CONGENITAL DISLOCATION OF THE RADIAL HEAD
Congenital dislocation of the radial head is an exceedingly rare orthopedic anomaly. However, it must be maintained high on the differential diagnosis when a patient presents with a chronically dislocated radial head in the absence of any historical or radiographic evidence of an ulnar fracture (such as a missed Monteggia fracture-dislocation). The condition is characterized by profound dysplastic changes in the osseous morphology of the proximal radioulnar joint (PRUJ) and the radiocapitellar articulation.
Pathoanatomy and Clinical Evaluation
The etiology of congenital radial head dislocation is believed to stem from a failure of normal embryological development of the radiocapitellar joint. It may present as an isolated unilateral anomaly, though older literature frequently cited bilaterality as a diagnostic criterion. Contemporary evidence confirms that unilateral presentations are well-documented. Furthermore, there is a recognized familial predisposition, particularly with paternal inheritance patterns, and it may present syndromically in association with chondro-osteodystrophy or other connective tissue disorders.
Clinically, patients often present with a visible or palpable prominence at the elbow and a restricted range of motion, most notably a limitation in forearm supination and pronation. Flexion and extension are typically preserved until secondary osteoarthritic changes develop later in life.
Radiographic Diagnostic Criteria
The radiographic findings of a congenital dislocation are pathognomonic and serve to differentiate it from traumatic dislocations.
Clinical Pearl: Differentiating Congenital vs. Traumatic Dislocation
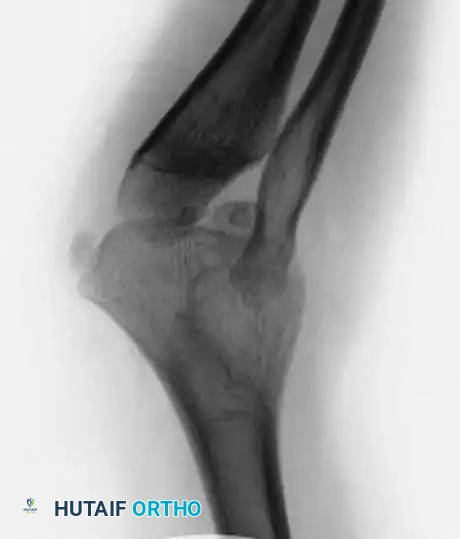
In a traumatic dislocation, the radial head is typically normal in shape, and the ulna may show signs of a healed fracture or plastic deformation. In congenital cases, the radial head is dysplastic (dome-shaped), the capitellum is hypoplastic, and the ulna exhibits adaptive bowing.
Key radiographic features include:
* Radial Shaft Hypertrophy: The radial shaft is abnormally long relative to the ulna.
* Ulnar Bowing: The ulna is usually abnormally bowed to accommodate the over-lengthened radius.
* Direction of Dislocation: The radial head is most frequently dislocated posteriorly, though anterior and lateral dislocations can occur.
* Dysplastic Radial Head: The head is rounded and dome-shaped, showing little to no central depression (fovea) for articulation with the capitellum. It is usually significantly smaller than normal.
* Capitellar Hypoplasia: The capitellum is often small and flattened due to the absence of normal articulating forces during skeletal development.
* Absent Radial Notch: The radial notch of the proximal ulna is typically small, dysplastic, or entirely absent.
* Ectopic Ossification: Occasionally, areas of heterotopic ossification are present in the soft tissues surrounding the radial head.
Management Principles
A congenitally dislocated radial head is fundamentally irreducible—both manually and surgically. This irreducibility is dictated by severe adaptive contractures of the surrounding soft tissues and the complete absence of normal articular congruity between the radius, ulna, and humerus.
Surgical Warning: Avoid Open Reduction in Children
Attempting open reduction and reconstruction of the annular ligament in a child with a congenital dislocation is strictly contraindicated. The dysplastic joint surfaces will not maintain reduction, and surgical intervention will inevitably lead to severe stiffness, pain, and accelerated degenerative changes.
Pediatric Management:
In children, any impairment of function is usually limited to a restriction of forearm rotation. The only indicated treatment during childhood is targeted physical therapy to maximize and maintain the existing range of motion.
Adolescent and Adult Management:
If significant pain or severe mechanical block persists into late adolescence or adulthood, surgical intervention may be warranted. The procedure of choice is radial head excision.
Historically, radial head resection was delayed until complete skeletal maturity to prevent proximal migration of the radius and subsequent ulnocarpal impaction. However, even after skeletal maturity, excision may not fully restore motion due to long-standing soft tissue contractures. Notably, Campbell, Waters, and Emans reported highly favorable results following radial head excision in a cohort of six patients (eight elbows) ranging in age from 10 to 15.5 years. Contrary to older dogmatic data, their study demonstrated that excision of the radial head in this specific age group resulted in both an increased range of motion and a significant decrease in elbow pain across all treated elbows.
Surgical Technique: Radial Head Excision
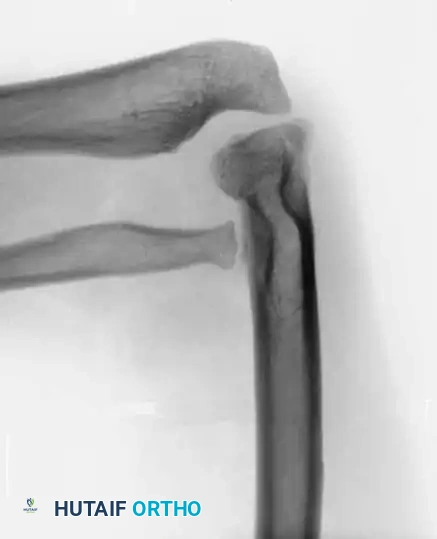
Positioning and Preparation:
* The patient is placed in the supine position with the operative arm extended on a radiolucent hand table.
* A well-padded non-sterile tourniquet is applied high on the brachium.
* The limb is prepped and draped in standard sterile fashion.
Surgical Approach (Kocher Interval):
* An oblique incision is made over the posterolateral aspect of the elbow, extending from the lateral epicondyle distally toward the ulnar crest.
* The internervous plane between the anconeus (radial nerve) and the extensor carpi ulnaris (posterior interosseous nerve) is identified and developed.
* Critical Step: The forearm is fully pronated to move the posterior interosseous nerve (PIN) anteriorly and away from the surgical field, minimizing the risk of iatrogenic injury.
Resection:
* The joint capsule is incised longitudinally to expose the dysplastic radial head.
* The radial neck is exposed subperiosteally. Care is taken not to dissect distal to the annular ligament to further protect the PIN.
* An oscillating saw is used to resect the radial head at the level of the radial neck, just proximal to the bicipital tuberosity.
* The resected bone edge is smoothed with a rasp or rongeur to prevent soft tissue irritation.
* The capsule and muscular intervals are closed in layers over a suction drain, followed by routine skin closure.
Postoperative Protocol:
* The elbow is placed in a soft bulky dressing or a posterior splint in 90 degrees of flexion for 3 to 5 days to allow for initial soft tissue healing.
* Active and active-assisted range of motion exercises (flexion, extension, pronation, supination) are initiated early to prevent capsular contracture.
* Strengthening exercises are delayed until 6 weeks postoperatively.
CONGENITAL PSEUDARTHROSIS OF THE RADIUS
Congenital pseudarthrosis of the radius is an extremely rare pathological entity, significantly less common than its tibial counterpart. It is almost exclusively associated with neurofibromatosis type 1 (NF1).
Etiology and Pathoanatomy
In patients with neurofibromatosis, the pseudarthrosis typically originates from a dysplastic cyst within the radius. These patients invariably present with classic cutaneous manifestations of NF1 (café-au-lait spots, axillary freckling, neurofibromas) or possess a strong familial history of the disease.
The pathoanatomy is highly consistent across reported cases:
* The pseudarthrosis invariably occurs in the distal third of the radial diaphysis.
* The distal osseous fragment is characteristically very short and osteoporotic.
* Because the lesion is situated in close proximity to the distal radial physis, the bone ends become severely attenuated and "pencil-like."
* The ulna continues to grow normally, resulting in relative ulnar overgrowth, bowing, and secondary subluxation or dislocation of the distal radioulnar joint (DRUJ) or radiocapitellar joint.
Surgical Management Strategies
The primary goal of surgery is to achieve stable osseous union, restore radial length, and realign the radiocarpal and radioulnar articulations. Achieving union in dysplastic, neurofibromatous bone is notoriously difficult.
1. Dual-Onlay Bone Grafting (Boyd Technique)
For decades, the treatment of choice has been the dual-onlay bone grafting technique, originally popularized by Boyd for congenital pseudarthrosis of the tibia.
Biomechanics of Dual-Onlay Grafting:
This technique provides a "viselike" mechanical grip on the short, osteoporotic distal fragment. It effectively increases the cross-sectional diameter of the attenuated bone ends, restores radial length, and provides massive osteogenic potential, resulting in satisfactory union rates.
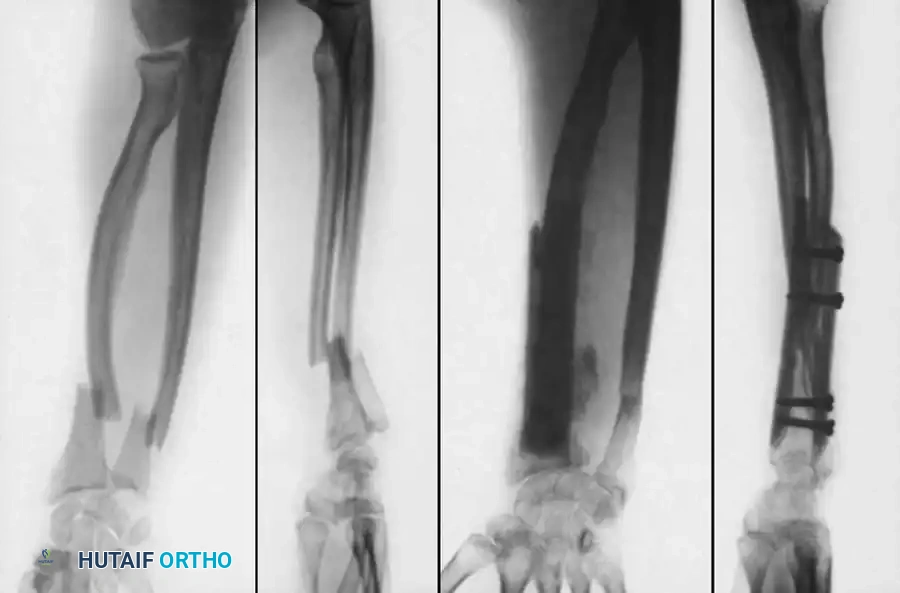
Surgical Steps:
* Approach: A volar Henry approach is utilized to expose the distal radius, carefully protecting the radial artery and the superficial branch of the radial nerve.
* Preparation: The pseudarthrosis site is radically debrided. All fibrous hamartomatous tissue and thickened periosteum must be excised until healthy, bleeding cortical bone is encountered. The medullary canals of both fragments are opened with a drill or curette.
* Graft Harvest: Corticocancellous bone grafts are harvested from the ipsilateral or contralateral iliac crest.
* Fixation: The grafts are contoured to fit the volar and dorsal aspects of the radius. They are applied as dual onlay plates bridging the defect.
* Fixation is achieved using cortical screws that pass through the volar graft, the host bone, and the dorsal graft, compressing the construct.
* Cancellous bone chips are packed into any remaining voids at the nonunion site.
2. Free Vascularized Fibular Grafting (FVFG)
Recent advancements in microsurgery have shifted the paradigm toward free vascularized fibular transfer, particularly for larger defects or recalcitrant nonunions. Kameyama and Ogawa reported excellent outcomes following complete en bloc resection of the involved radius (including surrounding dysplastic periosteum and soft tissue) followed by FVFG.
Timing and Fixation Controversies:
* Delayed vs. Early Intervention: Kameyama and Ogawa advocated delaying the procedure until skeletal maturity, utilizing a custom forearm brace in the interim. Conversely, Allieu et al. and Cheng et al. strongly recommended early FVFG intervention (e.g., at 20 months to 5 years of age) to prevent secondary radial head dislocation and irreversible ulnar deformity.
* Outcomes: In a comprehensive review, Witoonchart et al. demonstrated that FVFG yielded the highest union rate among all reported procedures, achieving success in 18 of 19 reported ulnar or radial pseudarthroses.
* Fixation Challenges in the Pediatric Patient: Witoonchart et al. highlighted the profound difficulty of obtaining stable internal fixation in very young children. Rigid plate and screw fixation carries a high risk of iatrogenic damage to the delicate vascular pedicle supplying the fibular graft's periosteum. However, relying solely on unstable fixation (such as intramedullary or crossed Kirschner wires) was implicated as the primary cause of delayed union in their cohort. A delicate balance must be struck, often utilizing customized mini-fragment plates or specialized external fixators that spare the vascular anastomosis.
CONGENITAL PSEUDARTHROSIS OF THE CLAVICLE
Congenital pseudarthrosis of the clavicle is a rare anomaly that almost exclusively affects the right clavicle. It is distinct from cleidocranial dysplasia and obstetric clavicle fractures. The defect typically occurs in the middle third of the clavicle and is thought to arise from a failure of fusion between the medial and lateral primary ossification centers.
Clinical Presentation and Indications for Surgery
Patients typically present with a painless, palpable mass over the mid-clavicle. As the child grows, the shoulder girdle may droop anteriorly and inferiorly, leading to cosmetic deformity and, occasionally, late-onset thoracic outlet syndrome or shoulder girdle fatigue.
Surgical intervention is indicated for:
1. Progressive cosmetic deformity.
2. Pain or functional impairment of the shoulder girdle.
3. Neurological or vascular compromise (rare).
Surgery is optimally performed between 3 and 7 years of age, allowing the clavicle to grow sufficiently to accept internal fixation while utilizing the child's massive osteogenic potential.
Surgical Technique: Open Reduction and Iliac Bone Grafting
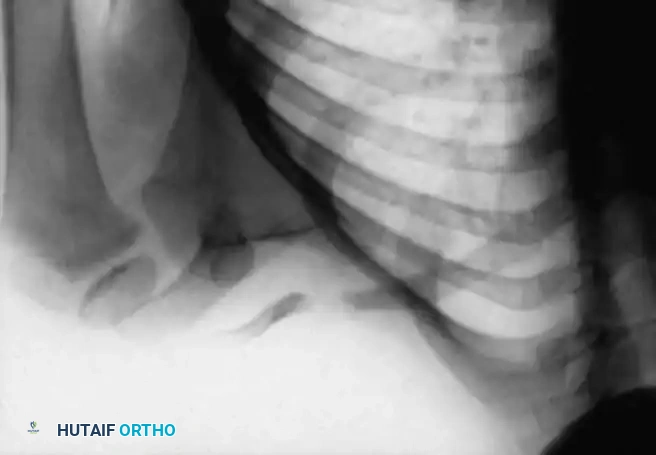
Positioning:
* The patient is placed in the beach-chair or supine position with a bump between the scapulae to allow the shoulders to fall posteriorly, aiding in the reduction of the clavicle.
* The entire forequarter and ipsilateral iliac crest are prepped and draped.
Surgical Approach:
* An incision is made directly over the clavicle, parallel to Langer's lines, centered over the pseudarthrosis.
* The platysma is incised, and the supraclavicular nerves are identified and protected.
* The periosteum is incised longitudinally.
Surgical Warning: Subclavian Vessel Protection
Dissection around the pseudarthrosis must remain strictly subperiosteal. The subclavian vein and artery, as well as the brachial plexus, lie immediately posteroinferior to the middle third of the clavicle. A blunt retractor (such as a Darrach or malleable retractor) must be placed deep to the bone during drilling and resection.
Preparation of the Pseudarthrosis:
* Thoroughly debride the site of the pseudarthrosis. All intervening fibrous and cartilaginous tissue must be excised down to normal, bleeding cortical bone medially and laterally.
* The sclerotic bone ends are resected, and the medullary canals are opened with a small drill bit to promote osteogenesis.
Internal Fixation and Grafting:
* Select a four-hole or six-hole plate (semitubular, dynamic compression, or acetabular reconstruction plate, depending on the child's size).
* Carefully bend and contour the plate to fit the superior or anterosuperior surface of the clavicle.
* Fix the plate to the clavicle using standard AO techniques, ensuring at least two (preferably three) bicortical screws are placed in both the medial and lateral fragments.
* Obtain autogenous cancellous and cortical grafts from the iliac crest.
* Place the bone grafts meticulously on the superior, inferior, and posterior aspects of the pseudarthrosis site, bridging the defect completely.

Closure:
* The periosteum and clavipectoral fascia are closed over the plate if possible.
* The platysma is closed as a distinct layer to prevent widened scarring.
* The skin is closed with a running subcuticular suture.
Postoperative Protocol
- Immobilization: A collar and cuff sling or a pediatric shoulder immobilizer is worn continuously for 2 to 3 weeks to protect the soft tissues and the fixation construct.
- Rehabilitation: Gentle pendulum exercises are initiated at 3 weeks. Active elevation above 90 degrees and lifting are restricted until radiographic evidence of bridging callus is observed (typically 6 to 8 weeks).
- Hardware Removal: Due to the subcutaneous nature of the clavicle, the plate often becomes prominent and symptomatic as the child grows. The plate can be safely removed at 12 to 24 months postoperatively, provided that solid radiographic union and cortical remodeling have occurred.
