Tapentadol ER (Nucynta ER): A Comprehensive Medical SEO Guide for Chronic Pain Management
As an expert in orthopedic pain management, understanding the nuances of advanced analgesic options is paramount. Tapentadol ER, marketed as Nucynta ER, represents a significant advancement in the treatment of severe chronic pain. This comprehensive guide delves into its intricate mechanisms, clinical applications, safety profile, and management strategies, providing an authoritative resource for both healthcare professionals and patients seeking to understand this powerful medication.
1. Introduction & Overview of Tapentadol ER (Nucynta ER)
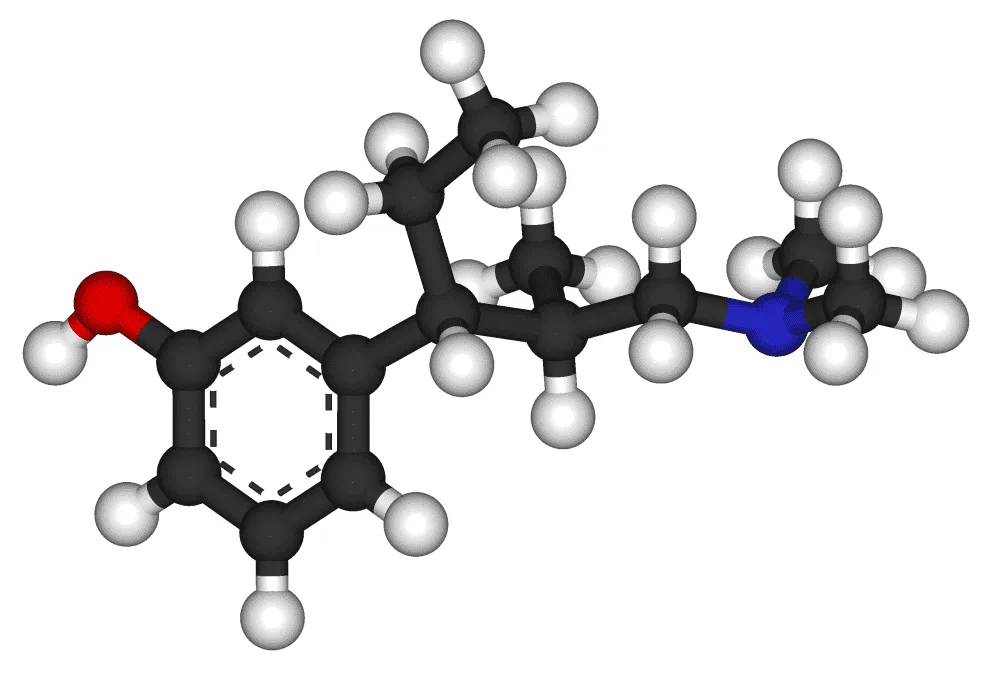
Tapentadol Extended-Release (ER), known by its brand name Nucynta ER, is a centrally acting opioid analgesic indicated for the management of severe chronic pain in adults requiring continuous, around-the-clock opioid analgesia for an extended period. What sets Tapentadol ER apart from traditional opioids is its unique dual mechanism of action, which combines mu-opioid receptor agonism with norepinephrine reuptake inhibition. This synergistic approach offers a broad spectrum of pain relief, including components of neuropathic pain, making it a valuable tool in complex pain syndromes often encountered in orthopedics.
Developed to provide effective and sustained pain control, Tapentadol ER is formulated for once or twice-daily dosing, ensuring consistent therapeutic levels and reducing fluctuations in pain intensity. Its introduction has provided clinicians with an alternative to traditional strong opioids, potentially offering a different side effect profile and efficacy for specific patient populations.
2. Deep-Dive into Technical Specifications & Mechanisms
2.1. Mechanism of Action: The Dual-Action Advantage
Tapentadol's unique analgesic profile stems from its two distinct pharmacological actions:
-
Mu-Opioid Receptor (MOR) Agonism: Tapentadol acts as a full agonist at the mu-opioid receptors in the central nervous system (CNS). This binding mimics the action of endogenous opioids, leading to:
- Inhibition of Pain Signal Transmission: It reduces the release of neurotransmitters involved in pain signaling from primary afferent neurons in the dorsal horn of the spinal cord.
- Modulation of Pain Perception: It alters the brain's perception of pain, increasing pain tolerance and reducing the emotional distress associated with chronic pain.
- Activation of Descending Inhibitory Pathways: It enhances the activity of pain-modulating pathways that descend from the brainstem to the spinal cord.
This MOR agonism is a primary contributor to its potent analgesic effects, similar to other opioids.
-
Norepinephrine Reuptake Inhibition (NRI): Tapentadol also inhibits the reuptake of norepinephrine (NE) in the CNS. This action contributes significantly to its analgesic properties, particularly in neuropathic pain. By increasing the concentration of norepinephrine in the synaptic cleft, Tapentadol:
- Enhances Descending Noradrenergic Pathways: Norepinephrine is a key neurotransmitter in the descending pain inhibitory pathways. By increasing its availability, Tapentadol strengthens these natural pain-suppressing systems.
- Modulates Neuropathic Pain: The noradrenergic system plays a crucial role in the processing of neuropathic pain. Inhibition of NE reuptake can reduce hypersensitivity and allodynia often associated with nerve damage.
The combination of these two mechanisms provides a synergistic effect, offering broader pain relief compared to agents that act solely via opioid receptors. This dual action is particularly beneficial for mixed pain states, where both nociceptive and neuropathic components are present, common in orthopedic conditions like chronic low back pain with radiculopathy or post-surgical neuropathic pain.
2.2. Pharmacokinetics
Understanding the pharmacokinetics of Tapentadol ER is crucial for appropriate dosing and management.
-
Absorption:
- After oral administration, Tapentadol is rapidly and extensively absorbed from the gastrointestinal tract.
- The extended-release formulation provides sustained plasma concentrations, typically reaching peak plasma concentration (Tmax) between 3 to 6 hours.
- Absolute bioavailability is approximately 32% due to significant first-pass metabolism.
- Food intake does not significantly affect the absorption or bioavailability of Tapentadol ER.
-
Distribution:
- Tapentadol has a large volume of distribution, indicating extensive tissue distribution.
- Plasma protein binding is low, approximately 20%, suggesting that drug interactions based on protein binding displacement are unlikely.
-
Metabolism:
- Tapentadol undergoes extensive metabolism, primarily via conjugation with glucuronic acid (Phase II metabolism).
- The major metabolic pathways involve UGT1A6, UGT1A9, and UGT2B7 enzymes, leading to the formation of inactive metabolites (e.g., tapentadol-O-glucuronide).
- Unlike many other opioids, metabolism via cytochrome P450 (CYP) isoenzymes is minor, primarily CYP2C9 and CYP2C19, forming N-desmethyltapentadol. This limited CYP involvement reduces the potential for drug interactions with CYP inhibitors or inducers, a significant advantage.
- No active metabolites contribute to its analgesic effect.
-
Elimination:
- Tapentadol and its metabolites are primarily excreted via the kidneys.
- Approximately 99% of the administered dose is excreted in urine, with about 70% as glucuronides.
- The terminal half-life of Tapentadol ER is approximately 4-6 hours, but due to the extended-release formulation, its duration of action allows for once or twice-daily dosing.
-
Special Populations:
- Hepatic Impairment: Patients with moderate hepatic impairment show significantly higher plasma concentrations (Cmax and AUC) of Tapentadol. Severe hepatic impairment is not recommended. Dose adjustments are necessary.
- Renal Impairment: Patients with severe renal impairment also exhibit increased plasma levels. Dose adjustments are required.
- Elderly: No specific dose adjustment based solely on age is generally needed, but caution is advised due to potential for decreased renal/hepatic function and increased sensitivity to opioids.
3. Extensive Clinical Indications & Usage
Tapentadol ER is a crucial medication for specific chronic pain conditions, especially those with a neuropathic component.
3.1. Detailed Indications
- Management of Severe Chronic Pain: Tapentadol ER is indicated for the management of severe chronic pain in adults who require continuous, around-the-clock opioid analgesia for an extended period. This includes various chronic musculoskeletal conditions, such as:
- Chronic low back pain
- Osteoarthritis pain (when other treatments are insufficient)
- Fibromyalgia (off-label, but may be used for chronic pain components)
- Post-surgical chronic pain states (e.g., after spinal fusion, joint replacement, if pain persists and warrants long-term opioid use).
- Management of Neuropathic Pain associated with Diabetic Peripheral Neuropathy (DPN): Tapentadol ER is specifically indicated for the management of neuropathic pain associated with diabetic peripheral neuropathy (DPN) in adults severe enough to require daily, continuous, long-term opioid treatment. This indication highlights its unique dual mechanism, which is particularly effective against neuropathic pain symptoms like burning, tingling, and shooting pain.
Important Note: As an extended-release formulation, Tapentadol ER is NOT indicated for as-needed (PRN) pain relief or for acute pain management. Its role is strictly for chronic pain requiring continuous opioid therapy.
3.2. Dosage Guidelines
Precise dosing and titration are essential to maximize efficacy and minimize risks.
- Initial Dosing for Opioid-Naïve Patients:
- The recommended starting dose for opioid-naïve patients is 50 mg orally twice daily (every 12 hours).
- Initial Dosing for Patients Currently Receiving Other Opioids:
- Patients transitioning from other opioids require careful conversion. It is generally recommended to start with the lowest dose (50 mg twice daily) and titrate slowly, monitoring for signs of withdrawal or over-sedation. A conservative approach is always best.
- Titration and Maintenance:
- The dose of Tapentadol ER should be individualized based on the patient's response, tolerability, and the balance between pain relief and side effects.
- Doses can be increased by 50 mg increments, no more frequently than every 3 days.
- The maximum recommended dose is 250 mg twice daily (500 mg/day). Doses above this have not been studied for safety and efficacy.
- Dosing in Special Populations:
- Moderate Hepatic Impairment: Reduce the starting dose to 50 mg once daily and titrate cautiously. Do not use in severe hepatic impairment.
- Severe Renal Impairment (CrCl < 30 mL/min): Reduce the starting dose to 50 mg once daily and titrate cautiously.
- Elderly Patients (≥65 years): While no specific dose adjustment is required, caution is advised due to increased sensitivity and potential for age-related decline in organ function.
- Administration Instructions:
- Tapentadol ER tablets must be swallowed whole. They should not be crushed, chewed, dissolved, or split. Doing so can lead to rapid release and absorption of a potentially fatal dose of tapentadol.
- Can be taken with or without food.
- Discontinuation:
- Abrupt discontinuation of Tapentadol ER in physically dependent patients can lead to severe opioid withdrawal symptoms.
- A gradual tapering schedule is essential to minimize withdrawal symptoms. Reduce the dose by 25% to 50% every 2 to 4 days, or as clinically appropriate, under medical supervision.
4. Risks, Side Effects, and Contraindications
Like all potent analgesics, Tapentadol ER carries significant risks that must be carefully considered.
4.1. Contraindications
Tapentadol ER is contraindicated in patients with:
* Significant respiratory depression: Acute or severe bronchial asthma or hypercarbia in an unmonitored setting or in the absence of resuscitative equipment.
* Known or suspected paralytic ileus: Opioids can exacerbate this condition.
* Concomitant use of monoamine oxidase inhibitors (MAOIs) or within 14 days of discontinuing MAOIs: Risk of hypertensive crisis and serotonin syndrome.
* Hypersensitivity: To tapentadol or any component of the formulation.
* Acute pain management: Due to its extended-release nature, it is unsuitable for acute pain.
4.2. Black Box Warnings (As per FDA)
- Addiction, Abuse, and Misuse: Tapentadol ER exposes patients and other users to the risks of opioid addiction, abuse, and misuse, which can lead to overdose and death.
- Respiratory Depression: Serious, life-threatening, or fatal respiratory depression may occur. Patients must be monitored, especially during initiation and dose titration.
- Accidental Ingestion: Accidental ingestion of even one dose, especially by children, can result in a fatal overdose.
- Neonatal Opioid Withdrawal Syndrome (NOWS): Prolonged use during pregnancy can result in NOWS, which may be life-threatening if not recognized and treated.
- Interactions with Alcohol: Co-ingestion with alcohol can result in the rapid release of tapentadol, leading to potentially fatal overdose.
- Concomitant Use with Benzodiazepines and Other CNS Depressants: Can result in profound sedation, respiratory depression, coma, and death.
4.3. Common Side Effects
Many side effects are typical of opioid medications and often dose-dependent:
* Nausea, vomiting
* Constipation
* Dizziness, somnolence (drowsiness)
* Headache
* Dry mouth
* Fatigue
* Pruritus (itching)
4.4. Serious Side Effects
- Respiratory Depression: The most serious risk.
- Serotonin Syndrome: Due to its norepinephrine reuptake inhibition, concurrent use with other serotonergic drugs can precipitate serotonin syndrome (agitation, hallucinations, tachycardia, fever, sweating, muscle rigidity, severe nausea/vomiting/diarrhea).
- Seizures: Tapentadol may lower the seizure threshold in some patients.
- CNS Depression: Profound sedation, confusion, impaired cognitive and motor function.
- Adrenal Insufficiency: Chronic opioid use can lead to adrenal insufficiency (fatigue, weakness, dizziness, nausea, vomiting, loss of appetite).
- Androgen Deficiency: Chronic opioid use may result in hypogonadism or androgen deficiency (decreased libido, impotence, amenorrhea, infertility).
- Hypotension: May cause severe hypotension, particularly in patients with compromised cardiovascular function.
4.5. Drug Interactions
- CNS Depressants (e.g., benzodiazepines, other opioids, sedatives, hypnotics, tranquilizers, muscle relaxants, general anesthetics, alcohol): Greatly increased risk of respiratory depression, profound sedation, coma, and death. Concomitant prescribing should be avoided or used with extreme caution and reduced dosages of one or both agents.
- Serotonergic Drugs (e.g., SSRIs, SNRIs, TCAs, triptans, MAOIs, linezolid, tramadol, St. John's Wort): Increased risk of serotonin syndrome. Monitor for symptoms.
- Monoamine Oxidase Inhibitors (MAOIs): Contraindicated. Severe, potentially fatal reactions (e.g., hypertensive crisis, serotonin syndrome) can occur.
- Drugs affecting UGT Enzymes: While Tapentadol is primarily metabolized by glucuronidation, concomitant use of strong UGT inhibitors or inducers could theoretically alter tapentadol levels, though significant clinical interactions are less common compared to CYP-mediated interactions.
4.6. Pregnancy and Lactation Warnings
- Pregnancy Category C: There are no adequate and well-controlled studies of Tapentadol in pregnant women. Animal studies show adverse developmental effects. Tapentadol should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus.
- Neonatal Opioid Withdrawal Syndrome (NOWS): Prolonged use of opioids during pregnancy can result in NOWS, which presents with irritability, hyperactivity, abnormal sleep pattern, high-pitched cry, tremor, vomiting, diarrhea, and failure to gain weight.
- Labor and Delivery: Opioids can cross the placental barrier and cause respiratory depression and other signs of opioid toxicity in the newborn. Not recommended for use in pregnant women during or immediately prior to labor when other analgesic techniques are more appropriate.
- Lactation: Tapentadol is excreted in human milk. Given the potential for serious adverse reactions in breastfed infants (e.g., sedation, respiratory depression), a decision should be made whether to discontinue nursing or discontinue the drug, taking into account the importance of the drug to the mother.
4.7. Overdose Management
An overdose of Tapentadol ER can be life-threatening and requires immediate medical attention.
- Symptoms of Overdose:
- Respiratory depression (decreased respiratory rate and/or tidal volume, Cheyne-Stokes respiration, cyanosis).
- Somnolence progressing to stupor or coma.
- Miosis (pinpoint pupils), though mydriasis may occur in severe hypoxia.
- Skeletal muscle flaccidity.
- Cold and clammy skin.
- Bradycardia, hypotension.
- Circulatory collapse, cardiac arrest, and death.
- Treatment of Overdose:
- Establish a patent airway and assist ventilation: The primary concern is to re-establish adequate respiratory exchange.
- Administer an opioid antagonist (Naloxone): Naloxone is a specific antidote for opioid overdose. Administer intravenously, intramuscularly, or subcutaneously. Repeated doses may be necessary, especially with an extended-release formulation like Tapentadol ER, as the opioid effects may outlast the duration of action of the antagonist.
- Supportive Care: Maintain blood pressure and circulation with intravenous fluids and vasopressors as indicated. Treat hypothermia.
- Monitoring: Patients should be monitored for a prolonged period (at least 24 hours) due to the extended-release nature of Tapentadol ER and the potential for recurrence of respiratory depression.
5. Massive FAQ Section
Q1: What is Tapentadol ER (Nucynta ER) and how is it different from other opioids?
A1: Tapentadol ER (Nucynta ER) is a prescription opioid analgesic used for severe chronic pain. Its key difference lies in its dual mechanism of action: it acts as a mu-opioid receptor agonist (like traditional opioids) AND a norepinephrine reuptake inhibitor. This dual action provides broader pain relief, including for neuropathic pain, and may have a different side effect profile compared to single-mechanism opioids.
Q2: Is Nucynta ER an opioid?
A2: Yes, Nucynta ER (Tapentadol ER) is an opioid. It contains tapentadol, which is classified as a Schedule II controlled substance due to its potential for abuse and dependence.
Q3: Can I take Nucynta ER for acute pain or "as-needed" pain?
A3: No, Nucynta ER is an extended-release formulation specifically designed for continuous, around-the-clock management of severe chronic pain. It is NOT intended for as-needed (PRN) pain relief or for the treatment of acute pain. Using it for acute pain can lead to inappropriate dosing and increased risks.
Q4: How long does it take for Nucynta ER to start working?
A4: As an extended-release medication, Nucynta ER is designed to provide sustained pain relief over an extended period. While some relief may be felt within a few hours of the first dose, it typically takes several days of consistent dosing for the medication to reach steady-state concentrations in your body and for you to experience its full therapeutic effect.
Q5: What should I do if I miss a dose of Nucynta ER?
A5: If you miss a dose, take it as soon as you remember, unless it is almost time for your next scheduled dose. In that case, skip the missed dose and resume your regular dosing schedule. Do NOT take two doses at once or take extra medication to make up for a missed dose, as this can lead to an overdose.
Q6: Can I drink alcohol while taking Nucynta ER?
A6: Absolutely not. Drinking alcohol while taking Nucynta ER is extremely dangerous and can lead to a rapid release of tapentadol from the extended-release formulation. This can cause severe respiratory depression, profound sedation, coma, and even death. Avoid alcohol completely while on this medication.
Q7: What are the signs of Nucynta ER addiction or dependence?
A7: Signs of addiction or dependence can include: craving the drug, taking more than prescribed, continuing use despite negative consequences, neglecting responsibilities, withdrawal symptoms when stopping, and spending excessive time obtaining or using the drug. If you or someone you know exhibits these signs, seek medical help immediately.
Q8: How do I stop taking Nucynta ER safely?
A8: Do NOT stop taking Nucynta ER abruptly. Abrupt discontinuation can lead to severe opioid withdrawal symptoms. Your doctor will create a gradual tapering schedule to slowly reduce your dose over time, minimizing withdrawal and ensuring your safety. Always consult your healthcare provider before making any changes to your medication regimen.
Q9: Is Nucynta ER safe during pregnancy or while breastfeeding?
A9: Nucynta ER is generally not recommended during pregnancy due to the risk of neonatal opioid withdrawal syndrome in the newborn. It can also cause respiratory depression in the baby if used during labor. Tapentadol is excreted in breast milk and can cause serious adverse effects in breastfed infants. Discuss the risks and benefits thoroughly with your doctor if you are pregnant, planning to become pregnant, or breastfeeding.
Q10: What should I do if I experience severe side effects like extreme dizziness or difficulty breathing?
A10: If you experience severe side effects such as extreme dizziness, fainting, severe drowsiness, confusion, significant difficulty breathing, shallow breathing, blue lips, or unresponsiveness, seek emergency medical attention immediately. These could be signs of serious complications like respiratory depression or overdose.
Q11: Can Nucynta ER cause serotonin syndrome?
A11: Yes, Nucynta ER (Tapentadol ER) can increase the risk of serotonin syndrome, especially when taken with other serotonergic drugs (e.g., SSRIs, SNRIs, triptans, MAOIs). Symptoms of serotonin syndrome include agitation, hallucinations, rapid heart rate, fever, sweating, muscle stiffness or twitching, loss of coordination, nausea, vomiting, or diarrhea. If you experience these symptoms, seek immediate medical attention.
Q12: How does Nucynta ER affect driving or operating machinery?
A12: Nucynta ER can cause dizziness, drowsiness, and impaired mental and physical abilities. You should avoid driving or operating heavy machinery until you know how the medication affects you and are certain you can perform these tasks safely.
This comprehensive guide aims to provide a robust understanding of Tapentadol ER (Nucynta ER) for effective and safe chronic pain management. Always consult with a qualified healthcare professional for personalized medical advice and treatment plans.