monofilament Sutures: While monofilament nylon or polypropylene (Prolene) glide easily, they exhibit higher rates of early gap formation and knot slippage compared to braided polyester.
* Absorbable Sutures: Polydioxanone (PDS) maintains strength longer than polyglycolic acid (Vicryl) and can be used in specific pediatric or delayed repair scenarios, but non-absorbable sutures remain preferred for primary flexor repairs.
Suture Caliber:
Biomechanical studies (Taras et al., Barrie et al.) demonstrate that 3-0 core sutures provide a twofold to threefold increase in fatigue strength compared to 4-0 sutures. For adult flexor tendons in Zones II-V, a 3-0 braided polyester suture is recommended if an early active motion program is planned. A 4-0 suture may be reserved for smaller digits or distal Zone I repairs.
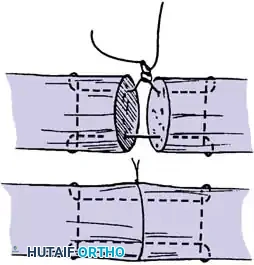
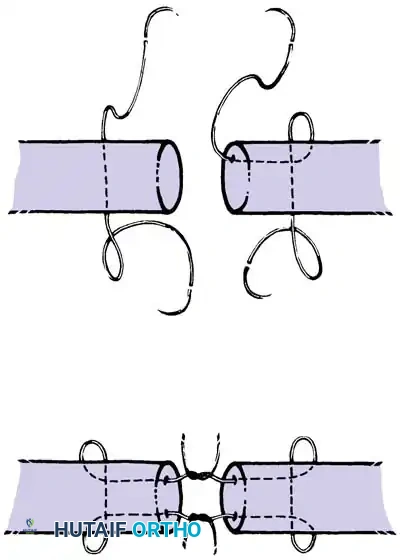
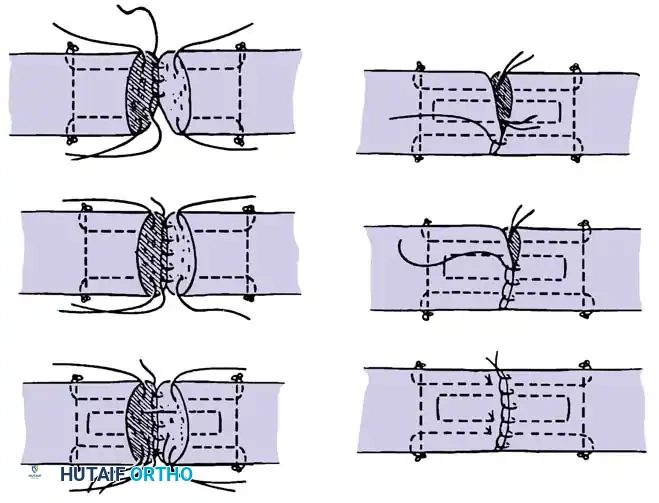
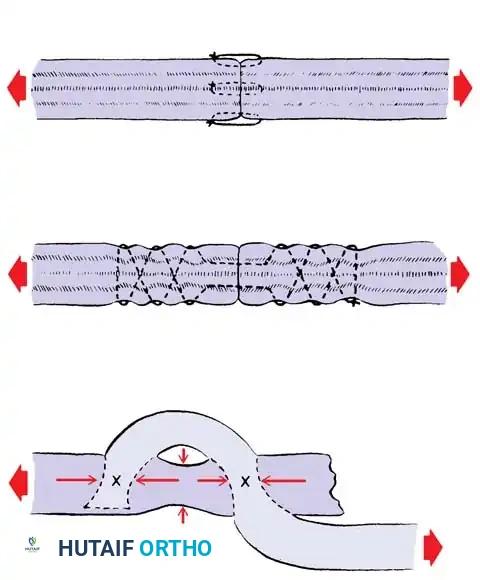
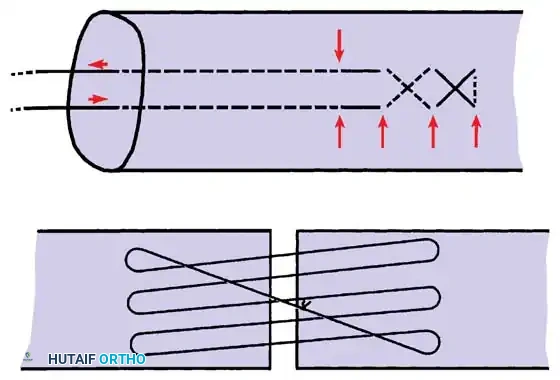
Core Suture Configurations
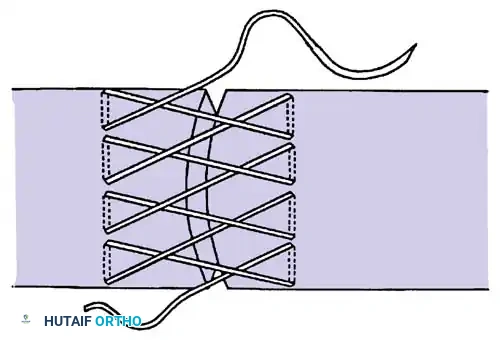
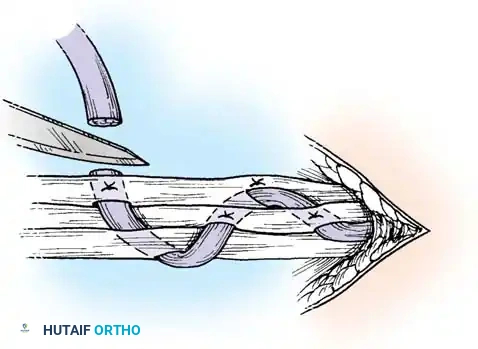
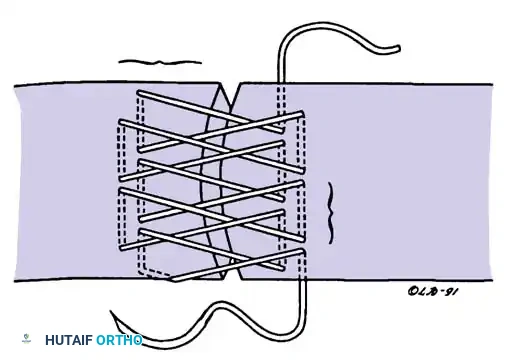
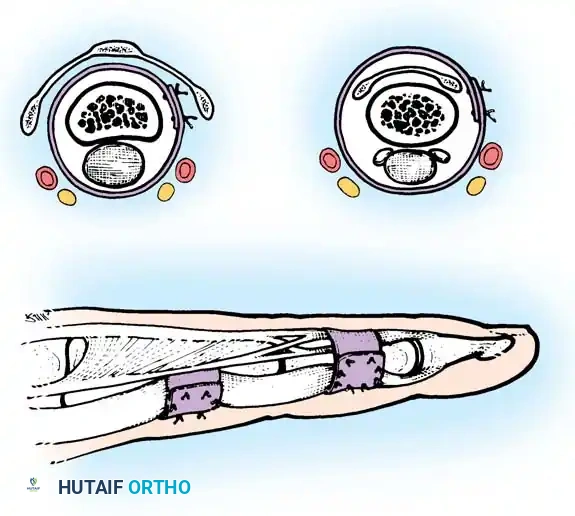
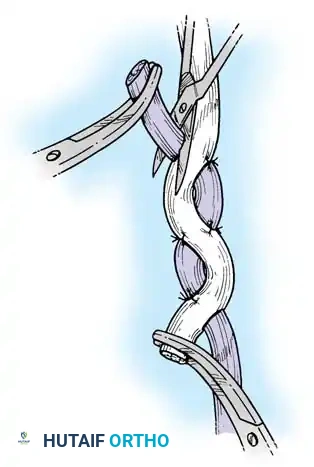
The strength of a tendon repair is directly proportional to the number of suture strands crossing the repair site.
* Two-Strand Repairs (e.g., Modified Kessler, Bunnell): Historically popular, but generally lack the tensile strength required for modern early active motion protocols.
* Four-Strand Repairs (e.g., Cruciate, Double Kessler): The current standard of care. They provide a reliable balance between high tensile strength, resistance to gap formation, and minimal disruption of intrinsic tendon vascularity.
* Six-Strand Repairs: Offer maximum strength but can increase tendon bulk, potentially causing triggering or friction within the A2/A4 pulleys.
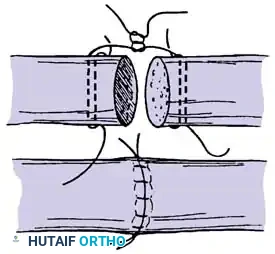
Core suture placement requires precise purchase of the tendon fibers, ensuring the knots are buried within the repair site to minimize epitenon friction.
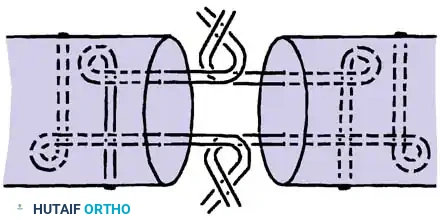
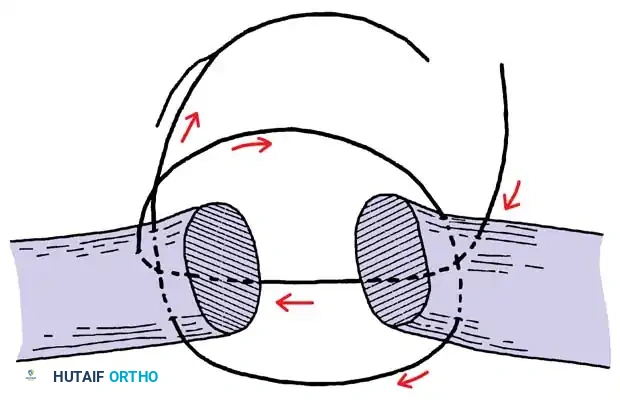
Epitendinous Repair
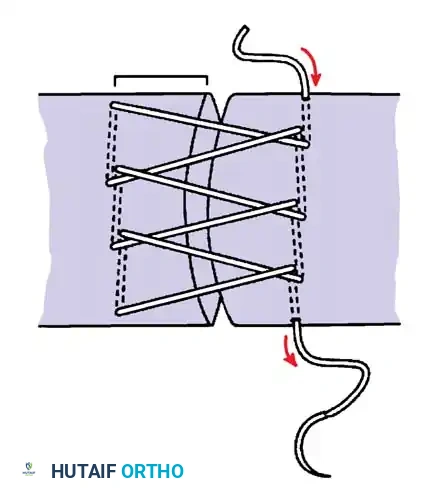
A continuous epitendinous suture (typically 5-0 or 6-0 monofilament nylon) is mandatory. It serves two critical functions:
1. Biomechanical: Increases the overall tensile strength of the repair by 20% to 30% and significantly reduces the risk of gap formation.
2. Gliding: Inverts the tendon edges, smoothing the repair site and reducing friction as the tendon passes through the retinacular pulleys.
Emerging Technologies
Recent advancements include intratendinous, crimped, single-strand, multifilament stainless steel devices (e.g., Teno Fix). Multicenter randomized trials (Su et al.) have shown these devices compare favorably with four-strand cruciate repairs, demonstrating lower rupture rates and excellent functional outcomes, though their bulk may limit use in tight Zone II injuries.
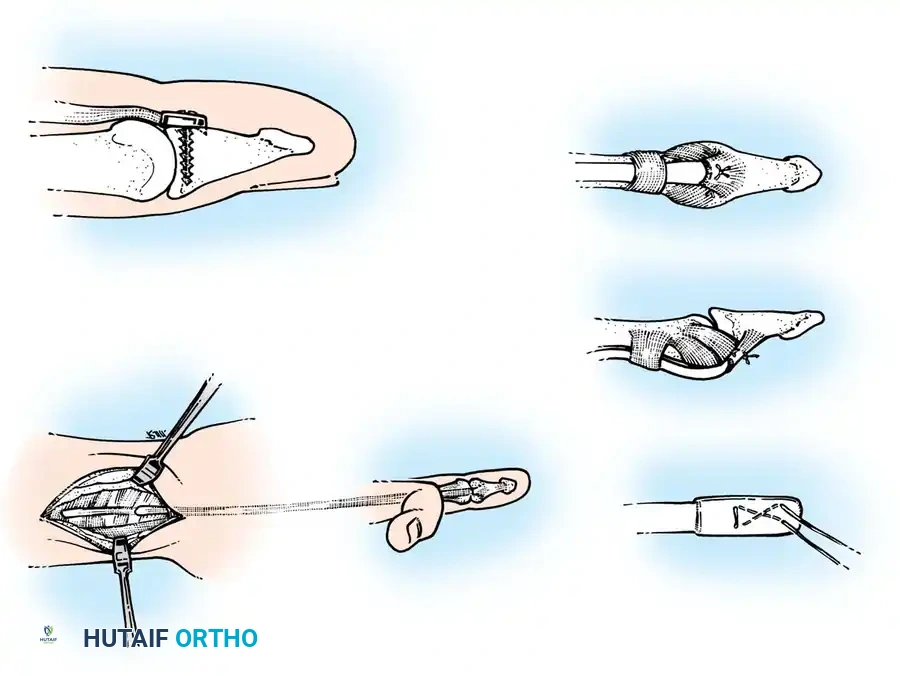
Step-by-Step Surgical Approach: Primary Flexor Tendon Repair
1. Positioning and Preparation
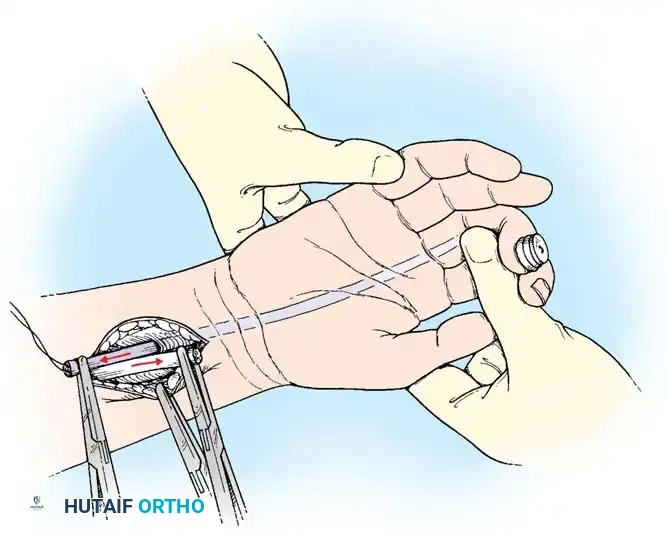
- Anesthesia: General anesthesia or regional block (axillary/supraclavicular). Wide-awake local anesthesia no tourniquet (WALANT) is increasingly popular, allowing intraoperative active movement testing to assess repair integrity and gliding.
- Positioning: Supine with the arm on a radiolucent hand table.
- Tourniquet: Exsanguinate the limb and inflate the pneumatic tourniquet to 250 mmHg (unless utilizing WALANT).
- Magnification: Surgical loupes (2.5x to 3.5x) are essential for precise epitenon handling.
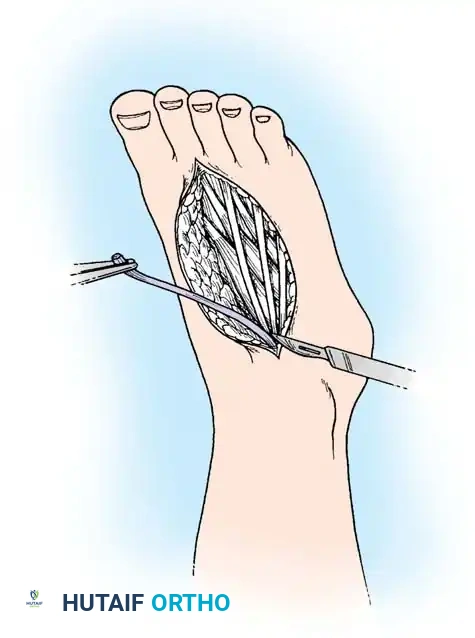
2. Incision and Exposure
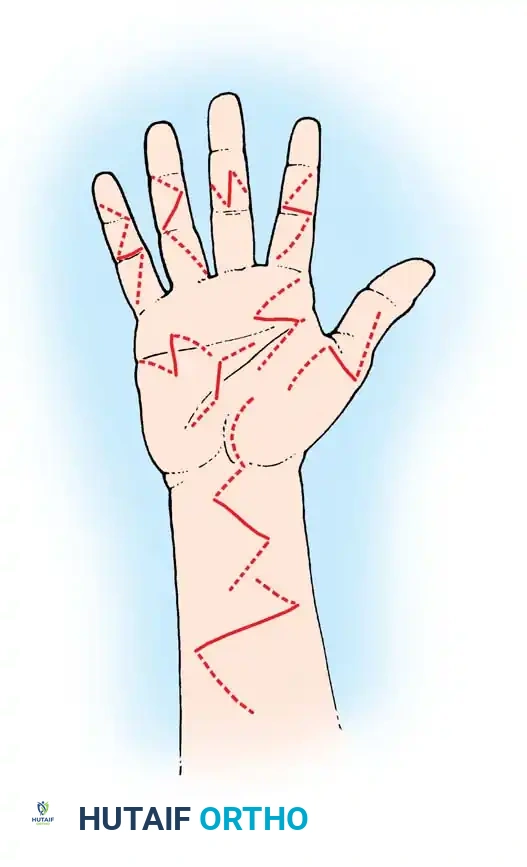
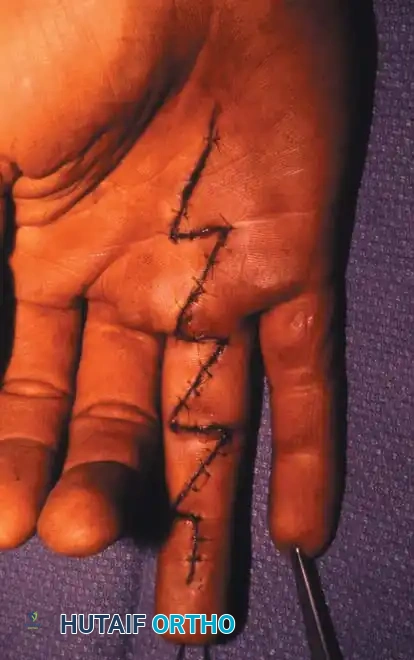
- Extend the traumatic laceration using Brunner (zigzag) or mid-lateral incisions. Avoid crossing flexion creases at a right angle to prevent flexion contractures.
- Elevate full-thickness flaps, keeping the neurovascular bundles protected within the volar flap.
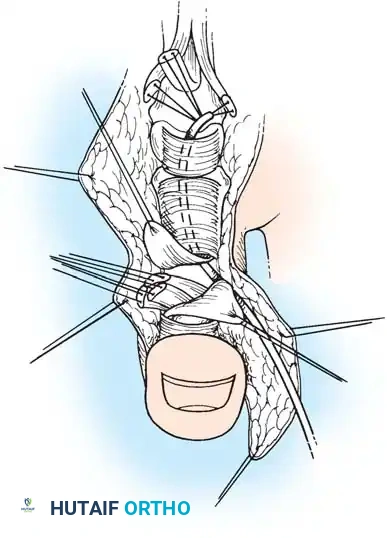
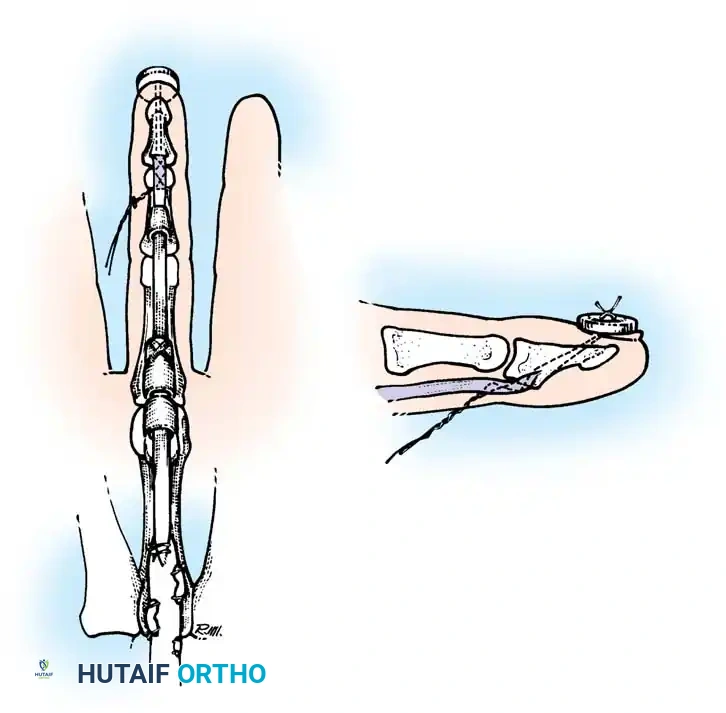
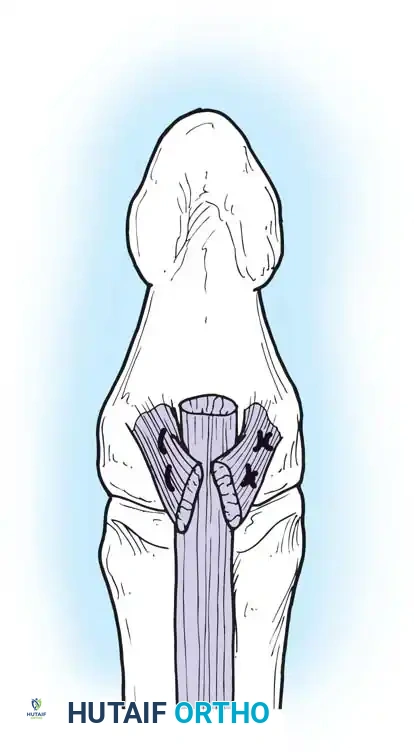
Careful elevation of the skin flaps exposes the flexor tendon sheath. The critical A2 and A4 pulleys must be identified and preserved.
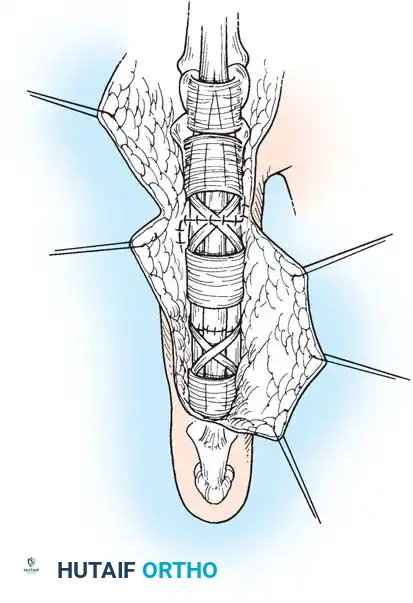
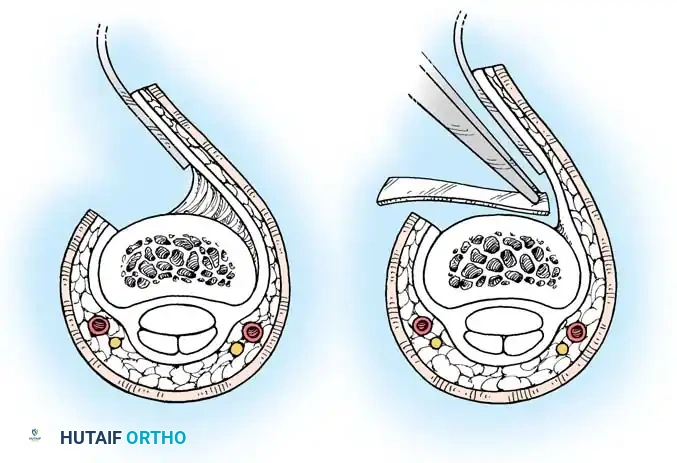
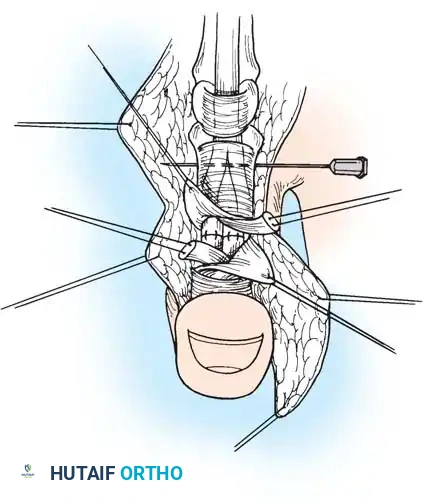
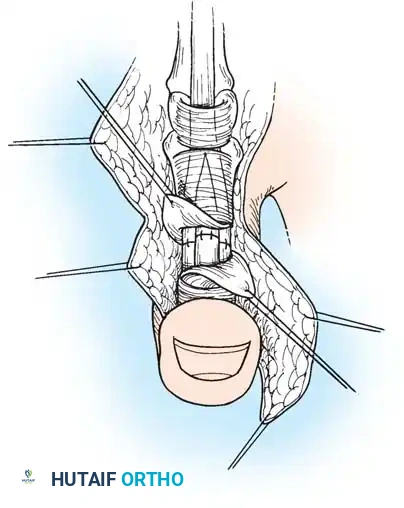
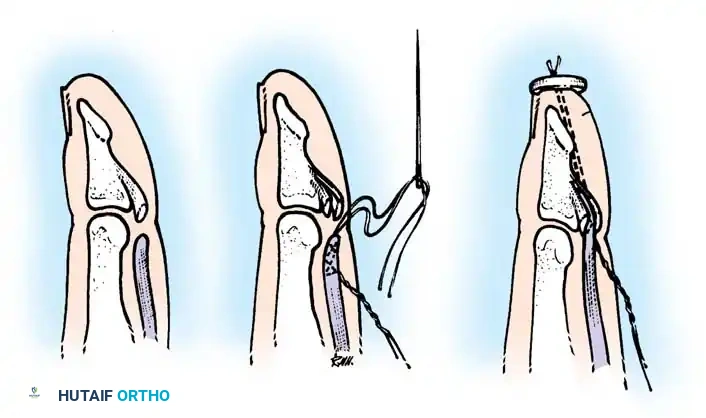
3. Sheath Management and Tendon Retrieval
- Open the flexor sheath through the cruciate pulleys (C1, C2) or membranous portions, strictly preserving the A2 and A4 pulleys to prevent bowstringing.
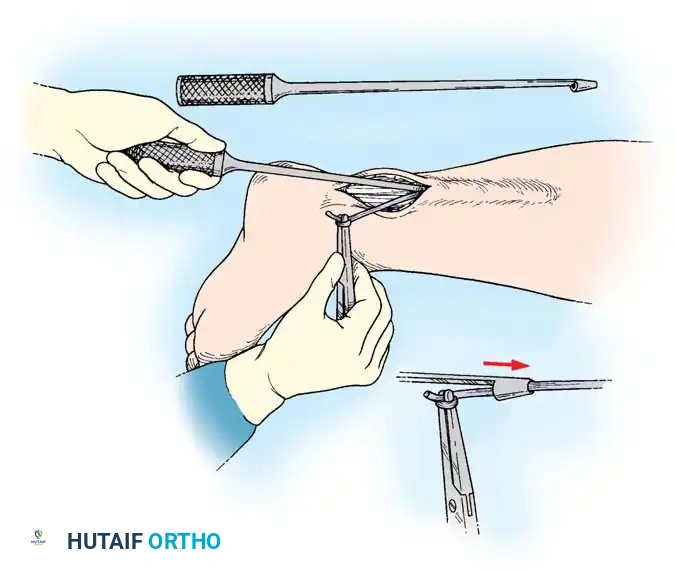
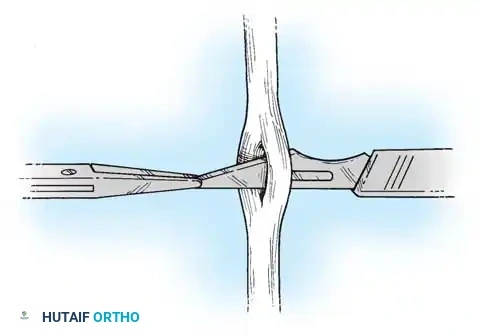
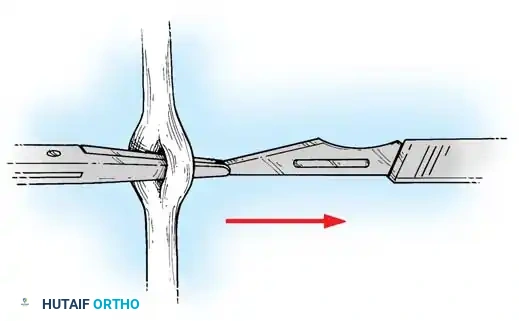
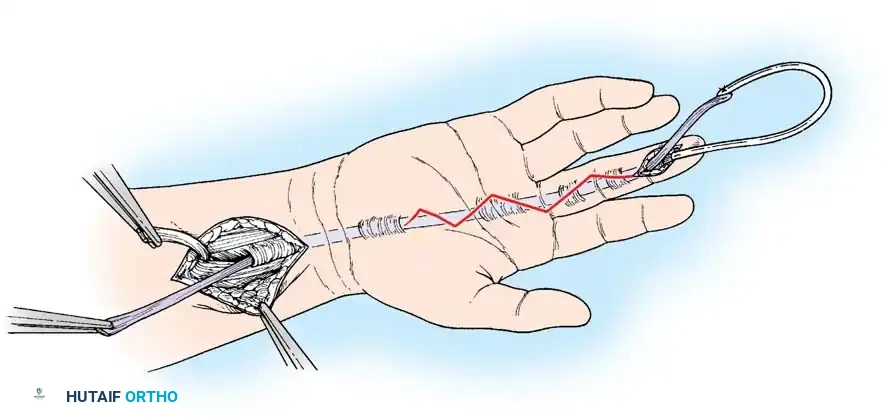
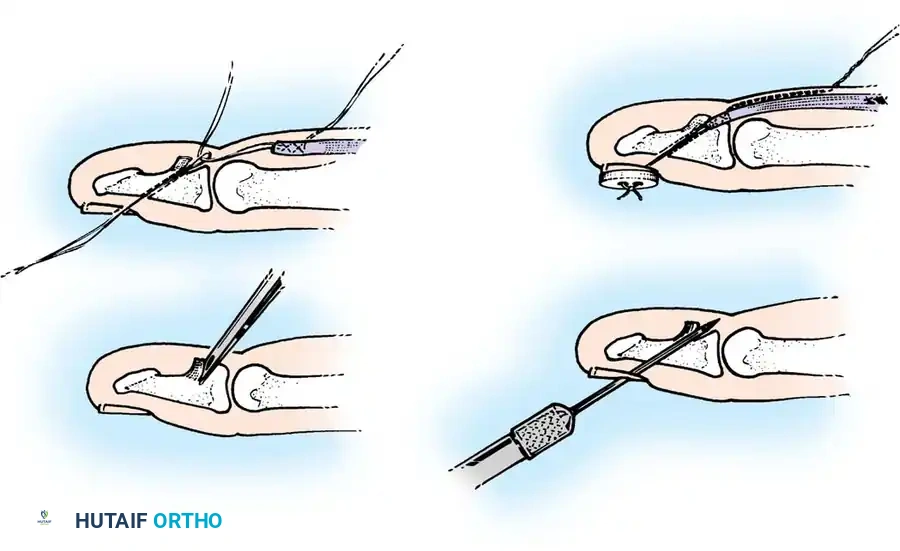
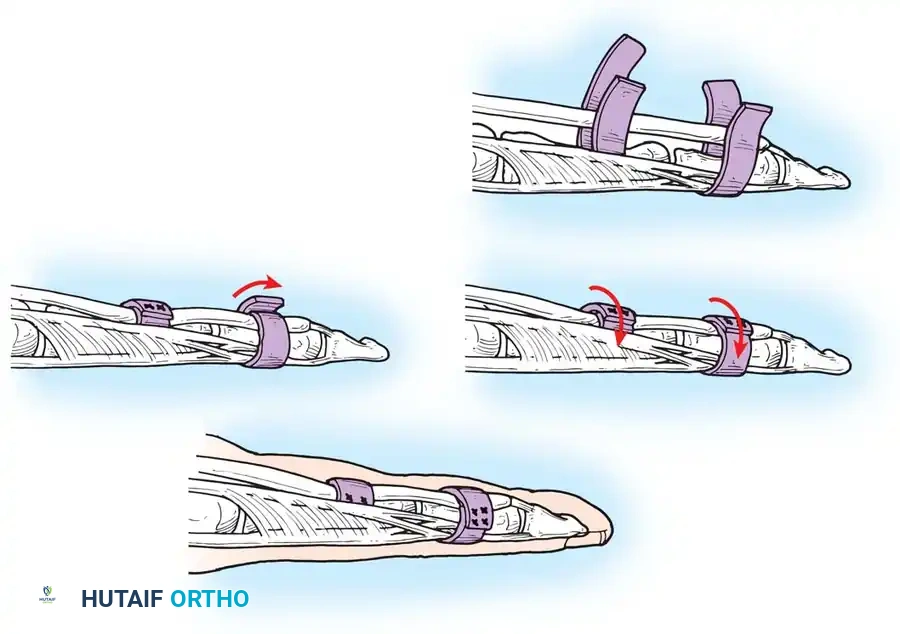
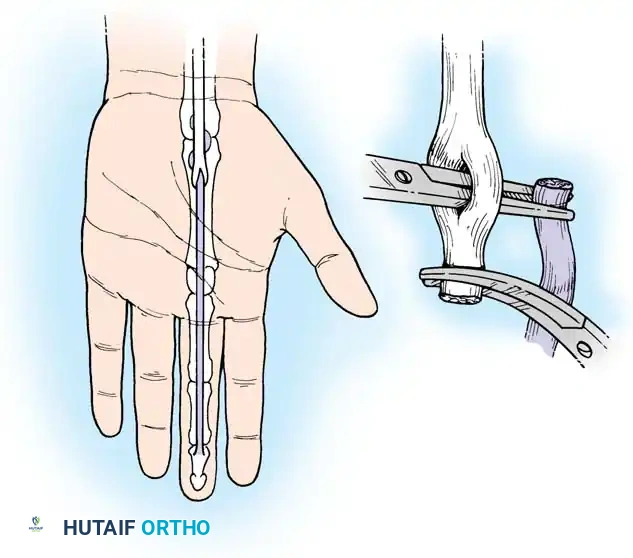
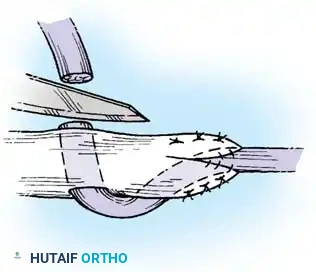
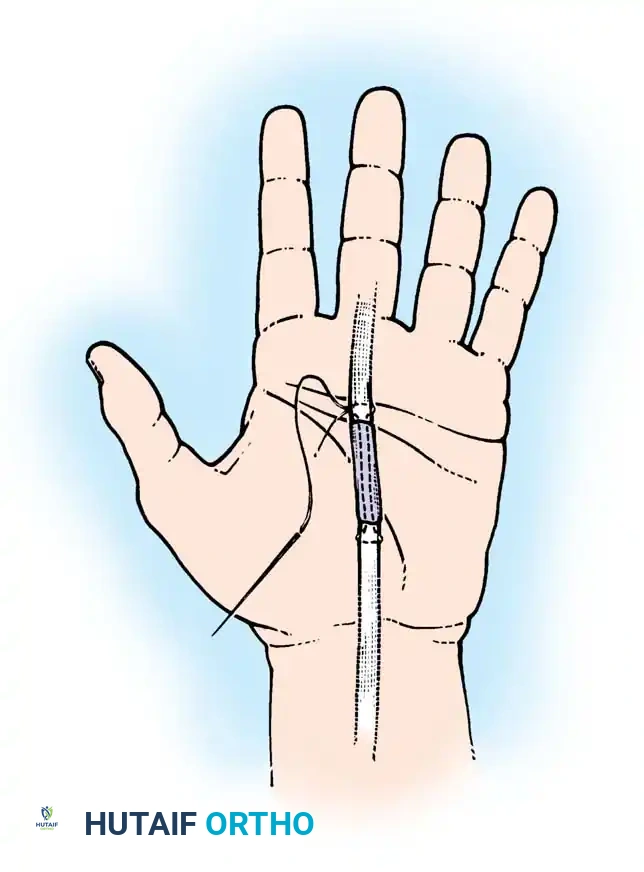
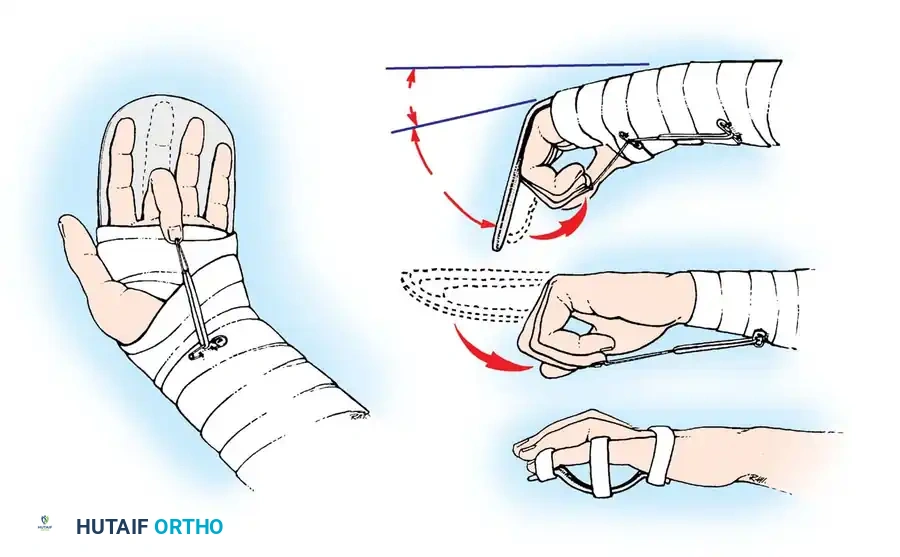
- Retrieve the proximal tendon stump. If retracted into the palm, use a pediatric feeding tube or flexible silicone catheter passed retrograde from the finger to the palm. Suture the tendon to the catheter and gently pull it distally into the finger.
- Secure the proximal stump temporarily with a transverse 25-gauge hypodermic needle passed through the tendon and adjacent pulley/bone to relieve tension during the repair.
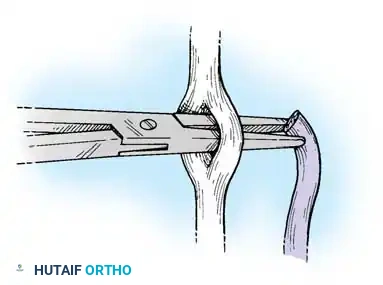
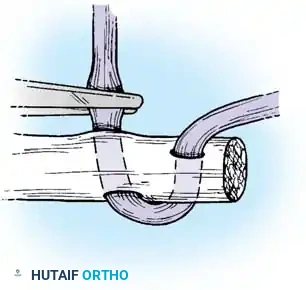
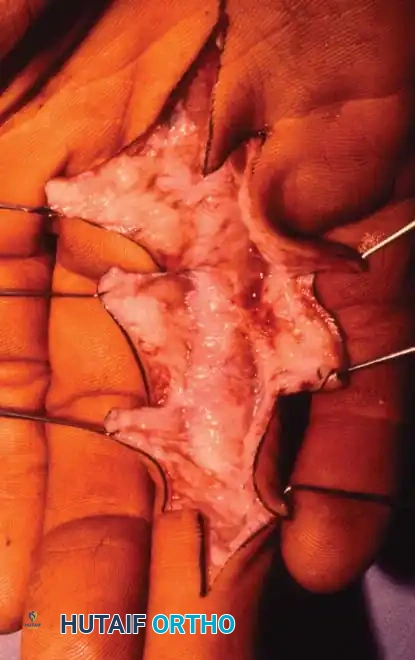
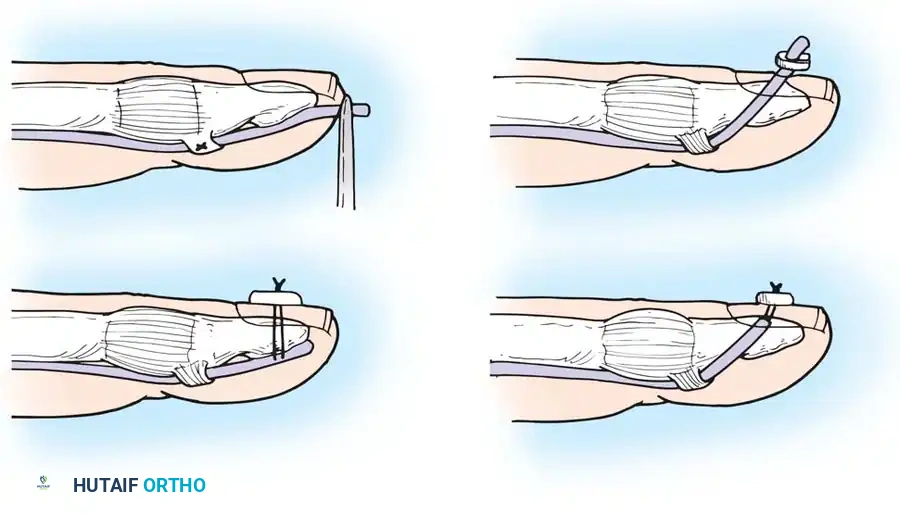
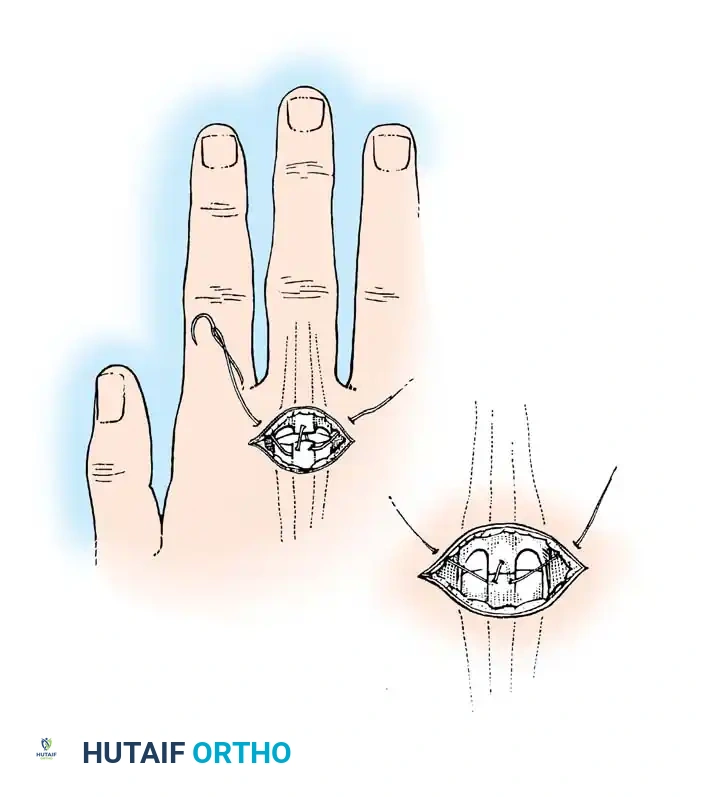
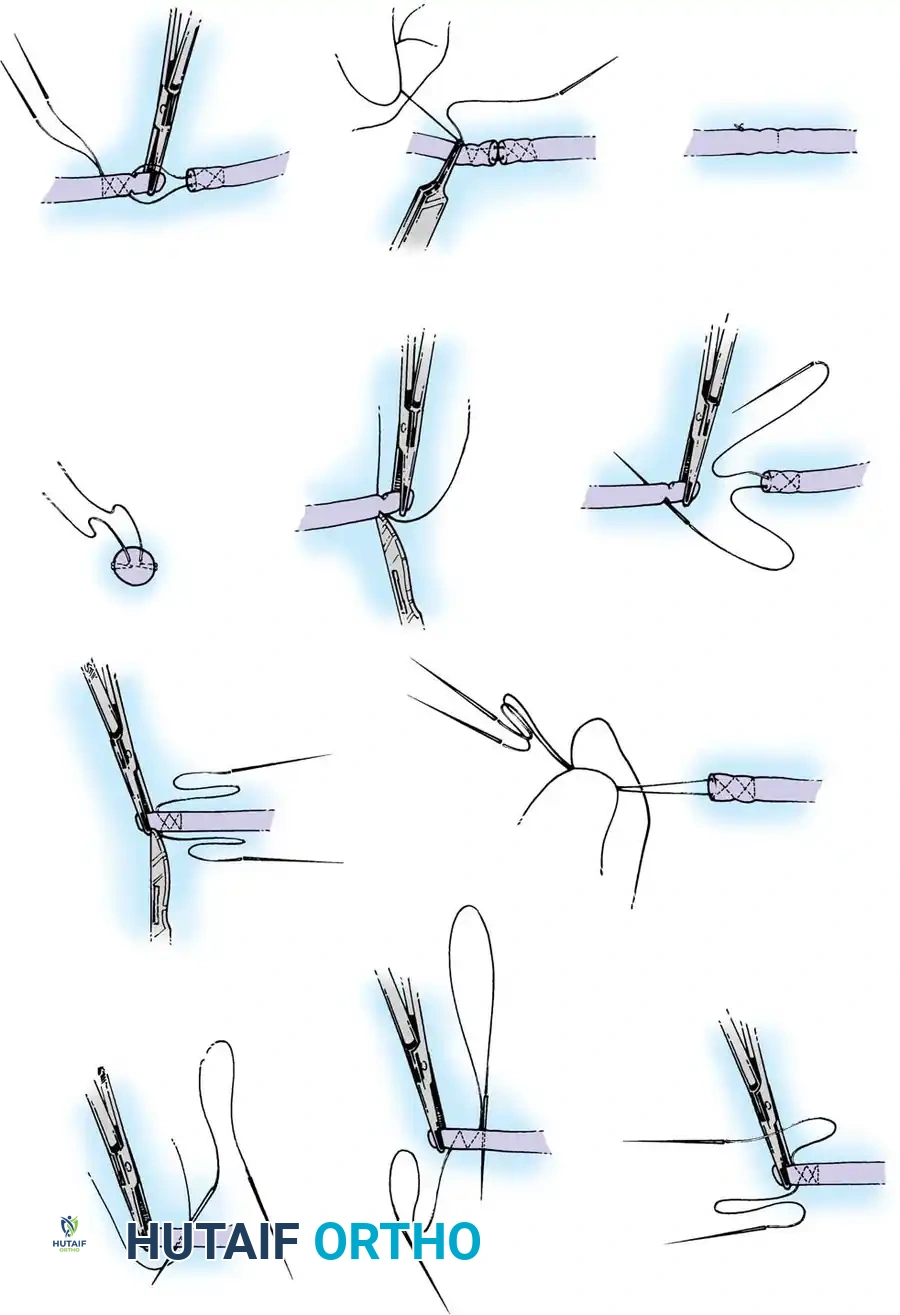
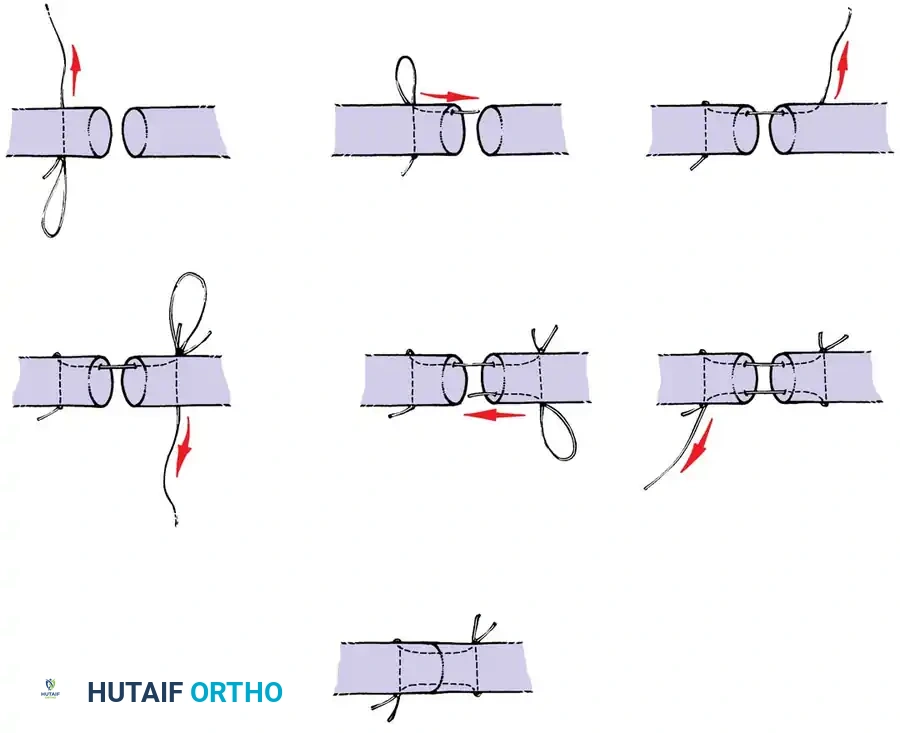
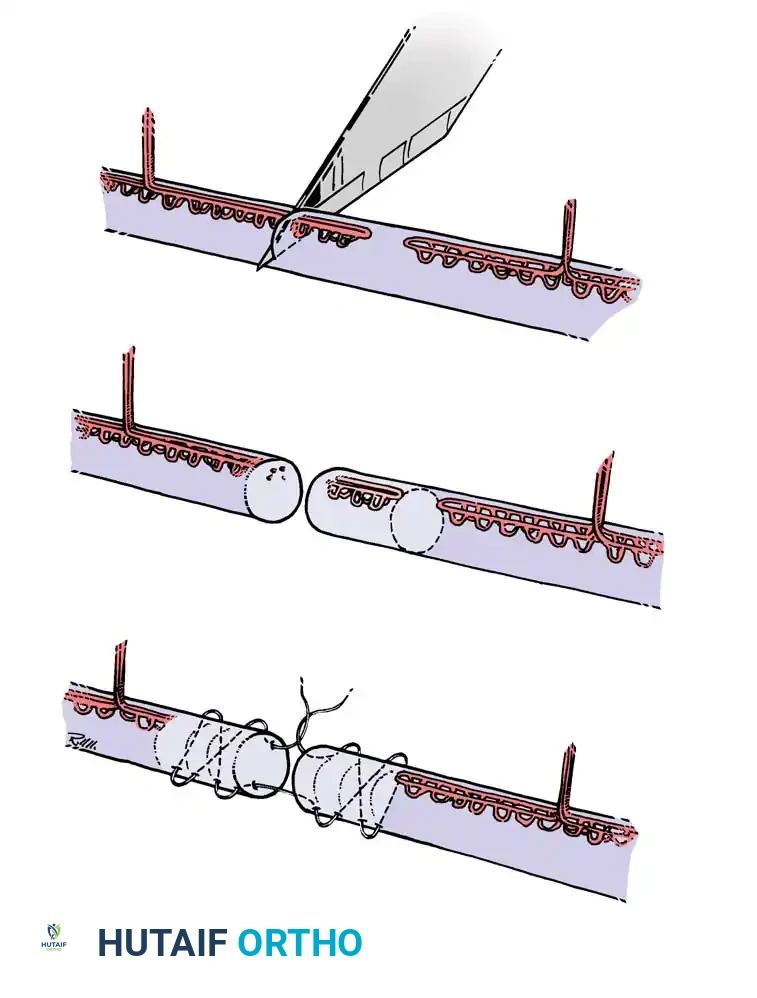
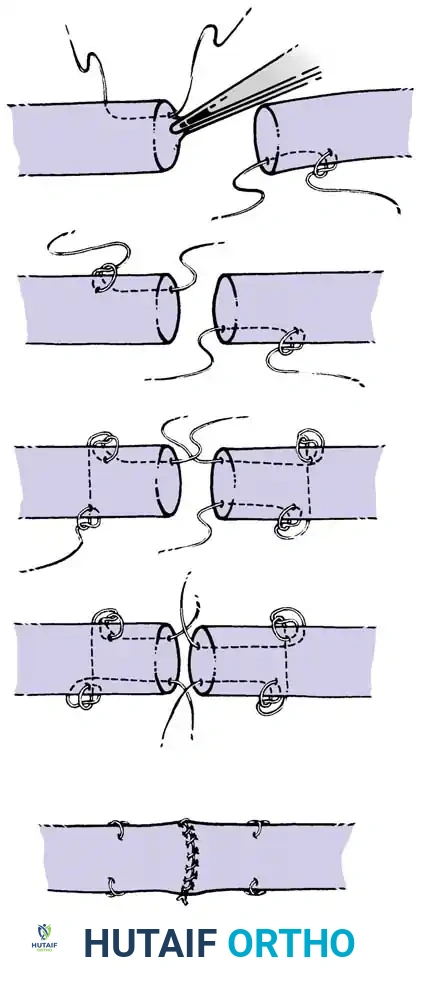
4. Core Suture Execution
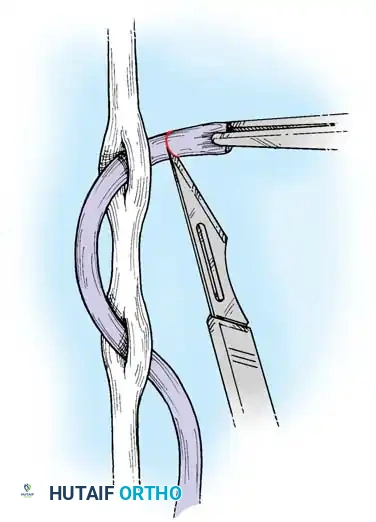
- Debride the tendon ends minimally to healthy, parallel fascicles.
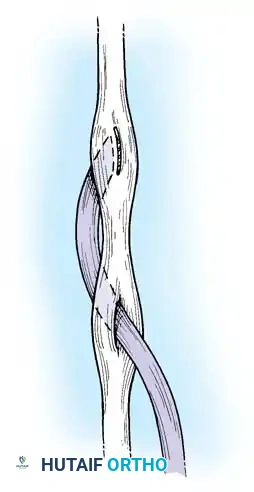
- Place a 4-strand core suture using 3-0 braided polyester. Ensure the suture purchases at least 7-10 mm of tendon on each side of the laceration.
- Place the core sutures volarly to avoid compressing the dorsal vincular blood supply.
- Tie the knots securely, ensuring they are buried within the tendon interface.
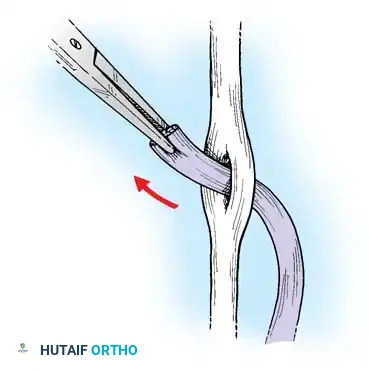
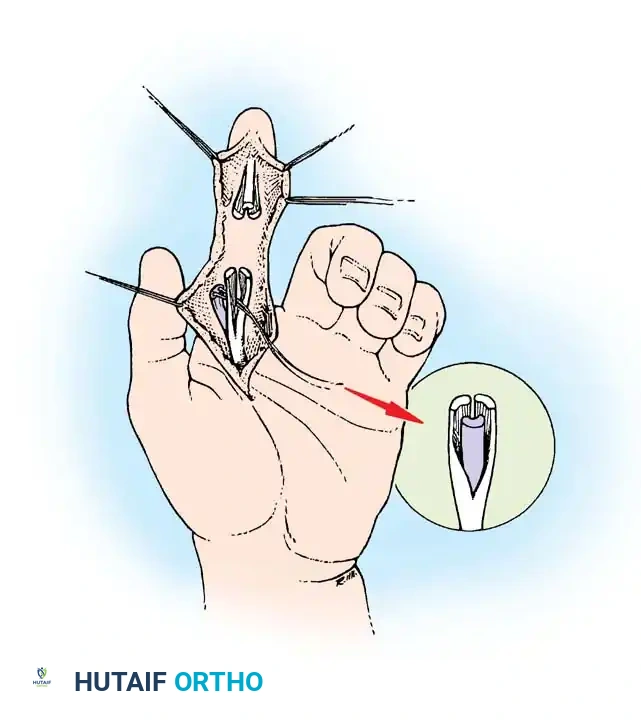
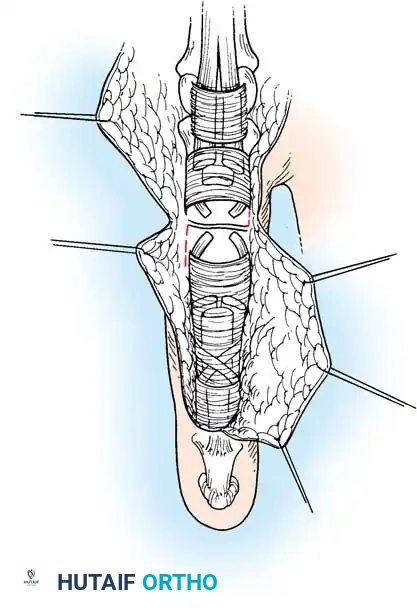
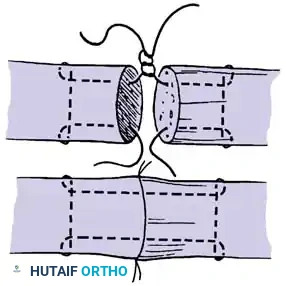
Execution of a multi-strand core repair. Tension must be perfectly balanced to prevent bunching or gapping at the repair site.
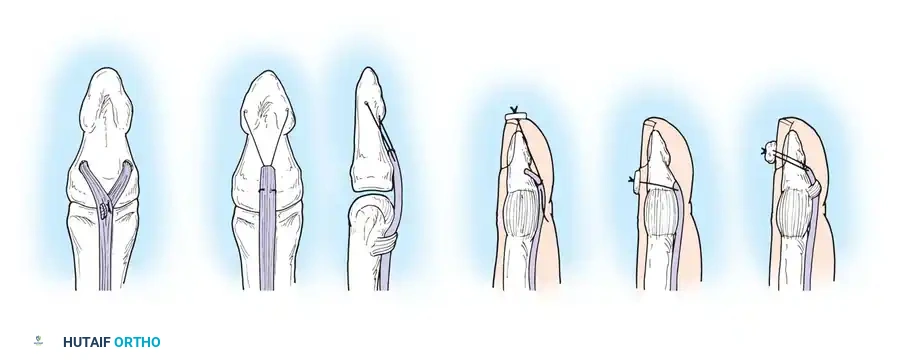
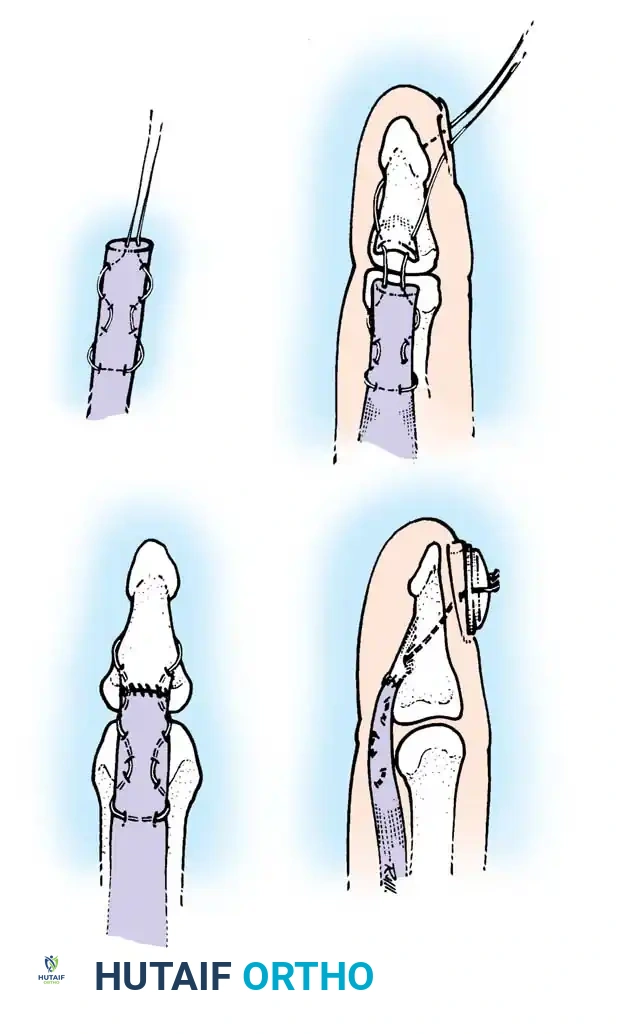
5. Epitendinous Suture and Closure
- Perform a running, locking epitendinous suture using 5-0 or 6-0 Prolene or Nylon. Ensure the edges are smoothly inverted.
- Remove the transverse stabilizing needle.
- Assess the repair by passively extending the wrist to observe the tenodesis effect. The tendon should glide smoothly beneath the A2 and A4 pulleys without catching.
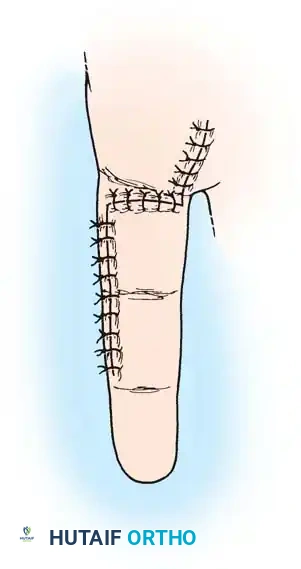
- Deflate the tourniquet, achieve meticulous hemostasis, and close the skin with non-absorbable sutures.
Postoperative Rehabilitation Protocols
The success of a flawless surgical repair can be entirely undone by improper rehabilitation. The goal is to apply controlled stress to the tendon to promote intrinsic healing and excursion while protecting the repair from rupture.
- Immobilization (0-3 days): The hand is placed in a dorsal blocking splint. The wrist is positioned in 20-30 degrees of flexion, metacarpophalangeal (MCP) joints in 50-70 degrees of flexion, and interphalangeal (IP) joints in neutral extension.
- Early Passive Motion (Duran/Kleinert): Historically, protocols relied on passive flexion (via rubber band traction or therapist manipulation) and active extension within the limits of the dorsal splint.
- Early Active Motion (EAM): Modern 4-strand and 6-strand repairs are robust enough to withstand EAM. Protocols like "Place and Hold" involve the therapist passively flexing the digits, after which the patient gently contracts the muscle to hold the position. True active flexion is gradually introduced under strict supervision, significantly reducing adhesion formation and improving final total active motion (TAM).
Clinical Pearl: Patient compliance is the most critical variable in postoperative rehabilitation. A robust 4-strand repair combined with a dedicated patient and a skilled certified hand therapist (CHT) yields the highest percentage of excellent outcomes.
Conclusion
The management of flexor and extensor tendon injuries is a testament to the precision required in orthopaedic hand surgery. By respecting the delicate vascular anatomy, utilizing biomechanically superior multi-strand core and epitendinous suture techniques, and implementing rigorous early active motion protocols, the surgeon can reliably restore function and minimize the debilitating complications of tendon rupture and restrictive adhesions.
===```
Associated Surgical & Radiographic Imaging