INTRODUCTION TO PERIPHERAL NERVE DIAGNOSTICS
The evaluation and management of peripheral nerve injuries (PNIs) demand a rigorous, evidence-based approach. Accurate diagnosis dictates not only the prognosis but also the precise timing and nature of surgical intervention. A comprehensive diagnostic algorithm relies on a synthesis of clinical examination, autonomic testing, and advanced electrodiagnostic studies. For the orthopedic surgeon, understanding the temporal evolution of Wallerian degeneration and axonal regeneration is paramount to interpreting these tests correctly and avoiding premature or delayed surgical exploration.
ELECTRODIAGNOSTIC STUDIES (EDX)
Electrophysiological studies have been utilized for decades as the gold standard to diagnose, localize, and prognosticate a wide variety of neuromuscular disorders and traumatic nerve injuries. Sequential EDX studies following a peripheral nerve insult provide the clinician with an indispensable, objective knowledge base.
The primary utilities of electrodiagnostic testing in the setting of peripheral nerve trauma include:
* Documentation of Injury: Establishing a baseline deficit, which is particularly critical in medicolegal or workers' compensation cases.
* Location of Insult: Differentiating between radiculopathy, plexopathy, and isolated peripheral nerve entrapment or transection.
* Severity of Injury: Distinguishing between neurapraxia (conduction block), axonotmesis, and neurotmesis.
* Recovery Pattern: Monitoring for subclinical signs of reinnervation before gross motor function returns.
* Prognosis: Predicting the likelihood of spontaneous recovery versus the need for microsurgical reconstruction.
* Impairment Documentation: Providing quantifiable data regarding motor unit loss.
* Pathology Identification: Differentiating demyelinating conditions from axonal loss.
* Surgical Planning: Aiding in the selection of optimal, fully innervated donor muscles for tendon transfer procedures.
The two most common and complementary electrodiagnostic methods used to study peripheral nerve injuries are Nerve Conduction Velocity (NCV) studies and Electromyography (EMG). Together, they provide a comprehensive picture of nerve conductivity, myelin integrity, axonal continuity, and muscle recruitment capability.
Nerve Conduction Velocity (NCV)
Nerve conduction studies evaluate the speed and amplitude of electrical signals propagating along a peripheral nerve. Stimulation of a peripheral nerve by a surface electrode placed on the skin overlying the nerve readily evokes a measurable response.
Standard nerve conduction techniques include:
* Orthodromic Motor Studies: Stimulating the nerve and recording the compound muscle action potential (CMAP) over the target muscle belly.
* Antidromic and Orthodromic Sensory Studies: Evaluating the sensory nerve action potential (SNAP). Preservation of the SNAP in the presence of anesthesia suggests a preganglionic injury (e.g., nerve root avulsion), as the dorsal root ganglion remains in continuity with the distal axon.
* Retrograde Studies (F-Wave and H-Reflex): F-wave studies are especially useful for investigating peripheral nerve injuries that are located proximally (e.g., brachial plexus or proximal sciatic nerve) and are less accessible through standard distal stimulation techniques.
Clinical Pearl: In NCV testing, conduction velocity primarily reflects the integrity of the myelin sheath, whereas the amplitude of the action potential reflects the number of functioning axons. A severe crush injury (axonotmesis) will result in a profound drop in amplitude, while a compressive neuropathy initially presents with slowed conduction velocity due to focal demyelination.
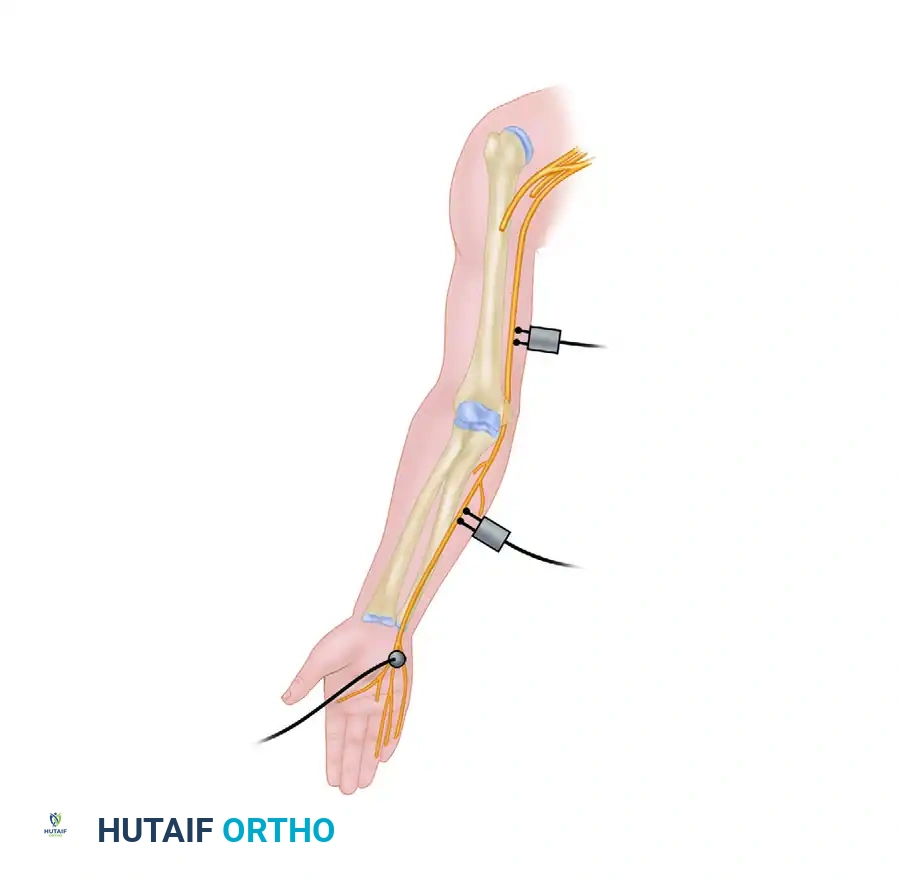
By stimulating the nerve proximal and distal to the suspected site of injury, the surgeon can map the neural injury pattern. If stimulation distal to the injury site elicits a normal response immediately after the injury, but fails to do so after 7 to 10 days, Wallerian degeneration has occurred, indicating an axonal injury (Sunderland Type 2 or higher). If distal stimulation remains normal indefinitely but proximal stimulation fails to cross the lesion, a pure neurapraxia (conduction block) is present.
Electromyography (EMG)
Myotomal sampling of the involved extremity with needle pick-up electrodes (e.g., monopolar, concentric, or single-fiber needles) yields profound insights into neuropathic pathology. The basic monopolar needle electrode samples approximately eight muscle fibers simultaneously. By assessing multiple sites within a muscle, a fair representation of the entire muscle group's integrity is obtained.
The EMG evaluation is divided into two phases: observation of the muscle at rest (insertional and spontaneous activity) and observation during volitional muscle recruitment.
The Temporal Evolution of EMG Findings
Understanding the timeline of EMG changes is critical for the orthopedic surgeon, as testing too early will yield false-negative results regarding axonal loss.
- Immediate Post-Injury Phase (Days 0-10): Needle sampling at rest is typically normal unless there has been a prior, pre-existing injury. Insertional activity lasts approximately 200 milliseconds and then ceases. During volitional effort, recruitment is decreased or absent depending on the severity of the injury. This early test is highly useful in delineating a cause-and-effect relationship in litigation cases by ruling out pre-existing denervation.
- 10 to 14 Days Post-Injury: Abnormal spontaneous rest potentials, specifically positive sharp waves, begin to appear in innervated myotomes where axonal injury and subsequent Wallerian degeneration have occurred.
- 14 to 18 Days Post-Injury: Fibrillation potentials appear. These are spontaneous, rhythmic discharges of individual denervated muscle fibers. Fibrillations of denervation will last indefinitely until the muscle either becomes reinnervated or undergoes irreversible fibrotic degeneration (typically 18 to 24 months post-injury).
- Approximately 3 Months Post-Injury: If regeneration is occurring, peripheral neural sprouting reaches the muscle. The EMG will show nascent motor unit potentials, which are typically low-amplitude, highly polyphasic configuration potentials.
- 2 to 6 Months Post-Injury: As reinnervation progresses, larger potentials are established. The motor unit potential amplitude progressively increases and remains enlarged until reinnervation is completed, at which time the configuration returns to a more normal-appearing pattern.
Surgical Warning: Although gross manual muscle testing is a practical clinical guide, significant undetected motor unit loss can occur while gross strength appears fairly well-maintained. EMG is exponentially more sensitive in detecting subtle neurological injury and early reinnervation. Many surgeons will monitor denervated myotomes for 3 months before committing to surgical exploration, relying on the appearance of polyphasic potentials to indicate spontaneous recovery.
CLINICAL DIAGNOSTIC SIGNS
The Tinel Sign
The Tinel sign (or Hoffmann-Tinel sign) is a cornerstone of the peripheral nerve examination. It is elicited by gentle percussion using a finger or a reflex hammer along the anatomical course of an injured nerve.
Technique and Interpretation:
The nerve should always be percussed in a distal-to-proximal direction. A positive Tinel sign is characterized by a transient tingling sensation (formication) felt by the patient in the specific sensory distribution of the injured nerve—not merely at the area being percussed. This sensation should persist for several seconds after stimulation.
A positive Tinel sign provides presumptive clinical evidence that regenerating axonal sprouts, which have not yet obtained complete myelinization, are progressing along the endoneurial tubes.
Correlation with Sunderland Classification:
* Sunderland Type 1 (Neurapraxia): Should not show an advancing Tinel sign because Wallerian degeneration and subsequent axonal regeneration do not occur. The nerve remains intact, and recovery is spontaneous.
* Sunderland Types 2 and 3 (Axonotmesis): A distally advancing Tinel sign should occur. The rate of this progression (approximately 1 mm per day or 1 inch per month) can be measured serially to establish a prognosis and confirm that conservative management is appropriate.
* Sunderland Types 4 and 5 (Neuroma-in-continuity and Neurotmesis): Will not show an advancing Tinel sign unless surgically repaired. A static Tinel sign at the site of injury indicates a neuroma.
With progressive regeneration, the positive response fades proximally. This occurs presumably because of progressive myelinization along the more proximal part of the regenerated segment, rendering it less mechanically irritable.
Pitfall: A few regenerating sensory fibers can result in a strongly positive Tinel sign. Therefore, the presence of an advancing Tinel sign cannot be construed as absolute evidence that motor fibers are regenerating adequately, nor does it guarantee that significant functional sensory return will be achieved. It is an encouraging sign, but must be correlated with EMG data.
AUTONOMIC TESTING
Sympathetic nerve fibers travel within peripheral nerves to innervate sweat glands and regulate vasomotor tone. These unmyelinated fibers are notably resistant to mechanical trauma. Evaluating autonomic function provides critical clues regarding the completeness of a nerve lesion.
Sweat Tests
The presence of sweating within the autonomous sensory zone of an injured peripheral nerve reassures the examiner, suggesting that complete interruption of the nerve has not occurred.
- Ophthalmoscope Technique: As described by Kahn, preservation of sweating can be determined simply and non-invasively by observing beads of sweat on the papillary ridges through the +20 diopter lens of a standard ophthalmoscope.
- Iodine Starch Test (Quinizarin Powder): This time-honored test consists of dusting the extremity with quinizarin powder. Sweating is then induced (e.g., via heat or physical exertion). The powder remains dry and light gray throughout the denervated (anhidrotic) area, while it assumes a deep purple color in areas of normal sweating.
- Ninhydrin Print Test: Recommended by Aschan and Moberg, this method assesses sweat patterns in the hand by detecting amino acids present in sweat, leaving a permanent fingerprint record of sudomotor function.
Skin Resistance Test
The skin resistance test is an objective method of evaluating autonomic interruption utilizing a Richter dermometer.
The autonomous zone with an absence of sweating (anhidrosis) lacks the conductive properties of moisture, thereby showing a markedly increased resistance to the passage of electrical current. Conversely, adjacent normally innervated areas possess normal resistance. Further decreased resistance in these normal areas can be elicited by exposing the patient to high external temperatures, which will induce sweating in intact zones but will not affect the denervated area. The boundary outlined by the Richter dermometer roughly approximates the autonomous sensory zone of the injured nerve.
ELECTRICAL STIMULATION
Electrical stimulation through intact skin has historical significance and retains specific clinical utilities.
- Faradic Stimulation: Often of little value in modern practice because normally innervated muscles may fail to respond to this short-duration current. Furthermore, if a response to faradic stimulation is still present after 3 weeks, the muscles are almost always capable of voluntary contraction, rendering the test redundant.
- Galvanic Stimulation: Useful in determining chronaxy and plotting the strength-duration curve. These determinations frequently give early evidence of denervation after nerve injury and are particularly useful in following the evolution of reinnervation, which is sometimes less readily assessed by other clinical methods in the very early stages of recovery.
SURGICAL TIMING AND MANAGEMENT PROTOCOLS
The synthesis of clinical exams, EDX, and autonomic testing culminates in the critical decision of surgical timing. The management of peripheral nerve injuries is broadly categorized by the mechanism of injury: open versus closed.
Management of Open Wounds with Nerve Injury
When a peripheral nerve deficit is identified in the setting of an open wound, systemic antibiotics and tetanus prophylaxis must be administered immediately. Following the stabilization of major visceral or vascular injuries, the peripheral nerve deficit is carefully assessed.
Clean, Sharp Lacerations:
If the wound is clean and sharply incised (e.g., a knife or glass wound), and the patient's condition permits a quiet, unhurried surgical environment with adequate microsurgical equipment, immediate primary repair (within 24-48 hours) is preferred.
* Delayed Primary Repair: If the general medical condition precludes immediate surgery, or if appropriate microsurgical expertise is unavailable, the wound should be irrigated, sutured, and dressed. Neurorrhaphy is then performed electively during the first 3 to 7 days post-injury.
Contaminated, Crush, or Blast Injuries:
Open wounds caused by blasting, abrading, or crushing agents present a different biological challenge. The zone of injury extends microscopically beyond the visible macroscopic damage.
* Initial Management: The wound is cleansed and thoroughly debrided of necrotic tissue.
* Nerve Tagging: If the severed ends of the nerve are identified, they should not be primarily repaired. Instead, they are marked with non-absorbable sutures (e.g., stainless steel or prolene) to facilitate easy identification during secondary exploration.
* Prevention of Retraction: In the absence of a significant gap, loose end-to-end apposition with a single epineurial suture prevents retraction. If a segmental gap exists, the nerve ends should be tacked to adjacent stable soft tissues.
* Definitive Repair: Soft tissue coverage is prioritized. The definitive nerve repair or grafting is performed at a later date—usually 3 to 6 weeks after injury—once the soft tissues have healed, the wound is sterile, and the longitudinal extent of neuroma formation is clearly demarcated, allowing for accurate resection back to healthy fascicles.
Management of Closed Nerve Injuries
A closed injury (e.g., traction, compression, or contusion) requires meticulous serial assessment.
Observation and Rehabilitation:
After initial pain subsides, early active motion of all joints in the involved extremity must be instituted. Gentle passive range of motion (ROM) exercises are critical to prevent joint contractures, provided they do not disrupt healing fractures or tendons.
* Rationale: The extremity must be kept in an optimal physiological state. If joints become stiff or muscles undergo severe fibrotic contracture, subsequent nerve regeneration will yield a poor functional outcome.
* Splinting: Dynamic and static splinting should be used intermittently to support paralyzed joints (e.g., a dynamic extension splint for radial nerve palsy) and prevent overstretching of denervated muscles.
Timing of Surgical Exploration in Closed Injuries:
When closed fractures are complicated by peripheral nerve deficits (e.g., a humeral shaft fracture with radial nerve palsy), the initial approach is expectant observation.
* Monitoring: The progress of reinnervation is evaluated with periodic EMG and NCV studies (typically at 6 weeks and 12 weeks) alongside frequent clinical exams (advancing Tinel sign).
* Exploration: If there is no clinical or electrodiagnostic evidence of reinnervation by 3 to 4 months, surgical exploration is indicated. Intraoperative nerve-evoked potentials and direct nerve stimulation are utilized to assess neuromas-in-continuity and determine the need for neurolysis versus resection and grafting.
* The Iatrogenic Exception: Conversely, if a nerve deficit immediately follows the closed manipulation or casting of a fracture in the absence of a prior deficit, the nerve may be entrapped in the fracture site. In this specific scenario, early surgical exploration is strongly favored to free the entrapped nerve and prevent irreversible neurotmesis.
===
