DIAGNOSIS AND CLINICAL EVALUATION OF SPINAL INFECTIONS
The diagnosis of spinal infections—encompassing pyogenic vertebral osteomyelitis, spondylodiscitis, and epidural abscesses—requires a meticulous, multidisciplinary approach. Because the clinical presentation is often insidious, a high index of suspicion is paramount for orthopedic surgeons and neurosurgeons. Delayed diagnosis frequently results in extensive biomechanical destruction of the anterior column, progressive sagittal deformity, and devastating neurological compromise.
Physical Examination and Clinical Presentation
The most common and reliable presenting symptom of spinal infection is localized back pain. According to seminal studies by Ross and Fleming, pain is the primary symptom in 85% of patients. This pain is typically mechanical, exacerbated by changes in position, ambulation, and weight-bearing activities, and ranges in intensity from mild, deep-seated aching to extreme, intractable agony.
Constitutional symptoms, such as fever, chills, and night sweats, may be absent in up to 50% of patients, particularly in the elderly or immunocompromised. Clinicians must be vigilant for signs of abscess pointing, which may manifest at a significant distance from the primary infectious focus.
Clinical Pearl: A paraspinal abscess commonly presents as a swelling in the groin below the Poupart ligament (inguinal ligament). This occurs due to the anatomical tracking of purulent material along the fascial planes of the psoas muscle. Patients may present with a hip flexion contracture and a positive psoas sign.
Standard provocative tests, such as the straight leg raise (SLR), are usually not helpful; they may be entirely negative or elicit only localized back pain rather than true radicular leg pain. Neurological findings are rarely radicular in nature; instead, they more frequently involve multiple nerve groups due to epidural extension or structural collapse.
When paralysis occurs from spinal cord compression, Eismont et al. noted that central cord syndrome is present in two-thirds of patients, while anterior cord syndrome is found in one-third. Neurological symptoms are anatomically dependent, becoming more frequent at higher spinal levels (cervical and thoracic) due to the narrower spinal canal, and are least common in the thoracolumbar and lumbar regions.
Surgical Warning: Differentiation between pyogenic and caseating (tuberculous) infections by physical examination alone is extremely difficult. The development of neurological signs suggests neural compression from epidural abscess formation, vertebral body collapse, or direct neural infection. When neurological symptoms appear, they progress rapidly unless active surgical decompression and drainage are undertaken immediately.
Laboratory Studies and Biomarkers
Laboratory diagnostics are essential for both the initial identification and the longitudinal monitoring of spinal infections.
- Erythrocyte Sedimentation Rate (ESR): The ESR is the most reliable traditional laboratory study to evaluate and clinically monitor osteomyelitic disc space infection. While not specific, it is highly sensitive. The ESR is elevated in 71% to 97% of children with vertebral osteomyelitis. In adults, 37% present with rates >100 mm/h, and 67% have rates >50 mm/h.
- C-Reactive Protein (CRP): CRP is a superior early indicator of infection. Its primary value lies in its rapid kinetic response; it normalizes quickly with the resolution of infection. Thelander and Larsson demonstrated that following uncomplicated spine surgery, CRP returns to normal by 14 days, whereas ESR may remain elevated for weeks. Richards and Emara confirmed that a secondary rise in CRP 4 to 6 days postoperatively strongly heralds a postoperative surgical site infection.
- Leukocytosis: White blood cell (WBC) counts are not especially helpful in diagnosing spinal infection. Counts may be normal or even decreased in infants, the elderly, and debilitated patients.
- Blood Cultures: Positive blood cultures during febrile episodes can identify the causative organism in up to 50% of cases, potentially obviating the need for an invasive biopsy.
- Immunocompromised Profiles (HIV): CD4 counts are predictive of the clinical course in HIV-positive patients. Casado et al. and Weinstein and Eismont found that spinal infection (discitis/osteomyelitis) typically occurs when the CD4 count is ≥ 200/mL, whereas atypical osteoarticular and soft-tissue infections predominate when the CD4 count falls below 200/mL.
ADVANCED IMAGING TECHNIQUES
The purpose of diagnostic imaging is the confirmation of the clinical impression and the precise anatomical mapping of the destructive process. No single diagnostic technique is 100% effective, and findings depend heavily on the chronicity of the infection.
Plain Radiographs
Plain radiographs of the involved spinal segment remain the standard initial study. However, radiographic findings lag behind clinical symptoms, typically appearing 2 weeks to 3 months after the onset of infection.
Early radiographic findings include subtle disc space narrowing and loss of the normal contour of the vertebral endplates.
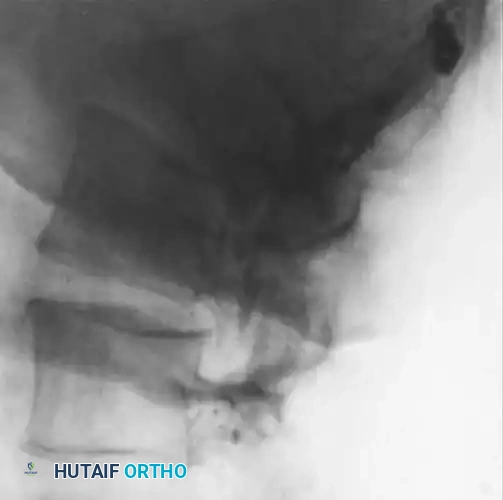
Fig. 40-7A: Early radiographic appearance of spinal osteomyelitis demonstrating minimal disc space narrowing, but a relatively normal endplate and subchondral region.
As the infection progresses (typically 2 to 8 weeks), the destructive cascade becomes evident. Findings include profound disc space narrowing, endplate destruction, defects in the subchondral bone, and reactive hypertrophic (sclerotic) bone formation.
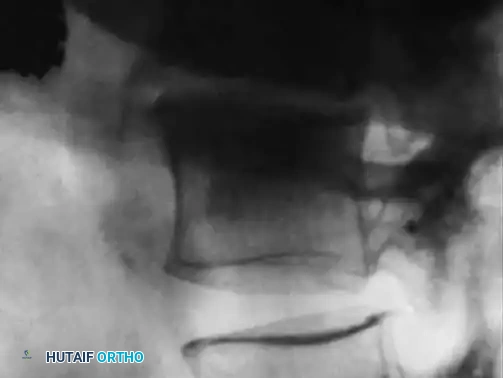
Fig. 40-7B: Progressive infection showing a severe reduction of disc height associated with the destruction of the endplate and the development of subchondral lytic defects.
Late radiographic findings (often taking up to 2 years to fully manifest) include vertebral body collapse, segmental kyphosis, and spontaneous bony ankylosis.
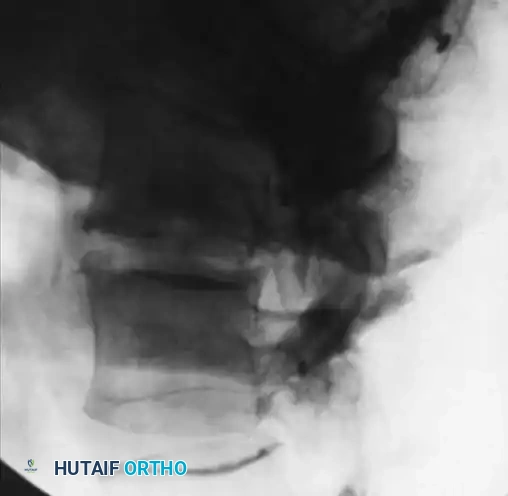
Fig. 40-7C: After successful treatment and eradication of the infection, note the sclerotic vertebra and the formation of a large bridging osteophyte indicating spontaneous fusion.
Diagnostic Pitfall: The only definable abnormality on plain radiographs and CT scans related specifically to spinal tuberculosis (Pott's disease) is fine calcification within the paravertebral soft-tissue space.
Computed Tomography (CT)
CT adds critical three-dimensional understanding to plain radiographs. It identifies paravertebral soft-tissue swelling and abscesses readily and is superior for evaluating the structural integrity of the trabecular bone. CT findings include lytic defects in the subchondral bone, multiple holes visible in cross-sectional views of the endplate, hypodensity of the disc, and disruption of the circumferential cortical bone. Postmyelogram CT can clearly define compression of neural elements by abscess or retropulsed bone fragments when MRI is contraindicated.
Magnetic Resonance Imaging (MRI)
High-quality MRI with and without gadolinium contrast is the undisputed gold standard for identifying spinal infection. Modic et al. reported an MRI sensitivity of 96%, a specificity of 92%, and an accuracy of 94% for disc space infections.
- T1-Weighted Images: Demonstrate decreased (hypointense) signal intensity in the vertebral bodies and disc spaces. The anatomical margin between the disc and the adjacent vertebral body becomes blurred or obliterated.
- T2-Weighted Images: Demonstrate increased (hyperintense) signal intensity in the intervertebral disc and adjacent infected vertebral bodies due to edema.
- Contrast Enhancement: Gadolinium-labeled DTPA enhances the delineation of epidural abscesses and hyperemic inflammatory tissue.
MRI is also invaluable for identifying primary spinal cord infections (myelitis) without epidural or bone involvement. Kastenbauer et al. noted hyperintensities on T2-weighted images involving the gray matter in bacterial myelitis.
Despite its superiority, MRI has drawbacks: degradation from patient motion (due to severe pain), claustrophobia, and absolute contraindications such as incompatible pacemakers. Furthermore, the small calcifications characteristic of tuberculous paravertebral abscesses are poorly visualized on MRI compared to CT.
Radionuclide Scanning
When MRI is contraindicated or hardware artifact obscures the region of interest, radionuclide studies are highly effective.
* Technetium-99m (99mTc) Bone Scan: Highly sensitive but lacks specificity. It shows focal uptake in delayed static images due to osteoblastic activity.
* Gallium-67 (67Ga) Scan: An excellent adjunct to bone scanning. Modic et al. reported 100% specificity when combined with 99mTc. It is particularly useful for documenting clinical improvement as uptake decreases rapidly with infection resolution.
* Indium-111 Labeled Leukocyte (111In WBC) Scan: Useful for detecting acute abscesses and differentiating infectious from noninfectious postoperative fluid collections (e.g., hematomas, seromas).
SURGICAL BIOPSY AND DECOMPRESSION: APPROACHES AND PROTOCOLS
While medical management with targeted intravenous antibiotics is the first line of treatment for uncomplicated spinal infections, surgical intervention is frequently required. The definitive diagnosis relies on the culture of the organism from infected tissue.
Surgical Indications
- Progressive neurological deficit (e.g., epidural abscess causing cord compression).
- Biomechanical instability or progressive kyphotic deformity due to anterior column destruction.
- Failure of medical management (persistent elevated CRP/ESR and intractable pain despite appropriate IV antibiotics).
- Need for definitive tissue diagnosis when blood cultures are negative and empiric therapy fails.
Biomechanics of the Infected Spine
The anterior column of the spine (vertebral bodies and intervertebral discs) bears approximately 80% of the axial load. Pyogenic spondylodiscitis primarily destroys this anterior load-bearing structure. As the vertebral bodies collapse, the spine hinges on the intact posterior elements (facet joints), leading to a progressive, rigid focal kyphosis. Surgical reconstruction must therefore focus on anterior column support (corpectomy and structural grafting) combined with posterior tension-band stabilization.
Step-by-Step Surgical Approaches
1. Image-Guided Percutaneous Biopsy
When blood cultures are negative, a CT-guided percutaneous biopsy is the standard of care to obtain tissue for culture and histopathology.
* Positioning: The patient is positioned prone on the CT gantry.
* Approach: A transpedicular or extrapedicular posterolateral approach is utilized to avoid the neural elements and thecal sac.
* Technique: Under CT guidance, a Jamshidi needle is advanced into the infected disc space or lytic vertebral body. Multiple core samples are taken for aerobic, anaerobic, mycobacterial, and fungal cultures, as well as permanent pathology to rule out neoplasia.
2. Open Anterior Debridement and Reconstruction (Corpectomy)
For extensive anterior destruction with cord compression, an anterior approach allows for direct decompression and placement of a structural strut graft.
* Positioning: Lateral decubitus position for a retroperitoneal (lumbar) or transthoracic (thoracic) approach.
* Exposure: The segmental vessels over the infected levels are ligated. The psoas muscle is mobilized posteriorly.
* Debridement: Radical debridement of all necrotic bone, infected disc material, and epidural purulence is performed until healthy, bleeding cancellous bone is reached.
* Reconstruction: A structural autograft (e.g., tricortical iliac crest, fibula) or a titanium mesh cage filled with local autograft is impacted into the defect to restore sagittal alignment and bear the axial load.
3. Posterior Instrumentation and Fusion
Because the infected anterior column is mechanically compromised, supplemental posterior pedicle screw instrumentation is often required to provide immediate rigid stabilization, promoting fusion and preventing graft subsidence.
* Positioning: Prone on a Jackson spinal table to allow the abdomen to hang free, reducing venous pressure.
* Technique: Pedicle screws are placed at least one to two levels above and below the infected segment. Decompression (laminectomy) may be performed simultaneously if a dorsal epidural abscess is present.
Postoperative Protocols
- Antimicrobial Therapy: Intravenous antibiotics are initiated immediately after deep tissue cultures are obtained. Therapy is tailored based on sensitivities and typically continued for 6 to 8 weeks.
- Immobilization: Depending on the rigidity of the internal fixation, a rigid orthosis (TLSO or cervical collar) may be utilized for 6 to 12 weeks to support the spine while bony fusion occurs.
-
Monitoring: Clinical progress is monitored via serial neurological examinations and weekly CRP and ESR levels. A steady decline in CRP is the most reliable indicator of successful eradication of the infection. Follow-up plain radiographs are obtained at 6 weeks, 3 months, and 6 months to assess the incorporation of the bone graft and the maintenance of sagittal alignment.
