CYSTIC LESIONS OF BONE: AN OVERVIEW
Cystic lesions of bone encompass a spectrum of non-neoplastic, reactive, and developmental anomalies that frequently present in orthopedic practice. While these lesions are not true neoplasms, their capacity for local bone destruction, cortical expansion, and subsequent pathological fracture demands rigorous diagnostic evaluation and meticulous surgical management. The most clinically significant entities in this category include the Unicameral Bone Cyst (UBC) and the Aneurysmal Bone Cyst (ABC), alongside less common variants such as intraosseous ganglion cysts and epidermoid cysts.
Understanding the distinct pathophysiology, imaging hallmarks, and biomechanical implications of each lesion is paramount for the practicing orthopedic surgeon to formulate an optimal, evidence-based treatment algorithm.
UNICAMERAL BONE CYST (UBC)
Unicameral bone cysts (also known as simple bone cysts) are common, fluid-filled, reactive lesions of childhood. They are widely considered to be developmental anomalies rather than true osseous tumors.
Demographics and Pathophysiology
Approximately 85% of unicameral bone cysts occur within the first two decades of life, exhibiting a 2:1 male predominance. While any bone in the appendicular skeleton can be affected, the vast majority arise in the proximal humerus and proximal femur. In the adult population, these lesions are exceedingly rare in long bones and are more frequently identified in the ilium and calcaneus.
The exact pathogenesis remains a subject of debate, but the prevailing theory suggests a localized venous lymphatic obstruction at the level of the physis. This obstruction leads to increased focal venous pressure, accumulation of interstitial fluid, and subsequent osteoclastic resorption of the surrounding cancellous bone.
Clinical Pearl: Unicameral bone cysts are most active during periods of rapid skeletal growth. They typically originate as metaphyseal lesions abutting the physis. As the child grows, normal bone is deposited between the physis and the cyst, causing the lesion to "migrate" toward the diaphysis. Cysts are classified as active when they are within 0.5 cm of the physis and latent when they have migrated further into the diaphysis.
Clinical Presentation and Radiographic Evaluation
UBCs are overwhelmingly asymptomatic unless complicated by a microfracture or a frank pathological fracture. Approximately two-thirds of patients present acutely following a fracture, which, paradoxically, can stimulate spontaneous healing of the cyst due to the influx of osteogenic fracture hematoma.
Plain radiography is usually diagnostic. Radiographs reveal a centrally located, purely lytic lesion with a well-marginated, sclerotic outline.
The cyst may expand concentrically, thinning the cortex, but it strictly respects the periosteal boundary and never penetrates the cortex unless fractured. Prominent osseous ridges on the inner cortical wall may superimpose on radiographs, giving a pseudo-multiloculated appearance.
Diagnostic Hallmark: In approximately 20% of cases presenting with a fracture, a small fragment of the thinned cortical wall breaks off and settles at the dependent base of the fluid-filled lesion. This is known as the "Fallen Fragment" sign and is pathognomonic for a unicameral bone cyst.
Surgical Management of Unicameral Bone Cysts
The primary goal of treatment is to prevent pathological fracture, promote osseous consolidation, and avoid growth arrest in active lesions near the physis.
Percutaneous Aspiration and Injection
For the majority of symptomatic or mechanically compromising UBCs, percutaneous injection is the first-line treatment.
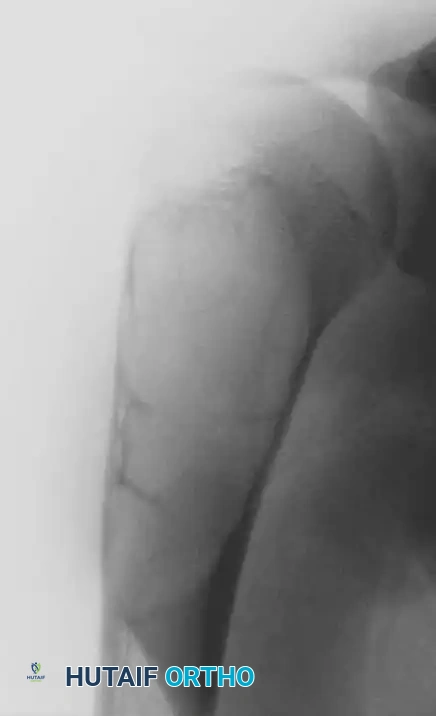
Preoperative radiograph demonstrating a large unicameral bone cyst in the right proximal humerus of a 13-year-old boy.
Step-by-Step Injection Technique:
1. Positioning and Anesthesia: The patient is placed supine under general anesthesia. Fluoroscopy is positioned to allow orthogonal views of the lesion.
2. Needle Placement: Two large-bore bone marrow biopsy needles (e.g., Jamshidi) are advanced percutaneously into the cyst under fluoroscopic guidance. One acts as an inflow/injection portal, and the other as a vent.
3. Aspiration and Venography: The characteristic serous, straw-colored fluid is aspirated. Contrast material can be injected to confirm the cyst is unilocular and to rule out rapid venous outflow, which would wash out the therapeutic injectate.
4. Cyst Disruption: The cyst lining is mechanically disrupted using the needles or a flexible curette to stimulate bleeding and an inflammatory healing response.
5. Injection: The therapeutic agent is injected through one needle until it extrudes from the vent needle, ensuring complete filling.
6. Materials Used: Autogenous bone marrow aspirate (often harvested from the iliac crest) mixed with allograft demineralized bone matrix (DBM) is highly effective. Other options include high-porosity hydroxyapatite, calcium sulfate, and medical-grade corticosteroids (methylprednisolone).
One year post-operative radiograph following aspiration and injection with bone marrow aspirate and demineralized bone matrix, showing excellent consolidation.
Surgical Pitfall: Poor prognostic factors for percutaneous treatment include a multiloculated appearance, massive size, radiographically active lesions abutting the physis, and patient age younger than 10 years. If healing is not observed after three sequential injections, open surgical intervention is mandated.
Curettage, Bone Grafting, and Internal Fixation
Open curettage and grafting are reserved for cysts refractory to injection, or for large, painful lesions in high-stress weight-bearing areas (e.g., the proximal femur) that are at imminent risk of a catastrophic fracture. In the proximal femur, prophylactic internal fixation (such as a pediatric dynamic hip screw or flexible intramedullary nails) is strongly recommended to prevent varus collapse during the healing phase. Allograft bone or commercially available bone graft substitutes are preferred to avoid the significant morbidity associated with large-volume autograft harvest in pediatric patients.
ANEURYSMAL BONE CYST (ABC)
In stark contrast to the relatively benign nature of a UBC, an Aneurysmal Bone Cyst (ABC) is a locally destructive, highly vascular, blood-filled reactive lesion. While not classified as a true malignant neoplasm, its rapid growth and aggressive local behavior can clinically and radiographically mimic a malignancy, such as telangiectatic osteosarcoma.
Demographics and Pathophysiology
ABCs predominantly affect patients younger than 20 years of age, with a slight female predominance. They can arise in any bone, but exhibit a strong predilection for the metaphyses of long bones (proximal humerus, distal femur, proximal tibia) and the posterior elements of the spine. Spinal lesions account for 15% to 20% of all ABCs and frequently extend into the vertebral body or adjacent spinal levels, posing a severe risk of neurological compromise.
The pathogenesis is believed to stem from a localized venous hemodynamic disturbance, leading to increased venous pressure, vascular engorgement, and the production of a massive local hemorrhage. This expanding hematoma causes reactive osteoclastic resorption and the formation of a thin, expanded periosteal shell.
Clinical Presentation and Advanced Imaging
Patients typically present with an insidious onset of mild to moderate pain, swelling, and a palpable mass evolving over weeks to months. Spinal lesions may present acutely with radicular pain or progressive neurological deficits due to epidural extension.
Radiographic Findings:
Plain radiographs demonstrate an eccentric, expansile, purely lytic lesion located in the metaphysis. The lesion aggressively elevates the periosteum but typically remains contained by a paper-thin shell of reactive cortical bone.
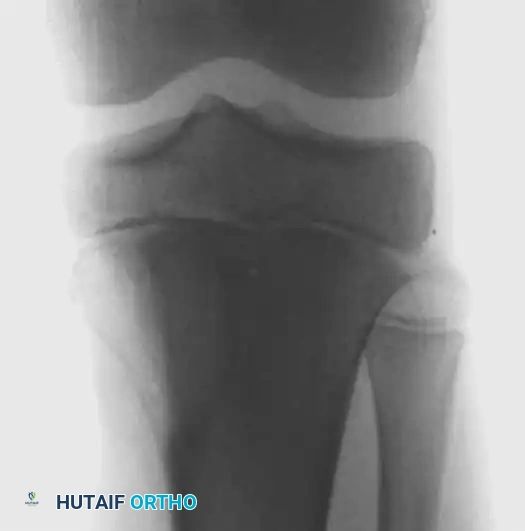
Radiolucent aneurysmal bone cyst of the left proximal tibia metaphysis in a 10-year-old boy.
Advanced Imaging (MRI and CT):
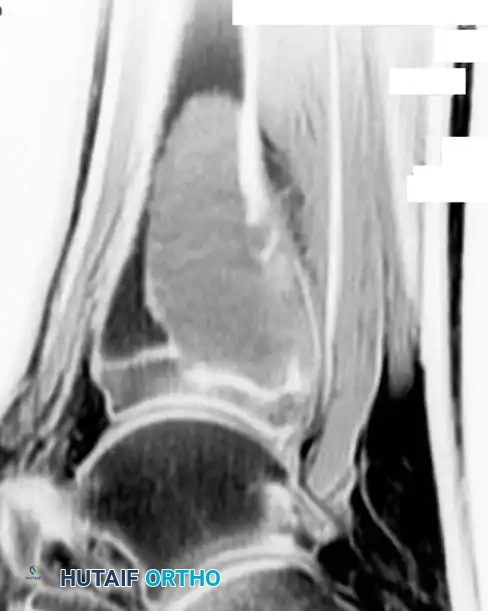
MRI is the gold standard for differentiating an ABC from a UBC or a necrotic malignancy. The hallmark MRI finding of an ABC is the presence of multiple double-density fluid-fluid levels within intralesional septations. These levels represent the settling of blood degradation products (serum layered over red blood cells) within the cavernous spaces.
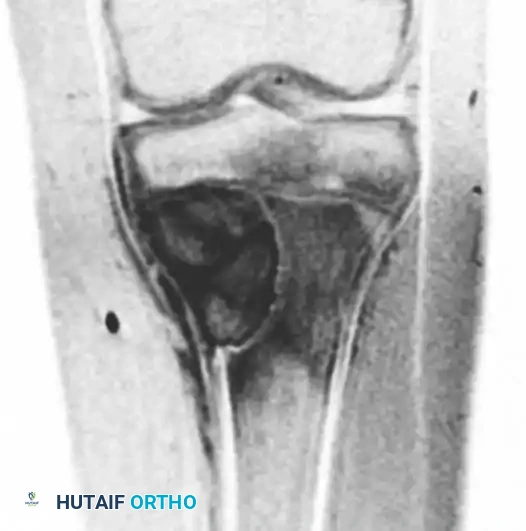
Coronal MRI image demonstrating the multiloculated nature of the lesion.
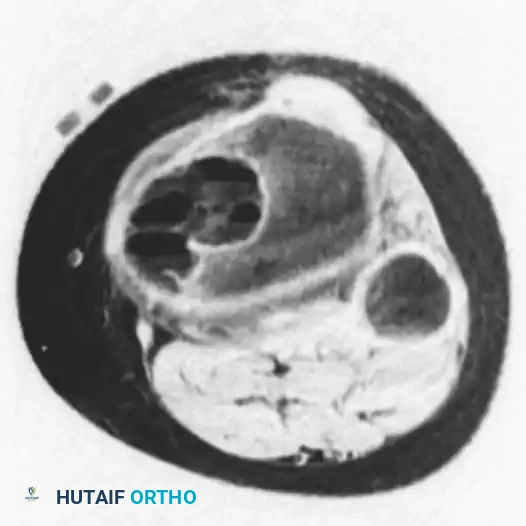
Axial MRI image clearly showing multiple fluid-fluid levels, highly consistent with an aneurysmal bone cyst.
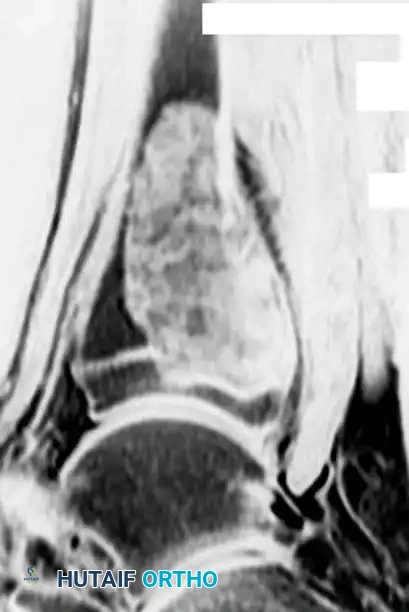
CT scanning is particularly valuable for preoperative planning in areas of complex osseous anatomy, such as the pelvis or spine, to delineate the integrity of the thin cortical rim surrounding the cyst.
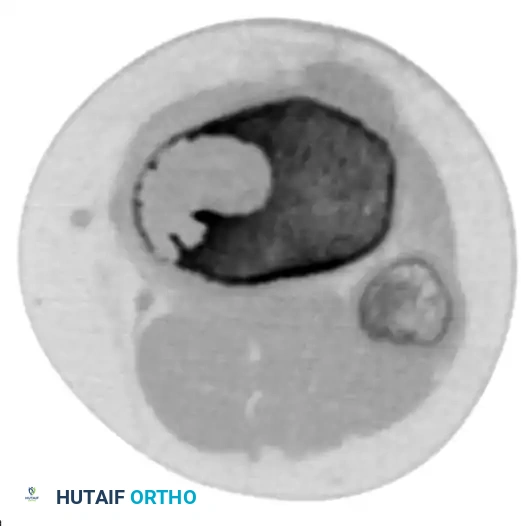
Axial CT scan demonstrating the extremely thin rim of remaining cortex overlying the expansile lesion.
Histopathology
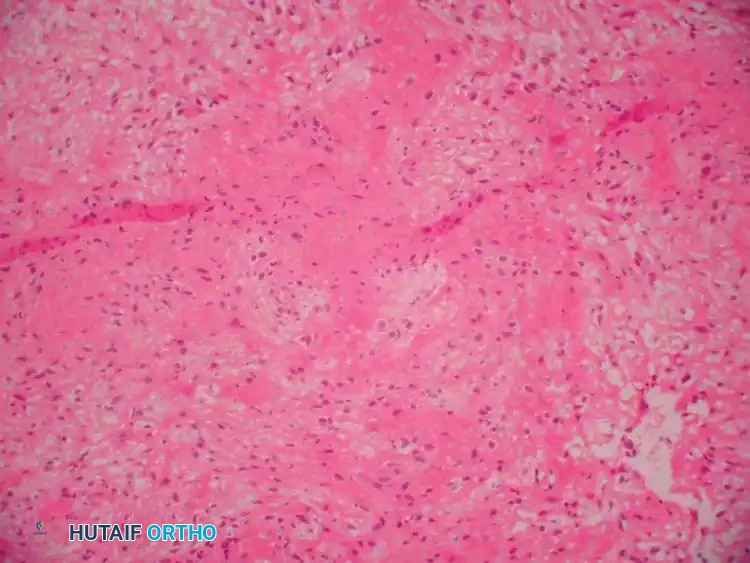
Grossly, an ABC resembles a blood-filled sponge. It is a cavitary lesion partitioned by fibrous septa. Microscopically, the tissue is highly hemorrhagic with cavernous spaces separated by a cellular stroma. The lining of these spaces lacks an endothelial layer (differentiating it from a true vascular tumor) and instead consists of compressed fibroblasts, histiocytes, hemosiderin-laden macrophages, and multinucleated giant cells.
Surgical Management: Extended Curettage
The gold standard treatment for appendicular Aneurysmal Bone Cysts is extended intralesional curettage combined with local adjuvant therapy and bone grafting.
Surgical Warning: ABCs can bleed profusely upon entry. Strict tourniquet control is mandatory for extremity lesions. For pelvic or spinal lesions where a tourniquet is impossible, preoperative selective arterial embolization (performed 24-48 hours prior to surgery) is highly recommended to minimize catastrophic intraoperative blood loss.
Step-by-Step Operative Technique:
- Exposure and Cortical Window: Following standard surgical approach and tourniquet inflation, the thin, reactive cortical shell is identified. A large cortical window is created to allow complete visualization of the entire cyst cavity.
- Gross Curettage: The blood-filled contents and fibrous septations are aggressively evacuated using large curettes.
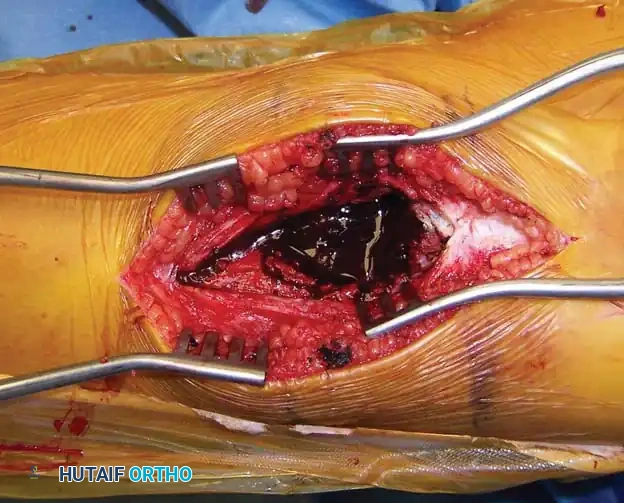
Intraoperative photograph revealing the multiloculated, blood-filled cavity characteristic of an ABC.
- High-Speed Burr (Extended Curettage): Standard curettage alone leaves microscopic reactive tissue in the bony interstices, leading to high recurrence rates. A high-speed power burr is systematically used to remove an additional 1-2 mm of bone from the entire cavity wall, extending the margin into normal-appearing cancellous bone.
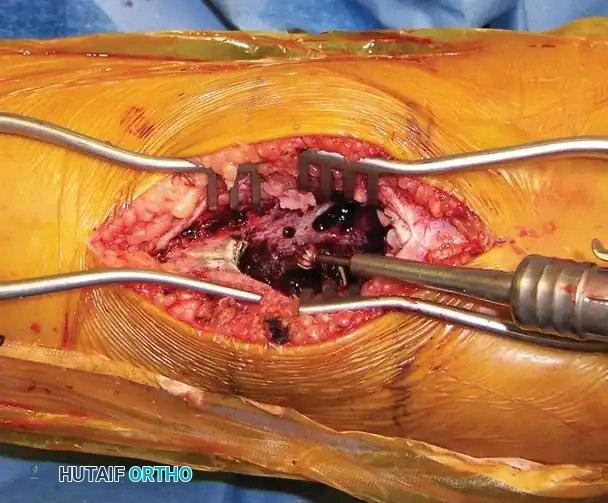
Following the removal of the gross cyst lining with large curettes, the tumor cavity is systematically enlarged and cleared using a high-speed power burr.
- Adjuvant Therapy: To further eradicate microscopic disease, a chemical or thermal adjuvant is applied. Argon beam coagulation is highly effective, providing uniform thermal necrosis to the cavity walls while minimizing deep tissue damage. Alternative adjuvants include liquid nitrogen (cryotherapy) or phenol, though these carry higher risks of adjacent nerve/tissue damage.
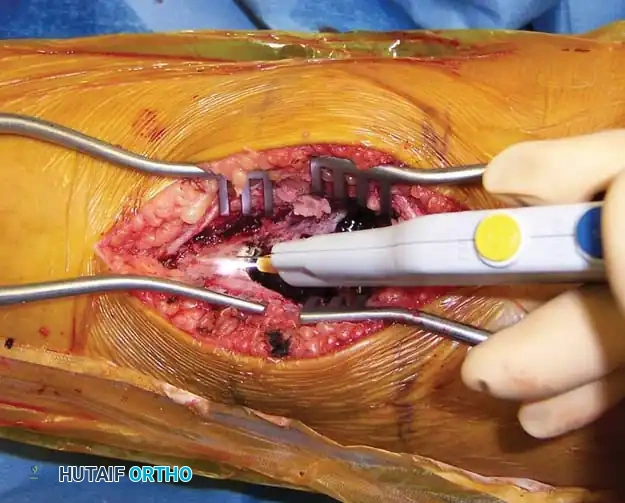
The tumor cavity is meticulously treated with argon beam coagulation to achieve thermal necrosis of any remaining microscopic cyst lining.
- Reconstruction and Grafting: The sterilized cavity is thoroughly irrigated and packed tightly with bone graft. A combination of freeze-dried cancellous allograft chips and demineralized bone matrix (DBM) provides an excellent osteoconductive and osteoinductive scaffold. If the cortical defect is massive or the bone is mechanically compromised, prophylactic internal fixation with plates and screws is applied.
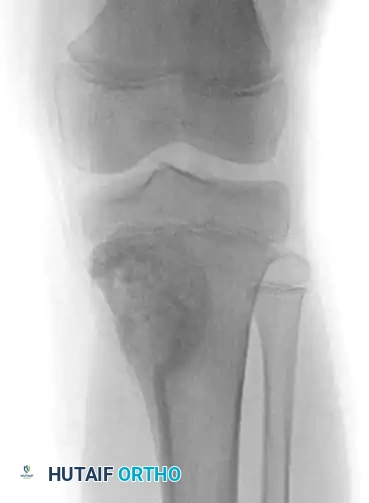
Immediate postoperative anteroposterior radiograph demonstrating the cavity densely packed with bone graft substitute.
Postoperative Protocol and Prognosis
Patients are typically kept non-weight-bearing or touch-down weight-bearing until radiographic evidence of graft incorporation and cortical consolidation is observed (usually 8 to 12 weeks).
The recurrence rate following extended curettage with adjuvant therapy is approximately 10% to 20%. Recurrence is strongly correlated with patient age younger than 15 years, centrally located cysts, and, most importantly, incomplete surgical removal of the cystic contents. Routine radiographic surveillance is required for at least 2 to 3 years postoperatively.
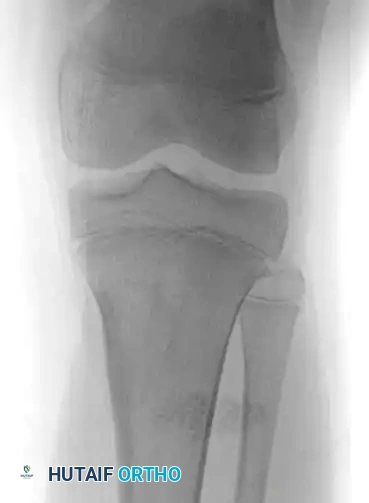
Radiograph obtained 1 year postoperatively showing complete osseous remodeling, graft incorporation, and no evidence of local recurrence.
Alternative Therapies: In surgically inaccessible locations (e.g., complex sacral or spinal lesions), serial arterial embolization can be used as a definitive, standalone treatment. Low-dose irradiation has historically been effective in inducing rapid ossification; however, it is strictly contraindicated in modern practice due to the unacceptable risk of secondary radiation-induced sarcomas in young patients.
INTRAOSSEOUS GANGLION CYST
Intraosseous ganglion cysts are benign, mucoid-filled lesions that typically occur in the epiphyseal or metaphyseal ends of long bones. They are most frequently observed in middle-aged men. The distal tibia (medial malleolus) is the most common site, followed by the periarticular regions of the knee and shoulder.
Pathogenesis and Imaging
These cysts are widely considered to be intraosseous extensions of soft-tissue ganglia that penetrate the bone cortex, or the result of mucoid degeneration of intraosseous connective tissue. Subperiosteal variants have also been documented.
Radiographically and on MRI, intraosseous ganglia appear as uniloculated or multiloculated, well-demarcated lytic defects located adjacent to a joint surface. They are characteristically surrounded by a distinct, thin rim of sclerotic reactive bone. Unlike subchondral cysts associated with osteoarthritis, intraosseous ganglia typically occur in joints with preserved articular cartilage space.
Surgical Management
Asymptomatic lesions discovered incidentally require only observation. For symptomatic lesions causing localized pain or threatening the structural integrity of the articular surface, surgical intervention is indicated. Treatment consists of local excision of any communicating overlying soft-tissue ganglion, followed by meticulous intralesional curettage of the involved bone. The resulting defect is grafted with cancellous bone. Recurrence following thorough curettage is exceedingly uncommon.
EPIDERMOID CYST OF BONE
Epidermoid cysts of bone are rare, benign lesions filled with keratinous material and lined by a flattened, stratified squamous epithelium. Microscopically, they are indistinguishable from common epidermal inclusion cysts found in the skin.
Pathogenesis and Clinical Presentation
In the osseous skeleton, epidermoid cysts are most frequently encountered in the skull (often developmental) and the distal phalanges of the fingers. When occurring in the phalanges, they are almost universally considered traumatic in origin. The prevailing theory suggests that a penetrating injury (such as a crush injury or a deep puncture wound) drives fragments of the germinal layer of the epidermis deep into the underlying cancellous bone. Over years, these epithelial cells proliferate, secrete keratin, and form an expanding intraosseous cyst.
Radiographic Evaluation and Treatment
Radiographically, epidermoid cysts present as well-circumscribed, rarefied lytic defects within the distal phalanx, often causing expansion of the bone and surrounded by a dense sclerotic margin. They can occasionally cause thinning of the cortex to the point of pathological fracture.
Treatment involves surgical exploration, meticulous curettage of the entire epithelial lining to prevent recurrence, and bone grafting of the defect if structurally necessary. Because the lesion is benign, marginal excision is curative, and the prognosis for full functional recovery of the digit is excellent.
