DORSAL AND LUMBAR SPINE ARTHRODESIS: PRINCIPLES AND INDICATIONS
The indications for arthrodesis of the dorsal and lumbar spine have evolved considerably from the pioneering eras of Hibbs and Albee. Today, fusion of the thoracolumbar and lumbosacral regions for degenerative, traumatic, neoplastic, and congenital lesions is a cornerstone of operative orthopedics. While the fundamental biological goal—achieving a solid osseous union across spinal segments—remains unchanged, the indications, techniques, and postoperative care protocols vary significantly based on the pathological entity, the surgeon’s experience, and the biomechanical demands of the construct.
Many orthopedic surgeons prefer posterior arthrodesis, most commonly utilizing a modification of the intertransverse process-type fusion augmented with a large quantity of autogenous iliac bone. Internal fixation is routinely used in conjunction with posterior arthrodesis to provide immediate biomechanical stability, correct deformity, and enhance fusion rates.
Clinical Pearl: Before the utilization of any spinal instrumentation, the current status of the implant—including its specific biomechanical risks, clinical indications, and FDA approval status—must be reviewed carefully and completely with the patient during the informed consent process.
Posterolateral or intertransverse process fusions are the most frequently employed techniques. They may be used in isolation or, increasingly, in combination with an anterior interbody fusion (e.g., ALIF, LLIF, TLIF) to provide 360-degree circumferential stability. The decision to perform an interbody fusion from a posterior, anterior, retroperitoneal, or transperitoneal approach remains a topic of academic debate. Ultimately, the surgical strategy must be tailored to the specific pathological entity, the expected biomechanics of the construct, and the biological healing potential of the patient.
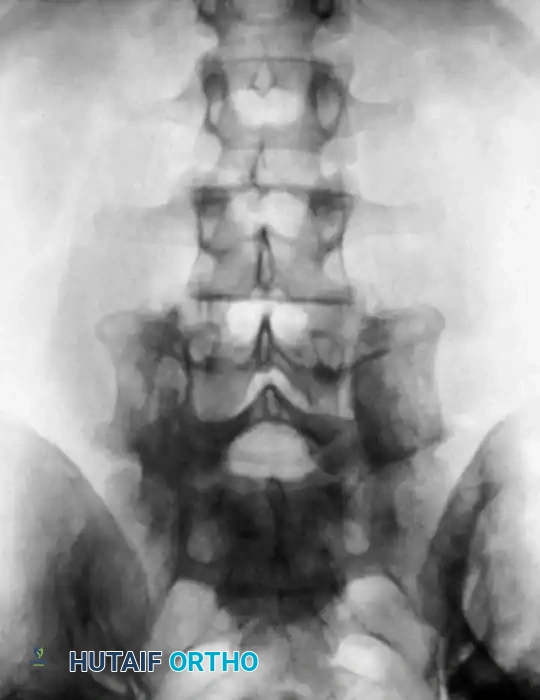
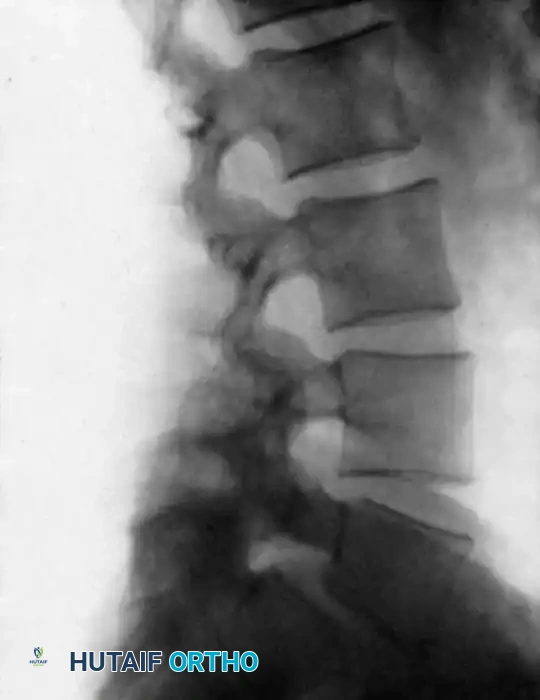
Biomechanics and Operative Planning
When formulating an operative plan, the surgeon must balance the need for neural decompression against the iatrogenic instability such decompression may create. Extensive laminectomies and facetectomies destabilize the posterior tension band, necessitating robust stabilization strategies to promote fusion.
After determining the optimal operative plan, the surgeon must decide on the execution technique—weighing the extensive exposure of traditional open techniques against the reduced soft-tissue morbidity of minimally invasive approaches (MIS). Regardless of the approach, meticulous preparation of the fusion bed is non-negotiable.
PATIENT POSITIONING AND LEVEL LOCALIZATION
Accurate visual identification of a specific vertebral level is notoriously difficult in the dorsal and lumbar spine, except when the sacrum can be definitively exposed and identified. Despite anatomical peculiarities of spinous processes, laminae, and articular facets, relying solely on visual landmarks is fraught with error.
Surgical Warning: Operating on the wrong spinal level is a catastrophic "never event." It is mandatory to obtain intraoperative marker radiographs before proceeding with irreversible steps such as decortication or decompression.
We strongly recommend a standardized method consisting of the radiographic identification of a radiopaque marker clamped securely to a spinous process within the operative field. The closer to the base of the spinous process the marker is inserted, the more accurate the identification. Cross-table lateral radiographs or anteroposterior (AP) fluoroscopic images taken on the operating table must be compared meticulously with high-quality preoperative radiographs.
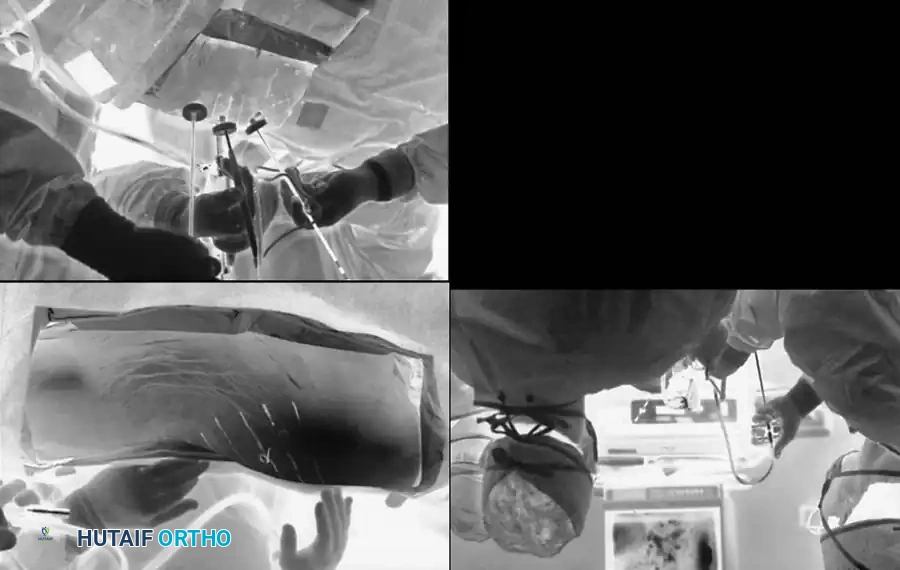
Patient positioning is equally critical. The patient must be positioned on a specialized spinal table (e.g., Jackson table) to maintain physiological lumbar lordosis and minimize intra-abdominal pressure, which in turn reduces epidural venous bleeding.
ANTERIOR COLUMN RECONSTRUCTION AND STRUT GRAFTING
While posterolateral fusions are the workhorse of lumbar spine surgery, severe anterior column deficiencies (e.g., burst fractures, tumors, or severe kyphotic deformities in the dorsal spine) often require anterior decompression and structural strut grafting. The principles of strut grafting, originally popularized in the cervical spine (e.g., the Robinson technique for cervical spondylotic myelopathy), are highly applicable to the upper dorsal and thoracolumbar spine when spinal cord or conus medullaris compression is present.
🔪 Surgical Technique: Anterior Decompression and Fibular Strut Grafting
- Decompression: Maintain the sides of the corpectomy trough in a strict parasagittal plane. When the posterior vertebral cortex has been reached and thinned to paper thickness, use a small curet to pull the bone anteriorly, detaching it safely from the posterior longitudinal ligament (PLL).
- PLL Resection: In cases of ossification of the posterior longitudinal ligament (OPLL) causing spinal cord compression, the PLL must be addressed. Develop a plane just ventral to the dura. Exercise extreme caution, as the dura can be quite attenuated and is easily torn. Small curets, 1-mm or 2-mm Kerrison rongeurs, and micro blunt hooks are essential for removing the PLL and uncovertebral osteophytes.
- Graft Preparation: On completion of the neural decompression, harvest or prepare a full segment of fibula (autograft or structural allograft) for strut placement.
- Endplate Preparation: Prepare the superior and inferior vertebral endplates to accept the graft by removing the cartilaginous endplate down to bleeding subchondral bone. Preserve the anterior portion of the vertebral cortex (the anterior lip) to prevent catastrophic anterior graft dislodgment.
- Notching and Keying: Notch the fibular graft at each end so that it keys perfectly into the prepared recesses in each endplate. Place the recess at the cephalad endplate slightly more posterior than the recess at the caudal end to facilitate easier graft insertion.
- Insertion: Apply longitudinal traction to the spinal column. Insert the graft into the superior vertebra first. Use a gentle impactor to sink the inferior portion of the graft into the caudal endplate recess, locking it into place as traction is released. Ideally, two-thirds of the graft should lie posterior to the anterior aspect of the vertebral column to optimize load-bearing.
- Fixation: Anterior plate fixation can be added to increase construct rigidity. Ensure proper plate length so that screws do not violate the graft-recipient site interface. Check the final graft position via intraoperative radiographs.
POSTERIOR ARTHRODESIS: THE HIBBS TECHNIQUE
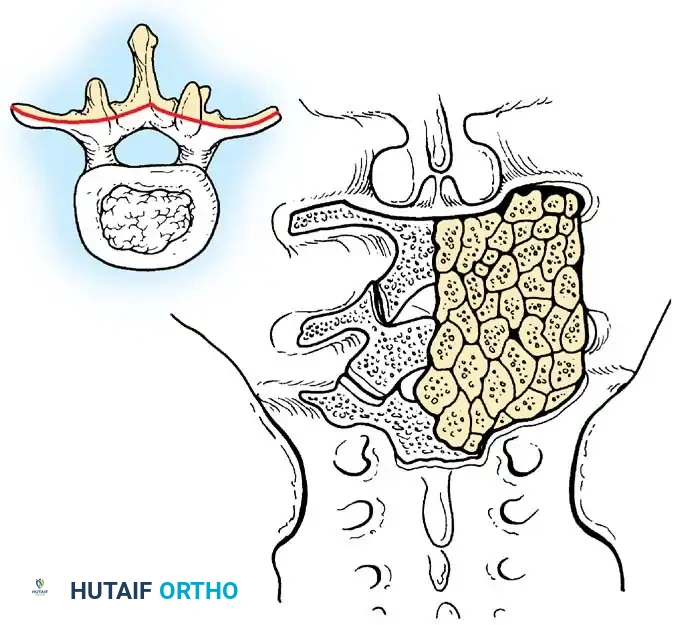
Posterior arthrodeses of the spine are fundamentally based on the principles originated by Russell Hibbs in 1911. In the classic Hibbs operation, fusion of the neural arches is induced by overlapping numerous small osseous flaps elevated from contiguous laminae, spinous processes, and articular facets. In the thoracic spine, the arthrodesis is extended laterally to the tips of the transverse processes to widen the fusion mass.
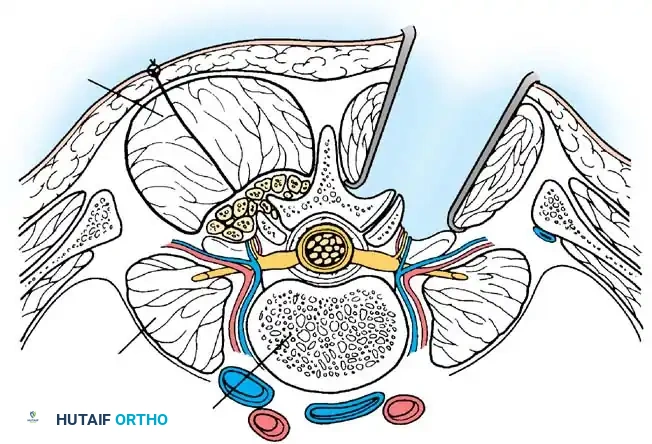
🔪 Surgical Technique: Hibbs Posterior Fusion
- Incision and Exposure: Incise the skin and subcutaneous tissues in the midline along the spinous processes. Divide the deep fascia and supraspinous ligament in line with the skin incision. Using a Kirmisson or Cobb elevator, sharply remove the supraspinous ligament from the tips of the spinous processes.
- Subperiosteal Dissection: Strip the periosteum from the sides of the spinous processes and the dorsal surface of the laminae using a curved elevator. Control bleeding meticulously with bipolar electrocautery and long, thin sponge packs (Hibbs sponges).
- Ligamentous Excision: Incise the interspinous ligaments longitudinally. Elevate the paraspinal muscles from the ligamentum flavum to expose the fossa distal to the lateral articulation, overlying the pars interarticularis and the base of the transverse process. Excise the overlying fat pad.
- Spinous Process Resection: Thoroughly denude the spinous processes of all soft tissue. Split them longitudinally and transversely with an osteotome, and remove them using a Hibbs biting forceps. Save this local autograft for later use.
- Facet Joint Preparation: Using a thick chisel elevator, strip away the capsules of the lateral articulations (facet joints). Excise the articular cartilage and cortical bone from the facets using special thin osteotomes (straight or angled at 30, 45, or 60 degrees).
- Creating the Fusion Bed: A.D. Smith emphasized that the lateral articulations of the vertebra above the planned fusion area must remain strictly undisturbed to prevent adjacent segment disease and future pain. However, all joints within the fusion construct must be completely obliterated; failure to do so jeopardizes the entire arthrodesis. Narrow the remaining facet defect by making small cuts into the articular processes parallel to the joint line, allowing thin slices of bone to separate and fill the space.
- Bone Flap Elevation: Using a gouge, cut chips from the fossa below each lateral articulation and turn them into the decorticated facet gaps. Alternatively, impact fragments of the harvested spinous processes into these gaps.
- Graft Placement: Denude the laminae of cortical bone. Place the harvested bone chips in the interlaminar spaces, ensuring intimate contact with raw, bleeding bone on both sides. Supplement with autogenous iliac crest bone graft or high-quality allograft if local bone is insufficient (e.g., in cases of spina bifida, prior laminectomy, or severe spondylolisthesis).
Clinical Pearl: When large grafts are harvested from the posterior ilium, postoperative donor site pain can be severe. Exercise extreme care to avoid injury to the superior cluneal nerves, which can lead to debilitating neuroma formation.
POSTEROLATERAL OR INTERTRANSVERSE FUSIONS
In 1948, Cleveland, Bosworth, and Thompson described a technique for repairing pseudarthrosis by placing grafts posteriorly over the laminae, lateral margins of the articular facets, and bases of the transverse processes. Watkins later refined this into the classic posterolateral fusion of the lumbar and lumbosacral spine. This technique is highly favored today because it provides a broad, highly vascularized bed for fusion while avoiding the spinal canal.
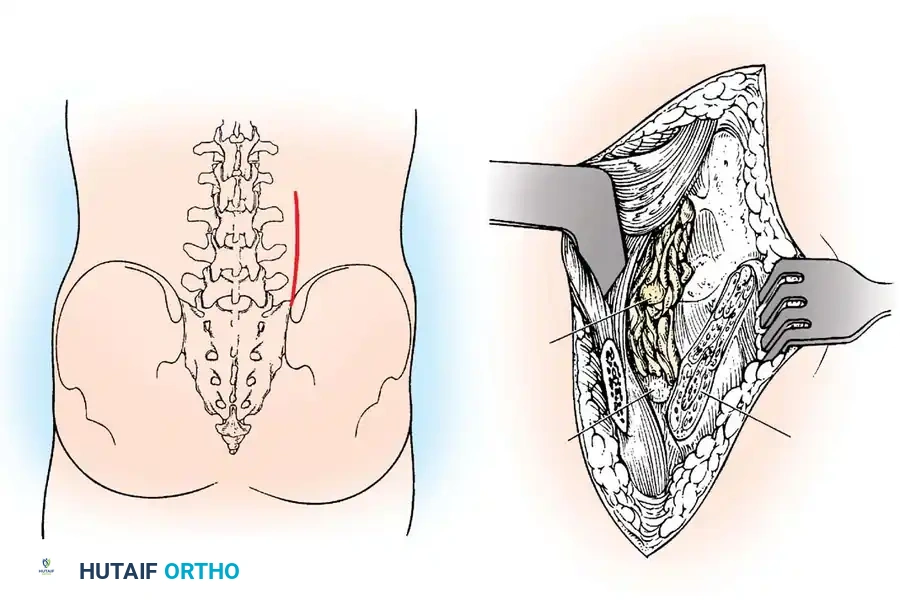
🔪 Surgical Technique: Watkins Posterolateral Fusion
- Approach: Make a longitudinal skin incision along the lateral border of the paraspinal muscles, curving it medially at the distal end across the posterior crest of the ilium. (Alternatively, a standard single midline skin incision can be used, followed by bilateral fascial incisions to mobilize the muscle bulk laterally).
- Fascial Incision: Divide the lumbodorsal fascia and establish the cleavage plane between the lateral border of the paraspinal muscles and the fascia overlying the transversus abdominis. The tips of the transverse processes can be palpated in the depths of this intermuscular interval.
- Iliac Crest Harvest: Release the iliac attachment of the muscles with an osteotome. Continue subperiosteal dissection over the posterior crest of the ilium. Harvest the crest almost flush with the sacroiliac joint, obtaining enough corticocancellous bone to provide robust grafts.
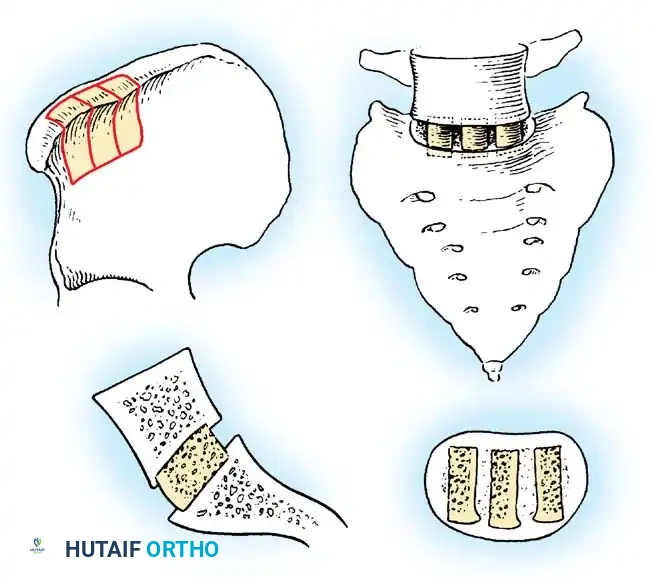
- Transverse Process Exposure: Retract the sacrospinalis muscle medially toward the midline. Denude the transverse processes of their dorsal muscle and ligamentous attachments. Expose the articular facets by excising the joint capsules.
- Decortication: Remove the cartilage from the facets with an osteotome. Level the area to allow the graft to fit snugly against the facets, pars interarticularis, and the base of the transverse processes at each level. Comminute the facets with a small gouge, turning bone chips up and down from the facet area and transverse processes.
- Graft Application: Split the resected iliac crest longitudinally. Shape the grafts to fit the prepared posterolateral bed. Impact the grafts firmly in place with their cancellous (cut) surfaces facing the decorticated spine. Pack additional ribbons and chips of cancellous iliac bone around the primary grafts to eliminate dead space.
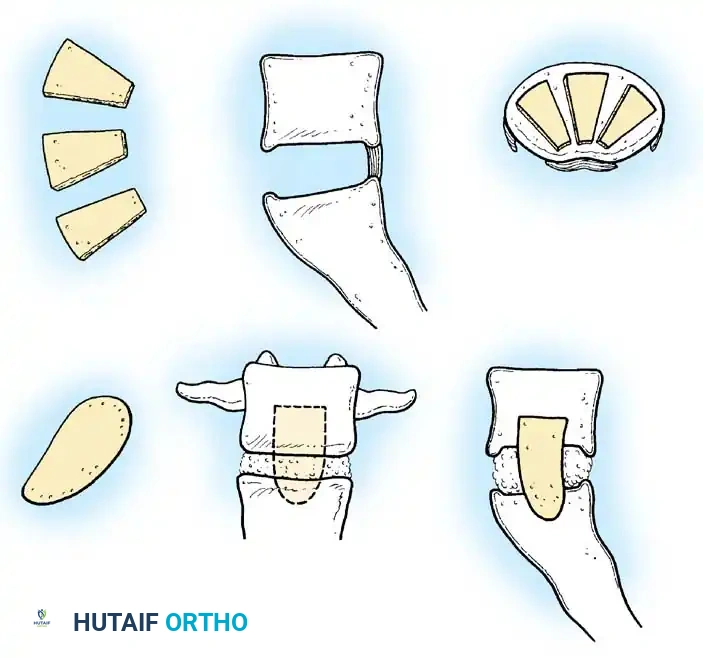
Surgical Warning: Bone grafts must not extend beyond the laminae or transverse processes of the terminal vertebrae in the construct. Projecting graft ends can impinge on adjacent mobile segments, causing severe mechanical irritation and adjacent segment pain.
- Closure: Allow the paraspinal muscles to fall back into position over the fusion mass. Suture the periosteum, ligaments, and muscles snugly over the bone chips with interrupted sutures to contain the graft. Close the subcutaneous tissue meticulously to eliminate dead space, and close the skin with a subcuticular suture.
POSTOPERATIVE MANAGEMENT AND REHABILITATION
Postoperative care is highly dependent on the rigidity of the internal fixation, the quality of the patient's bone, and the specific technique employed.
- Drain Management: We routinely utilize closed-suction drains placed deep to the fascial layer to prevent epidural hematoma formation. Drains are typically maintained for 12 to 36 hours and must be removed by 48 hours to minimize infection risk.
- Mobilization: Depending on the level of arthrodesis and the presence of rigid pedicle screw instrumentation, early mobilization is encouraged. Walking is generally allowed within 24 to 48 hours as pain permits.
- Orthotics and Bracing: The necessity of postoperative bracing remains controversial.
- Uninstrumented Fusions: If internal fixation is not used (or if screw purchase is deemed suboptimal due to osteoporosis), rigid immobilization is mandatory. For uninstrumented cervical/upper dorsal strut grafts, halo-vest immobilization may be required for up to 3 months. For lumbar fusions, a rigid Thoracolumbosacral Orthosis (TLSO) is utilized.
- Instrumented Fusions: For fusions with marked preoperative instability (e.g., burst fractures), rigid bracing is often continued for 8 to 12 weeks despite instrumentation. For highly stable constructs (e.g., single-level degenerative spondylolisthesis with robust pedicle screw fixation), bracing may be minimized or omitted entirely.
- Allograft Considerations: Postoperative immobilization and activity restrictions may need to be extended beyond 3 months when structural allografts (e.g., fibular allografts) are used, as the biological incorporation and creeping substitution of dense cortical allograft bone is significantly slower than that of autogenous cancellous iliac bone.
Through meticulous adherence to these biomechanical principles, precise surgical execution, and tailored postoperative care, orthopedic surgeons can achieve high rates of successful arthrodesis, restoring stability and function to the dorsal and lumbar spine.
📚 Medical References
- Lumbar spine arthrodesis: a comparison of hospital costs between 1986 and 1993, Am J Orthop 24:854, 1995.
- Pattee GA, Bohlman HH, McAfee PC: Compression of a sacral nerve as a complication of screw fi xation of the sacro-iliac joint, J Bone Joint Surg 68A:769, 1986.
- Pearch M, Protek I, Shepherd J: Three-dimensional x-ray analysis of normal movement in the lumbar spine, Spine 9:294, 1984.
- Peh WC, Ooi GC: Vacuum phenomena in the sacroiliac joints and in association with sacral insuffi ciency fractures: incidence and signifi cance, Spine 22:2005, 1997.
- Pinzur MS, Meyer PR Jr, Lautenschlager EP, et al: Measurement of internal fi xation device: a report in experimentally produced fractures of the dorsolumbar spine, Orthopedics 2:28, 1979.
- Post MJD, Green BA, Stokes NA, et al: Value of computed tomography in spinal trauma, Spine 7:417, 1982.
- Pringle RG: The conservative management of spinal injured patients, Semin Orthop 4:34, 1989.
- Purcell GA, Markolf KL, Dawson EG: Twelfth thoracic-fi rst lumbar vertebral mechanical stability of fractures after Harrington rod instrumentation, J Bone Joint Surg 63A:71, 1981.
- Rao S, Patel A, Schildhauer T: Osteogenesis imperfecta as a differential diagnosis of pathologic burst fractures of the spine, Clin Orthop Relat Res 289:113, 1993.
- Rea GL, Zerick WR: The treatment of thoracolumbar fractures: one point of view, J Spinal Disord 8:368, 1995.
- Rechtine GR: Nonsurgical treatment of thoracic and lumbar fractures, Instr Course Lect 48:413, 1999.
- Rechtine GR, Bono PL, Cahill D, et al: Postoperative wound infection after instrumentation of thoracic and lumbar fractures, J Orthop Trauma 15:566, 2001.
- Rhyne A III, Banit D, Laxer E, et al: Kyphoplasty: report of eighty-two thoracolumbar osteoporotic vertebral fractures, J Orthop Trauma 18:294, 2004.
- Riska EB: Antero-lateral decompression as a treatment of paraplegia following vertebral fracture in the thoraco-lumbar spine, Int Orthop 1:22, 1977.
- Rodger RM, Missiuna P, Ein S: Entrapment of bowel within a spinal fracture, J Pediatr Orthop 11:783, 1991.
- Roy-Camille R, Saillant G, Mazel C: Plating of thoracic, thoracolumbar, and lumbar injuries with pedicle screw plates, Orthop Clin North Am 17:147, 1986.
- Roy-Camille R, Saillant G, Mazel C, et al: Posterior spinal fi xation with transpedicular screws and plates, Paris, 1961, Groupe Hopitalier, La Pitié Salpétriere. Rumball K, Jarvis J: Seat-belt injuries of the spine in young children, J Bone Joint Surg 74B:572, 1992.
- Rupp RE, Ebraheim NA, Chrissos MG, et al: Thoracic and lumbar fractures associated with
