DISTAL TIBIOFIBULAR FUSION TO PREVENT PROGRESSIVE VALGUS DEFORMITY
Progressive valgus deformity of the ankle and hindfoot frequently presents as a devastating complication following trauma, chronic syndesmotic instability, or paralytic conditions. When the lateral column fails, the fibula migrates proximally or laterally, leading to lateral talar shift, incongruity of the ankle mortise, and rapid joint degeneration. In cases where standard arthrodesis techniques are insufficient due to poor local biology, massive bone loss, or recalcitrant nonunion, achieving a robust distal tibiofibular fusion is paramount to halting deformity progression.
To bridge critical defects and provide immediate, vitalized osteogenic tissue, orthopaedic reconstructive surgeons increasingly rely on free vascularized bone grafts. The two most versatile and reliable donor sites for these complex reconstructions are the Medial Femoral Condyle (MFC) and the Iliac Crest.
FREE VASCULARIZED MEDIAL FEMORAL CONDYLE FLAP
The use of free vascularized periosteal and corticoperiosteal bone from the medial femoral condyle was first described by Sakai in 1991. Since its inception, the technique has revolutionized the management of small-to-medium recalcitrant nonunions. Fuchs et al. described its use extensively in the treatment of nonunions of the clavicle and scaphoid, demonstrating superior union rates compared to non-vascularized autografts. In the lower extremity, the MFC flap is highly effective for localized arthrodesis, including complex distal tibiofibular fusions, where local vascularity is compromised by previous surgeries or trauma.
Vascular Anatomy of the Medial Femoral Condyle
The MFC flap is uniquely versatile because it can be based off the descending genicular artery (DGA), the superior medial genicular artery (SMGA), or both, providing redundant and reliable perfusion to the corticoperiosteal segment.
The descending genicular artery branches off the superficial femoral artery approximately 13 cm above the knee joint line, just proximal to the adductor hiatus. Upon its origin, it divides into two or three of the following distinct branches:
* The osteoarticular branch
* The muscular branch (supplying the vastus medialis)
* The saphenous branch (supplying the overlying skin, allowing for a chimeric osteocutaneous flap)
Anatomical studies demonstrate that the descending genicular artery is present in approximately 90% of specimens. The critical osteoarticular branch arises about 11 cm above the knee, lying just deep or lateral to the adductor magnus tendon along the posterior aspect of the medial intermuscular septum.
The superior medial genicular artery is consistently present, arising from the popliteal artery 5 cm above the knee. It courses anteriorly to anastomose with the osteoarticular branch of the descending genicular artery, forming a robust periosteal plexus. In approximately 11% of specimens, the SMGA is the dominant arterial supply to the medial condyle.
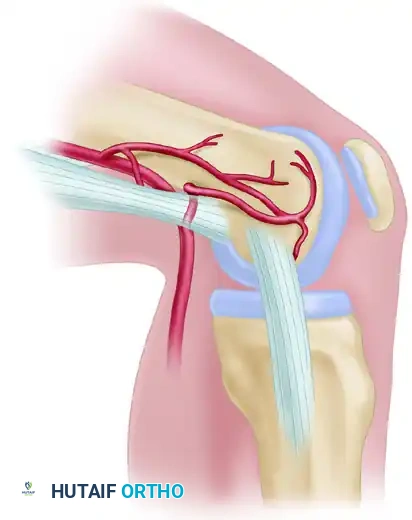

Figure 63-46: Arterial anatomy of the medial femoral condyle. The descending genicular artery and superiomedial genicular artery form a rich anastomotic network deep to the medial collateral ligament and adductor magnus tendon.
💡 Clinical Pearl: Preoperative Vascular Mapping
Always perform preoperative vascular imaging (CT angiography or MR angiography) prior to harvesting an MFC flap. While the DGA is present in 90% of patients, identifying a dominant SMGA preoperatively will alter your surgical approach and dissection trajectory, preventing inadvertent devascularization of the graft.
Biomechanics and Graft Properties
The MFC flap provides a thin, pliable layer of corticoperiosteal bone. Unlike the fibula or iliac crest, it is not inherently structural and cannot bridge massive segmental defects (>3-4 cm) under high mechanical load without supplemental rigid internal fixation. However, its immense osteogenic potential makes it the gold standard for treating avascular necrosis (e.g., Preiser disease, Kienböck disease) and recalcitrant nonunions where biological stimulation is prioritized over structural bridging.
SURGICAL TECHNIQUE: VASCULARIZED ILIAC CREST BONE FLAP
When a reconstruction requires both robust biological activity and significant structural integrity—such as bridging a large distal tibiofibular defect or reconstructing a massive segmental long bone loss—the vascularized iliac crest bone flap is the donor site of choice.
The iliac crest can be harvested based on either the Superficial Circumflex Iliac Artery (SCIA) or the Deep Circumflex Iliac Artery (DCIA). The DCIA provides a much more robust endosteal and periosteal blood supply, allowing for the harvest of large, structural bicortical grafts, whereas the SCIA is often preferred when a large overlying skin paddle is the primary requirement.
🔪 Surgical Technique 63-27: Iliac Crest Harvest
(Adapted from Taylor, Townsend, and Corlett; Daniel; Weiland et al.)
Preparation and Positioning
- Two-Team Approach: Microvascular reconstruction is highly demanding. Utilize two surgical teams working simultaneously—one preparing the recipient site (e.g., debriding the distal tibiofibular nonunion and isolating recipient vessels) and the other harvesting the flap.
- Positioning: Place the patient supine with a bump under the ipsilateral hip to elevate the iliac crest. Administer a general anesthetic with strict avoidance of peripheral vasoconstrictors.
Approach 1: Superficial Circumflex Iliac Artery (SCIA) System
The SCIA system is utilized when an osteocutaneous flap with a reliable skin paddle is required, though the bone supply is less robust than the DCIA.
- Vascular Mapping: Identify by palpation the femoral vessels. Using a sterile Doppler probe, outline the course of the superficial circumflex iliac artery paralleling the inguinal ligament toward the anterior superior iliac spine (ASIS) and iliac crest.
- Flap Design: If the SCIA system with overlying skin as an osteocutaneous flap is to be used, meticulously outline the skin flap and the course of the vessels with a surgical marker.
- Incision and Dissection: Make a vertical incision over the femoral vessels medially. Carefully dissect and identify the superficial circumflex iliac artery and the inferior epigastric vein.
- Lateral Elevation: Carry the dissection laterally from inferior to superior. Incise the fascia at the point of vessel penetration near the lateral border of the sartorius muscle.
- Protecting Perforators: Continue to elevate the skin flap superficial to the fascia as the dissection proceeds laterally. It is critical to maintain the attachments of the skin and soft tissue to the iliac crest while this dissection is proceeding to prevent shearing of the delicate musculocutaneous perforators.
- Osteotomy: When the flap has been fully elevated and the pedicle has been identified and protected, osteotomize the iliac crest using an oscillating saw, obtaining sufficient bone for the planned reconstruction.
- Transfer: Ligate the pedicle only when the recipient site is fully prepared. Transfer the bone graft to the recipient site immediately after the vessels have been transected to minimize ischemia time.
Approach 2: Deep Circumflex Iliac Artery (DCIA) System
The DCIA system is the preferred pedicle for structural bone grafts due to its excellent endosteal blood supply.
- Incision and Exposure: To use the deep circumflex iliac artery and vein, the dissection proceeds in a similar fashion initially. Identify the superficial circumflex iliac artery and vein and the inferior epigastric vein. Proceed superior to the inguinal ligament, continuing the skin incision to allow exposure paralleling the inguinal ligament.
- Pedicle Identification: Identify the deep circumflex iliac artery and vein as they arise from the external iliac artery and vein, just superior to the inguinal ligament.
- Muscle Transection: Incise transversely the external oblique, internal oblique, and transversus abdominis muscles, leaving a 2-3 cm cuff of muscle attached to the inner lip of the iliac crest to protect the vascular pedicle.
- Preperitoneal Dissection: Use blunt dissection to expose the preperitoneal fascia. Gently retract the peritoneum medially to expose the posterior aspect of the inner table of the iliac crest with its attached iliacus muscle.
- Tracing the DCIA: The deep circumflex iliac artery courses approximately 2.5 cm inferior to the iliac crest on its internal margin, running in a fibrous tunnel formed by the transversus abdominis and iliacus fasciae.
- Osteotomy: Outline the flap based on the deep circumflex artery. Perform the osteotomy beginning on the lateral surface of the iliac crest using an oscillating saw or a sharp osteotome. Preserve the overlying skin or subcutaneous tissue and its obligatory muscle cuff attachment to the iliac crest to avoid injury to the nutrient vessels.
- Graft Limitations: The natural curvature of the ilium prevents the harvesting of straight grafts much longer than 10 to 12 cm. If a straighter graft is required for a linear defect, closing wedge osteotomies of the graft can help to straighten the curve, provided the periosteal blood supply on the concave side is meticulously preserved.
⚠️ Surgical Warning: Anatomical Hazards during DCIA Harvest
During the preperitoneal dissection and isolation of the DCIA pedicle, extreme caution must be exercised to avoid injury to the spermatic cord (or round ligament in females), the external iliac vascular structures, and the genitofemoral branch of the femoral nerve. Retraction should be gentle, and bipolar electrocautery should be used sparingly near the pedicle.
POSTOPERATIVE PROTOCOL AND REHABILITATION
The success of a free vascularized bone graft depends as much on meticulous postoperative care as it does on intraoperative execution.
Flap Monitoring
- Immediate Postoperative Phase (0-72 hours): This is the most critical window for microvascular thrombosis. The flap must be monitored every 1-2 hours. If a skin paddle is present, assess color, capillary refill, and temperature.
- Doppler Ultrasound: An implantable venous Doppler or surface handheld Doppler should be used to confirm continuous arterial inflow and venous outflow. Any loss of Doppler signal or sudden change in flap turgor mandates immediate return to the operating room for pedicle exploration.
Immobilization and Weight-Bearing
- Strict Immobilization: The reconstructed limb (e.g., the ankle following distal tibiofibular fusion) must be strictly immobilized in a well-padded splint or external fixator. Shearing forces across the microvascular anastomosis or the osteotomy sites will lead to catastrophic failure.
- Weight-Bearing Restrictions: Patients are kept strictly non-weight-bearing on the affected extremity for a minimum of 6 to 8 weeks.
- Radiographic Follow-up: Serial radiographs are obtained at 2, 6, and 12 weeks. Because the graft is vascularized, it will not undergo the typical creeping substitution seen in non-vascularized grafts. Instead, primary bone healing should be observed at the host-graft interfaces. Hypertrophy of the graft in response to mechanical loading is a positive prognostic sign, typically visible by 3 to 6 months.
Donor Site Morbidity Management
- Iliac Crest: Patients frequently experience significant pain at the iliac crest donor site. Meticulous repair of the transversus abdominis and oblique muscles is mandatory to prevent postoperative abdominal hernias. Early mobilization (within the constraints of the recipient site) helps prevent deep vein thrombosis and pulmonary complications.
- Medial Femoral Condyle: The MFC donor site is generally well-tolerated. A suction drain is left in place for 24-48 hours to prevent hematoma formation. Patients may experience transient saphenous nerve neuropraxia if the sensory branches were manipulated during harvest.
CONCLUSION
The utilization of free vascularized bone grafts represents the pinnacle of orthopaedic reconstructive surgery. Whether employing the corticoperiosteal Medial Femoral Condyle flap for recalcitrant nonunions or the structural Deep Circumflex Iliac Artery flap for massive defects and complex distal tibiofibular fusions, a profound understanding of the vascular anatomy is non-negotiable. By adhering to strict microvascular principles, respecting the biomechanical limitations of the donor tissues, and executing meticulous postoperative monitoring, surgeons can achieve successful arthrodesis and limb salvage in the most challenging clinical scenarios.
