The Pathophysiology of Lumbar Degeneration
The evaluation and management of axial lumbar pain and spinal degeneration represent a cornerstone of orthopaedic spine surgery. To effectively treat these conditions, the spine surgeon must possess a profound understanding of the biomechanical and pathoanatomical alterations that occur within the functional spinal unit (FSU). The FSU, comprising two adjacent vertebral bodies, the intervertebral disc, and the paired facet joints, undergoes a predictable sequence of degenerative changes.
This sequence, classically described by Kirkaldy-Willis, is divided into three overlapping phases: Dysfunction, Instability, and Stabilization. Understanding this cascade is imperative to avoid both the overtreatment of self-limiting conditions and the undertreatment of progressive neurological compromise.
Phase I: Dysfunction
The dysfunction phase marks the insidious onset of the degenerative cascade, typically occurring in early adulthood (ages 15 to 45). It is characterized by subtle biomechanical alterations without gross morphological instability.
- Intervertebral Disc Pathology: The process often begins with repetitive microtrauma leading to circumferential tears in the outer annulus fibrosus. These tears can coalesce into radial tears, providing a pathway for the nucleus pulposus to migrate outward, potentially resulting in disc herniation. The loss of proteoglycans within the nucleus reduces its hydrostatic pressure, shifting axial loads directly onto the annulus.
- Facet Joint Pathology: Altered load distribution leads to facet joint synovitis and capsular hypermobility. The synovial lining becomes inflamed, and the articular cartilage begins to fibrillate.
- Clinical Presentation: Patients typically present with localized, intermittent axial back pain. The pain is often exacerbated by movement and relieved by rest. Neurological deficits are generally absent unless an acute disc herniation compresses a traversing or exiting nerve root.
Clinical Pearl: In the dysfunction phase, the primary pain generators are the highly innervated outer third of the annulus fibrosus and the facet joint capsules. Surgical intervention is rarely indicated for pure axial pain in this phase; conservative management is the gold standard.
Phase II: Instability
As degeneration progresses, the FSU enters the instability phase, typically affecting patients between 35 and 70 years of age. The structural integrity of the disc and facet joints is compromised, leading to abnormal translation and rotation.
- Pathoanatomy: Progressive disc resorption leads to a significant loss of disc height. This vertical collapse causes capsular laxity in the facet joints. The combination of annular incompetence and facet subluxation results in segmental instability.
- Neural Compromise: The loss of disc height decreases the cross-sectional area of the neuroforamen. Furthermore, dynamic subluxation can lead to lateral nerve entrapment and internal disruption of the spinal canal architecture.
- Clinical Presentation: Patients experience more frequent and severe episodes of back pain, often described as a "catching" or "giving way" sensation. Radicular symptoms may emerge due to dynamic foraminal stenosis or lateral recess stenosis.
Phase III: Stabilization
The final phase is the body's physiological attempt to arrest the abnormal motion of the unstable segment. This phase typically occurs in patients over the age of 60.
- Pathoanatomy: The chronic instability stimulates osteoblastic activity, leading to the enlargement of the articular processes (facet hypertrophy) and the formation of marginal osteophytes along the vertebral endplates.
- Stenosis: While the segment becomes stiffer and less painful from a mechanical standpoint, the hypertrophic bone and thickened ligamentum flavum encroach upon the neural elements. This results in one-level or multilevel spondylosis and central or lateral recess stenosis.
- Clinical Presentation: Axial back pain often decreases in severity, replaced by symptoms of neurogenic claudication (leg pain, heaviness, and cramping exacerbated by walking and relieved by lumbar flexion).
Clinical Evaluation of Axial Lumbar Pain
Axial lumbar pain is a ubiquitous human experience, occurring at some point in the lives of most individuals. Appropriate treatment for what can be excruciating acute pain must begin with a meticulous evaluation to rule out significant spinal pathology.
The Diagnostic Approach
Obtaining a comprehensive history and completing a rigorous physical examination is paramount. The goal is to establish a clinical diagnosis that is subsequently supported by diagnostic studies.
Surgical Warning: Matching a surgical treatment solely to the results of an imaging study—without strict clinical correlation—is fraught with difficulty and is a primary cause of failed back surgery syndrome (FBSS).
If significant pathology (e.g., fracture, tumor, infection, severe neurological deficit) is absent, the initial management should be conservative. A brief period of bed rest (1 to 3 days) combined with an anti-inflammatory regimen should be followed by a rapid progression to an active exercise program. Patients treated with this protocol generally demonstrate significant improvement within 4 to 8 weeks.
Selective Indications for Radiography
Routine radiography in the setting of acute, non-specific low back pain is discouraged due to its low diagnostic yield and unnecessary radiation exposure. Deyo and Diehl established highly effective selective ordering criteria (red flags) to identify patients who require immediate radiographic evaluation.
Box 39-1: Selective Indications for Radiography in Acute Low Back Pain
* Age > 50 years
* Significant trauma (e.g., motor vehicle accident, fall from height)
* Neuromuscular deficits (e.g., foot drop, saddle anesthesia, bowel/bladder dysfunction)
* Unexplained weight loss (> 10 lb in 6 months)
* Suspicion of ankylosing spondylitis
* History of drug or intravenous alcohol abuse
* History of cancer
* Prolonged use of corticosteroids
* Temperature ≥ 37.8ºC (≥ 100ºF)
* Recent visit (≤ 1 month) for the same problem with no improvement
* Patient seeking compensation for back pain
The Pitfalls of Advanced Imaging
The advent of Computed Tomography (CT) and Magnetic Resonance Imaging (MRI) revolutionized spinal diagnostics; however, it also introduced the danger of over-reliance on imaging.
Advanced imaging reveals disc herniations in 20% to 36% of completely asymptomatic normal volunteers. Alarmingly, this incidence increases to 76% in asymptomatic controls when matched to a population at risk for work-related lumbar pain. Therefore, mild-to-moderate nerve compression, disc bulging, and mild central stenosis do not correlate significantly with specific pain patterns. Only severe nerve compression shown on MRI or CT reliably correlates with symptoms of distal leg pain.
Psychosocial Factors and Occupational Impact
For patients who do not respond to standard treatment regimens, the spine surgeon must recognize that non-organic, psychosocial issues may be driving the clinical presentation.
The Biopsychosocial Model
Numerous studies on occupational back pain reveal that depression, occupational mental stress, job dissatisfaction, anxiety, and marital status are heavily correlated with complaints of pain and prolonged disability. In many instances, psychological stress precedes the onset of physical pain complaints.
Surgeons are notoriously poor at identifying these factors through standard clinical interviews. In a landmark study by Grevitt et al., experienced spinal surgeons successfully identified psychologically distressed patients only 26% of the time. This underscores the necessity of utilizing specific, validated psychometric instruments (e.g., the Waddell signs, Oswestry Disability Index, SF-36) during the evaluation of chronic back pain.
Return to Work Dynamics
Patients must understand that the persistence of mild pain does not indicate treatment failure. However, the treating physician must recognize a critical occupational metric: the longer a patient is limited by pain and out of work, the less likely they are to ever return to full activity.
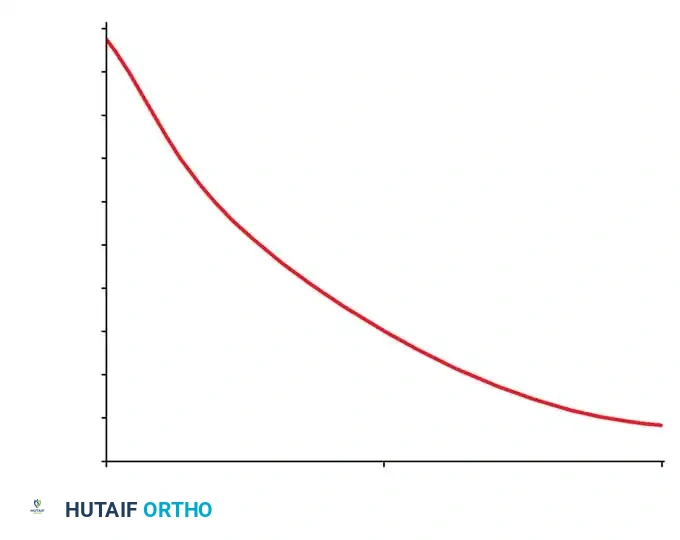
Fig. 39-5: Diminishing chance of return to work with increasing time out of work resulting from low back pain. Note the precipitous drop; when a patient is out of work for 6 months, there is only a 50% chance of returning to their previous employment.
Surgical Decision Making and Operative Principles
Careful assessment is imperative to avoid overtreatment (operating on asymptomatic MRI findings) and undertreatment (ignoring progressive myelopathy or profound instability). As Waddell noted, most patients with non-specific complaints are best managed by primary care or a multidisciplinary pain team.
However, surgical treatment provides profound benefits if it achieves one or more of the following:
1. Corrects a structural deformity.
2. Stabilizes a mechanically unstable segment.
3. Relieves neural compression.
Preoperative Planning
Once surgical indications are met (e.g., progressive motor deficit, intractable radiculopathy failing 6 weeks of conservative care, or gross radiographic instability), meticulous preoperative planning is required.
* Imaging Review: Correlate the patient's specific dermatomal pain and myotomal weakness with the exact level of compression on the MRI.
* Templating: If fusion is indicated, template pedicle screw trajectories and sizes using preoperative CT scans.
Standard Posterior Lumbar Approach: Step-by-Step
The posterior midline approach is the workhorse of lumbar spine surgery, utilized for laminectomies, discectomies, and posterior lumbar interbody fusions (PLIF).
1. Positioning and Anesthesia
* The patient is induced with general endotracheal anesthesia.
* A Foley catheter is placed, and neuromonitoring (SSEP/MEP) baseline potentials are obtained.
* The patient is carefully rolled prone onto a radiolucent Jackson spinal table. The abdomen must hang free to decrease intra-abdominal pressure, which in turn reduces epidural venous engorgement and intraoperative bleeding.
* All bony prominences are heavily padded.
2. Incision and Superficial Dissection
* Fluoroscopy is used to localize the exact surgical level.
* A midline longitudinal incision is made through the skin and subcutaneous tissue down to the lumbodorsal fascia.
* Hemostasis is achieved using bipolar electrocautery.
3. Fascial Incision and Subperiosteal Exposure
* The lumbodorsal fascia is incised strictly in the midline over the spinous processes.
* Using a Cobb elevator and electrocautery, the paraspinal musculature (multifidus and longissimus) is elevated subperiosteally off the spinous processes and laminae.
* Crucial Step: The dissection proceeds laterally to the medial border of the facet joints for a simple decompression. If a fusion is planned, the dissection is carried further laterally over the pars interarticularis to expose the transverse processes.
Pitfall: Straying lateral to the facet joint capsule during a routine laminectomy (without planned fusion) can inadvertently destroy the facet capsule, leading to iatrogenic postoperative instability.
4. Retractor Placement and Decompression
* Self-retaining retractors (e.g., McCulloch or Gelpi) are placed to maintain exposure.
* A Leksell rongeur is used to remove the spinous process and the interspinous ligament.
* A high-speed burr or Kerrison rongeur is utilized to perform the laminectomy, thinning the lamina until the ligamentum flavum is exposed.
* The ligamentum flavum is carefully detached using a curette and removed with a Kerrison rongeur, exposing the underlying dura and traversing nerve roots.
5. Discectomy (If Indicated)
* The nerve root is gently retracted medially using a Penfield dissector or nerve root retractor.
* The epidural veins are coagulated with bipolar cautery.
* An annulotomy is performed using a #15 blade on a long handle.
* Pituitary rongeurs are used to extract the herniated nucleus pulposus fragments. The disc space is irrigated to ensure no loose fragments remain.
6. Closure
* The surgical site is copiously irrigated with sterile saline.
* Hemostasis is confirmed. A subfascial drain may be placed at the surgeon's discretion.
* The lumbodorsal fascia is closed tightly with heavy, interrupted absorbable sutures (e.g., #1 Vicryl) to prevent postoperative fascial dehiscence and muscle herniation.
* The subcutaneous layer is closed in an inverted fashion, followed by a subcuticular skin closure.
Postoperative Protocol
- Mobilization: Early mobilization is critical. Patients are typically mobilized with physical therapy on postoperative day zero or one.
- Bracing: The use of a lumbar orthosis (LSO) depends on the procedure. Simple decompressions do not require bracing. Fusions may utilize an LSO for comfort and restriction of extreme motion for 4 to 6 weeks.
- Rehabilitation: A structured physical therapy program focusing on core stabilization and isometric strengthening begins at 4 to 6 weeks postoperatively.
-
Return to Work: Sedentary workers may return within 2 to 4 weeks. Heavy manual laborers may require 3 to 6 months of rehabilitation, emphasizing the need for early intervention before the psychosocial chronicity of pain sets in.
