INTRODUCTION TO SPINAL OSTEOLOGY AND EPIDEMIOLOGY
A profound, three-dimensional understanding of spine anatomy is the absolute prerequisite for any surgeon undertaking operative interventions on the axial skeleton. The intervertebral disc and its intimately associated neural elements form a highly complex biomechanical and neurological unit. Pathologies affecting this unit—ranging from degenerative disc disease to acute disc herniations and spinal stenosis—constitute a vast proportion of orthopedic and neurosurgical caseloads globally.
The decision to intervene surgically, particularly with procedures such as lumbar fusion, is heavily influenced by regional practices, anatomical understanding, and evolving biomechanical evidence.
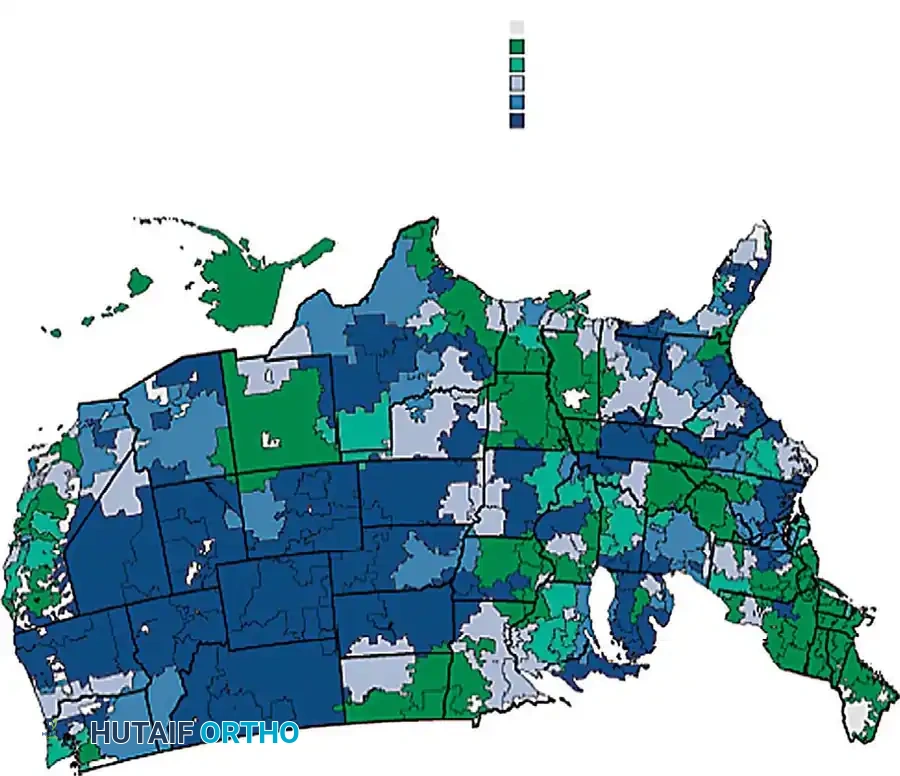
Fig. 39-1 Rates of lumbar fusion in United States between 2002 and 2003. (From Weinstein DO, Laurie JD, Olson PR, et al: United States’ trends and regional variations in lumbar spine surgery: 1992-2003, Spine 31:2707, 2006).
Understanding the microstructural anatomy, vascularity, and innervation of the spine is critical not only for diagnosing the precise pain generator but also for executing safe, tissue-sparing surgical approaches.
MICROSTRUCTURAL ANATOMY OF THE INTERVERTEBRAL DISC
The intervertebral disc is a highly specialized, avascular, and complex fibrocartilaginous structure designed to transmit loads, permit multidirectional spinal motion, and dissipate biomechanical stress. It consists of three distinct but seamlessly integrated components: the nucleus pulposus, the anulus fibrosus, and the cartilaginous end plates.
The Nucleus Pulposus
The central gelatinous nucleus pulposus possesses a highly organized extracellular matrix laid down by a relatively sparse population of cells. These cells are oval-shaped, closely resembling articular chondrocytes, and are primarily responsible for synthesizing Type II collagen and large aggregating proteoglycans (predominantly aggrecan). The high concentration of negatively charged glycosaminoglycans (GAGs) creates a massive osmotic swelling pressure, drawing water into the nucleus. This hydrostatic property allows the nucleus to act as a shock absorber, distributing axial loads radially outward against the anulus fibrosus.
The Anulus Fibrosus
The nucleus is contained peripherally by the anulus fibrosus, which is subdivided into an inner cartilaginous anulus and an outer collagenous anulus. The anulus exhibits a highly organized lamellar structure, consisting of 15 to 25 concentric sheets of collagen fibrils.
Biomechanics Pearl: The collagen fibers within each lamella run parallel to one another at an angle of approximately 30 degrees to the horizontal joint plane, but alternate in direction (roughly 120 degrees to each other) in adjacent lamellae. This alternating ply-structure provides exceptional tensile strength to resist torsional and bending forces.
The cells of the anulus are elongated, fibroblast-like, and synthesize predominantly Type I collagen, which is better suited to withstand tensile stresses. Collagen fibers from the outermost anulus (Sharpey's fibers) continue into the surrounding tissues, anchoring firmly into the vertebral body rim (ring apophysis), the anterior and posterior longitudinal ligaments, and the hyaline cartilage end plates.
The Cartilaginous End Plates
Cephalad and caudad, the disc is bounded by the cartilage end plates, which are secured to the osseous vertebral end plates by a layer of calcified cartilage. Notably, very few collagen fibers cross this calcified boundary, making the junction between the cartilage end plate and the subchondral bone a zone of mechanical vulnerability.
![Fig. 39-2 Histological fi ndings of human intervertebral discs. A, Specimen from 2.5-year-old child shows how regular concentric lamellae can be seen when specimen is viewed with polarized light. B, Specimen from neonate, showing how outer aspect of anulus fi brosus and cartilage end plate are vascularized with blood vessels (arrows) and vascular channels (asterisks) . ( A and B stained with hematoxylin and eosin; original magnifi cation, × 10 [A] and × 30 [B] .) (From Roberts S, Evans H, Trivedi J, et al: Histology and pathology of the human intervertebral disc, J Bone Joint Surg 88A:10, 2006.)](/media/hutaifortho/hutaifortho-disc-and-spine-anatomy-p2172-f498e6.webp)
VASCULARITY, NUTRITION, AND THE DEGENERATIVE CASCADE
Disc Nutrition and Avascularity
At birth, the intervertebral disc possesses a direct blood supply contained within the cartilaginous end plates and the outer anulus. However, these vessels rapidly recede during the first few years of life. By adulthood, the disc is the largest avascular structure in the human body.
The sparse cell population (accounting for only about 1% of the disc by volume) is sustained almost entirely by the diffusion of nutrients (glucose, oxygen) and the removal of metabolic waste (lactic acid) through the porous central concavity of the vertebral end plates. Histological studies demonstrate regions where marrow spaces are in direct contact with the cartilage, rendering the central portion of the end plate permeable.
Clinical Pearl: Motion and cyclical weight-bearing are essential for maintaining this diffusion gradient. Prolonged immobilization can accelerate disc starvation and subsequent degeneration. Despite its avascularity, the metabolic turnover of the disc is relatively high, though the glycosaminoglycan turnover is exceedingly slow, requiring approximately 500 days.
Pathophysiology of Disc Degeneration
Over time, the delicate nutritional balance is disrupted. Decreased permeability of the cartilage end plate—often due to subchondral sclerosis—prompts a cascade of degenerative changes:
* Desiccation: The water content of the gelatinous nucleus decreases due to an altered and depleted proteoglycan composition.
* Fibrosis and Fissuring: The nucleus assumes a more fibrous, disorganized consistency, ultimately leading to structural fissuring.
* Neovascularization: Blood vessels and nerve fibers grow deep into the disc through these outer anular fissures.
* Cellular Senescence: There is an increase in cellular proliferation (forming cell clusters) juxtaposed with an increase in apoptotic cell death. Herniated discs demonstrate a significantly greater number of senescent cells and higher concentrations of matrix metalloproteinases (MMPs) compared to non-herniated discs.
* End Plate Changes: The cartilage end plates thin and fissure, leading to reactive sclerosis of the subchondral bone (Modic changes).
NEURAL ELEMENTS AND TOPOGRAPHICAL ANATOMY
The organization of the neural elements within the spinal canal is strictly maintained from the cervical spine down through the conus medullaris and cauda equina.
Dural Sac Organization
Within the dural sac, the orientation of the nerve roots follows a highly predictable pattern. The most cephalad roots lie laterally, preparing to exit the neuroforamina, while the most caudad roots lie centrally. At all levels, the motor roots are situated ventral to the sensory roots, held in position by the delicate arachnoid mater.
The Pedicle: The Surgical Rosetta Stone
The pedicle is the fundamental anatomical landmark for understanding surgical spinal anatomy and executing instrumentation (e.g., pedicle screw fixation). The relationship of the pedicle to the exiting neural elements varies by spinal region:
- Cervical Spine: There are seven cervical vertebrae but eight cervical nerve roots. By accepted nomenclature, each cervical root exits cephalad (above) the pedicle of the vertebra for which it is named. For example, the C6 nerve root exits above the C6 pedicle (through the C5-C6 foramen).
- Cervicothoracic Junction: The C8 root exits between the C7 and T1 pedicles.
- Thoracic and Lumbar Spine: From T1 downwards, all nerve roots exit caudal (below) the pedicle for which they are named. For example, the L4 nerve root exits below the L4 pedicle (through the L4-L5 foramen).
Disc Nomenclature and Herniation Patterns
Discs are named for the vertebral level immediately cephalad (e.g., the C6 disc is between C6 and C7).
Surgical Warning: Understanding the difference between traversing and exiting nerve roots is critical for diagnosing radiculopathy.
* In the lumbar spine, a classic paracentral disc herniation at L4-L5 will impinge the traversing L5 nerve root.
* Conversely, a far-lateral (extraforaminal) disc herniation at L4-L5 will impinge the exiting L4 nerve root.
* In the cervical spine, due to the horizontal trajectory of the roots, a C5-C6 disc herniation typically impinges the exiting C6 nerve root.
SPINAL INNERVATION AND PAIN GENERATORS
The perception of discogenic and facet-mediated pain is governed by a complex, multi-level innervation network. At the level of the intervertebral foramen lies the Dorsal Root Ganglion (DRG), which houses the cell bodies of afferent sensory axons. Distal to the DRG, the spinal nerve divides into three distinct branches:
- Ventral Ramus: The most prominent branch, supplying all structures ventral to the neural canal, including the anterior longitudinal ligament (ALL) and the ventral surface of the disc.
- Dorsal Ramus: Courses dorsally, piercing the intertransverse ligament. It divides into lateral, intermediate, and medial branches. The medial branch is of paramount clinical importance as it provides primary innervation to the facet joint at its own level, as well as the facet joints immediately above and below. (This is the target for medial branch blocks and radiofrequency ablation).
- Sinu-vertebral Nerve (Nerve of Luschka): A small, recurrent filamentous nerve originating from the ventral ramus. It courses back through the intervertebral foramen to innervate the posterior aspect of the disc, the posterior longitudinal ligament (PLL), and the ventral dura.
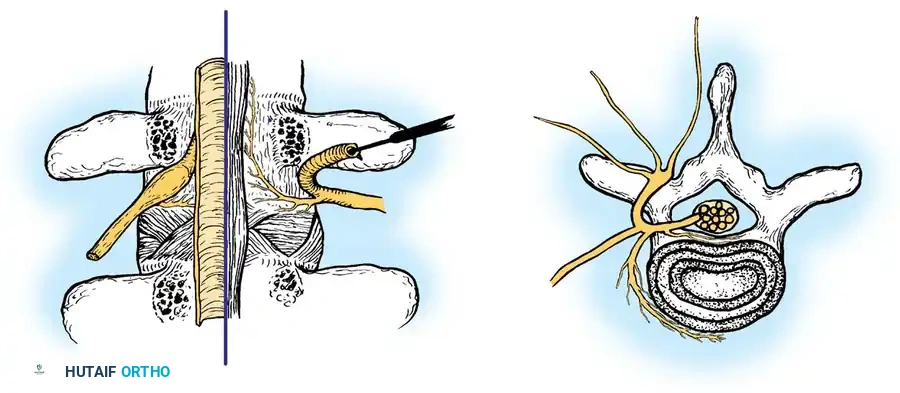
Fig. 39-4 A, Dorsal view of lumbar spinal segment with lamina and facets removed. B, Cross-sectional view of spine at level of end plate and disc. Note that sinu-vertebral nerve innervates dorsal surface of disc and posterior longitudinal ligament. Dorsal ramus arises from root immediately on leaving foramen. This ramus divides into lateral, intermediate, and medial branches. Medial branch supplies primary innervation to facet joints dorsally.
Multi-level and Sympathetic Innervation
Animal and human histological studies reveal that disc innervation is highly complex and non-segmental. The lateral anulus is innervated by fibers coursing from the index level and up to two superior levels via the sinu-vertebral nerves. Furthermore, the paravertebral sympathetic trunk provides innervation via the gray rami communicantes, connecting to the DRG up to three levels superiorly. Contralateral DRG involvement also occurs. This diffuse, multi-level innervation explains the poorly localized, referred nature of discogenic back pain.
Additionally, the basivertebral nerve (a branch of the sinu-vertebral nerve) enters the vertebral body alongside intraosseous vessels to innervate the vertebral end plates. The density of nociceptive fibers here suggests that end plate pathology (e.g., Modic changes) is a profound generator of axial back pain.
SURGICAL APPLICATIONS AND OPERATIVE PRINCIPLES
Mastery of the aforementioned anatomy is directly applied during operative interventions. The following outlines the standard principles for a Posterior Lumbar Microdiscectomy, the gold standard for symptomatic lumbar disc herniation failing conservative management.
Surgical Indications
- Progressive motor weakness or profound sensory deficit correlating with a specific dermatome/myotome.
- Cauda equina syndrome (absolute surgical emergency requiring immediate decompression).
- Intractable radicular pain failing 6 to 12 weeks of targeted conservative therapy (NSAIDs, physical therapy, epidural steroid injections) with corroborating MRI evidence of neural compression.
Patient Positioning
- Position: Prone on a radiolucent Jackson spinal table or Wilson frame.
- Biomechanics of Positioning: The abdomen must hang completely free. Any abdominal compression increases intra-abdominal pressure, which translates to the inferior vena cava, causing engorgement of the epidural venous plexus (Batson's plexus). Engorged epidural veins lead to excessive intraoperative hemorrhage and obscure the neural elements.
- Lordosis: Maintaining physiological lumbar lordosis is critical if fusion is planned; however, for a simple microdiscectomy, slight flexion of the hips and knees can open the posterior interlaminar space, facilitating access to the spinal canal.
Surgical Approach: Step-by-Step (Lumbar Microdiscectomy)
- Incision and Dissection: A midline incision is made centered over the fluoroscopically confirmed target level. The lumbodorsal fascia is incised just off the midline.
- Subperiosteal Exposure: Using Cobb elevators, the paraspinal musculature (multifidus and longissimus) is elevated subperiosteally off the spinous process and lamina of the cephalad vertebra, moving laterally to the medial aspect of the facet joint.
- Laminotomy: A high-speed burr or Kerrison rongeur is used to perform a hemilaminotomy. The inferior border of the superior lamina and the superior border of the inferior lamina are resected to expose the ligamentum flavum.
- Flavectomy: The ligamentum flavum is carefully detached using a curette and excised with a Kerrison rongeur. This exposes the epidural space and the underlying dural sac and traversing nerve root.
- Neural Retraction: The epidural fat is gently mobilized. The traversing nerve root is identified (often displaced posteriorly or laterally by the herniation). Using a Penfield dissector or nerve root retractor, the root and dural sac are gently retracted medially.
> Surgical Warning: Excessive medial retraction of the dural sac can cause traction injury to the nerve root or a dural tear resulting in a cerebrospinal fluid (CSF) leak. Retraction must be intermittent and gentle. - Annulotomy and Discectomy: The bulging posterior longitudinal ligament and anulus fibrosus are identified. A crucial annulotomy is performed using a #11 blade. Pituitary rongeurs are then utilized to extract the extruded nucleus pulposus fragments. The disc space is irrigated, and the neuroforamen is probed to ensure the nerve root is completely free and pulsatile.
Postoperative Protocols
- Immediate Post-Op: Patients are typically mobilized on the day of surgery. Neurological checks are performed to ensure no iatrogenic deficits.
- Restrictions: Bending, lifting (greater than 10 lbs), and twisting (the "BLT" restrictions) are strictly prohibited for the first 4 to 6 weeks to prevent re-herniation through the healing anular defect.
- Rehabilitation: Core stabilization and isometric strengthening begin at 4 weeks, progressing to dynamic lumbar stabilization exercises to support the paraspinal musculature and reduce load on the intervertebral discs.
By integrating a profound knowledge of disc microarchitecture, neural topography, and biomechanics, the orthopedic surgeon can execute precise, tissue-sparing interventions that reliably relieve radicular pain and restore spinal function.
