Cefazolin 500 mg: An In-Depth Medical SEO Guide
As an expert in orthopedic medicine and a seasoned medical SEO copywriter, I understand the critical role that specific medications play in patient care and recovery. Cefazolin 500 mg is a cornerstone antibiotic, particularly within surgical settings and in the management of various bacterial infections. This comprehensive guide aims to provide an exhaustive, authoritative overview of Cefazolin 500 mg, covering its mechanism, clinical applications, safety profile, and more, tailored for both medical professionals and informed patients.
1. Comprehensive Introduction & Overview of Cefazolin 500 mg
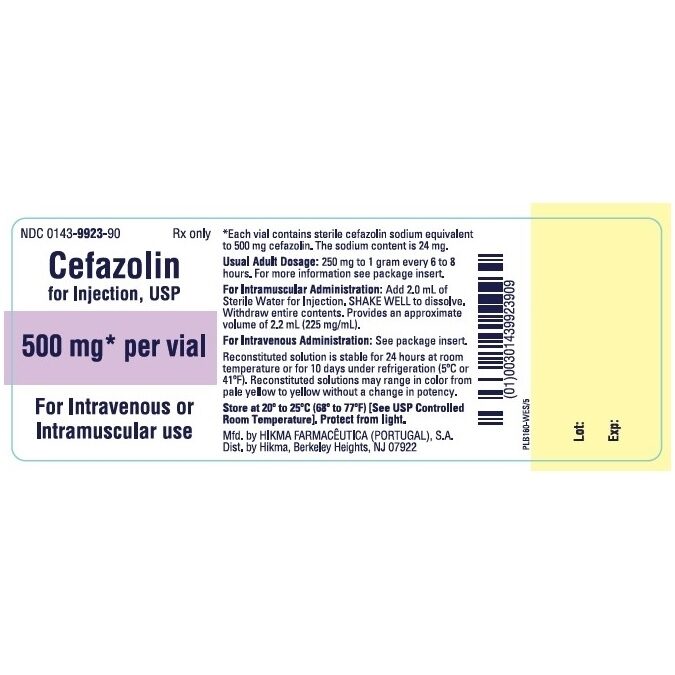
Cefazolin is a first-generation cephalosporin antibiotic, a class of beta-lactam antibiotics that are widely used due to their efficacy and relatively favorable safety profile. The "500 mg" in "Cefazolin 500 mg" refers to the dosage strength, indicating the amount of the active pharmaceutical ingredient present in each vial or dose. It is typically administered intravenously (IV) or intramuscularly (IM).
Key Characteristics of Cefazolin:
- Broad-Spectrum Activity: Primarily effective against Gram-positive bacteria, including many strains of Staphylococcus aureus (excluding MRSA) and Streptococcus species. It also exhibits activity against some Gram-negative bacteria like Escherichia coli, Klebsiella pneumoniae, and Proteus mirabilis.
- Bactericidal Action: Kills bacteria rather than merely inhibiting their growth.
- Clinical Versatility: Widely employed for surgical prophylaxis, treating skin and soft tissue infections, bone and joint infections, and certain urinary tract infections.
- Orthopedic Relevance: Cefazolin is a drug of choice in orthopedic surgery for preventing surgical site infections (SSIs) and for treating established osteomyelitis or septic arthritis.
This guide will delve into the intricate details of Cefazolin, providing a robust resource for understanding its therapeutic potential and safety considerations.
2. Deep-Dive into Technical Specifications & Mechanisms
Understanding how Cefazolin works at a molecular level is crucial for appreciating its clinical efficacy.
2.1. Mechanism of Action
Cefazolin, like other beta-lactam antibiotics, exerts its bactericidal effect by interfering with bacterial cell wall synthesis.
- Target: The primary target of Cefazolin is a group of enzymes known as Penicillin-Binding Proteins (PBPs), which are located in the bacterial cell membrane.
- Binding: Cefazolin irreversibly binds to these PBPs. Specifically, it inhibits the transpeptidase enzymes (a type of PBP) responsible for cross-linking peptidoglycan strands, which are essential components of the bacterial cell wall.
- Result: By preventing the formation of stable peptidoglycan cross-links, Cefazolin weakens the bacterial cell wall. This compromised wall loses its structural integrity, leading to an inability to withstand osmotic pressure differences between the bacterial cytoplasm and the surrounding environment.
- Consequence: The bacterial cell swells and ultimately lyses (bursts), resulting in bacterial death. This makes Cefazolin a bactericidal agent.
2.2. Pharmacokinetics
Pharmacokinetics describes how the body handles a drug – absorption, distribution, metabolism, and excretion (ADME).
- Absorption:
- Intramuscular (IM): Rapidly and completely absorbed. Peak plasma concentrations are typically achieved within 1-2 hours.
- Intravenous (IV): Administered directly into the bloodstream, resulting in immediate and complete bioavailability. Peak plasma concentrations are reached shortly after the end of infusion.
- Distribution:
- Wide Distribution: Cefazolin distributes widely into most body fluids and tissues, including bone, synovial fluid (joint fluid), pleural fluid, pericardial fluid, bile, and aqueous humor.
- Therapeutic Concentrations: Achieves therapeutic concentrations in many areas relevant to orthopedic infections, such as bone and joint tissues.
- Poor CNS Penetration: Does not readily penetrate the blood-brain barrier, especially when meninges are not inflamed, making it less suitable for central nervous system infections.
- Protein Binding: Highly protein-bound, approximately 70-86%, primarily to albumin.
- Metabolism:
- Cefazolin is minimally metabolized in the body. It is excreted largely unchanged.
- Elimination:
- Primary Route: Excretion is predominantly renal. Approximately 80-90% of an administered dose is excreted unchanged in the urine within 24 hours via glomerular filtration and tubular secretion.
- Half-life: The plasma elimination half-life is approximately 1.8 hours in individuals with normal renal function. This half-life is significantly prolonged in patients with impaired renal function, necessitating dose adjustments.
3. Extensive Clinical Indications & Usage
Cefazolin 500 mg is a versatile antibiotic with a broad range of approved indications. Its efficacy against key Gram-positive pathogens makes it invaluable in many clinical scenarios.
3.1. Detailed Indications
Cefazolin is indicated for the treatment of infections caused by susceptible microorganisms.
- Surgical Prophylaxis: This is one of Cefazolin's most critical uses, especially in orthopedic surgery.
- Orthopedic Procedures: Joint replacements (hip, knee), fracture repair, spinal surgeries, and other major bone and joint procedures.
- General Surgery: Abdominal surgery, biliary tract surgery, hysterectomy, cardiac surgery.
- Timing: Administered intravenously within 60 minutes prior to surgical incision to ensure adequate tissue concentrations during the critical period of wound contamination.
- Duration: Typically continued for a short duration post-operatively (usually 24 hours, but can be extended based on clinical judgment or specific protocols) to minimize the risk of surgical site infections.
- Skin and Soft Tissue Infections:
- Cellulitis, erysipelas, abscesses (often after drainage), impetigo, wound infections.
- Effective against Staphylococcus aureus (methicillin-susceptible) and Streptococcus pyogenes.
- Bone and Joint Infections:
- Osteomyelitis: Infection of the bone, which can be acute or chronic. Cefazolin's excellent bone penetration makes it a suitable choice.
- Septic Arthritis: Infection within a joint, often requiring surgical drainage in addition to antibiotics.
- Prosthetic Joint Infections: Often part of a multi-drug regimen or for prophylaxis during revision surgery.
- Respiratory Tract Infections:
- Pneumonia, bronchitis, pleurisy caused by susceptible Streptococcus pneumoniae or Staphylococcus aureus.
- Urinary Tract Infections (UTIs):
- Complicated and uncomplicated UTIs, including pyelonephritis and cystitis, caused by susceptible E. coli, Proteus mirabilis, and Klebsiella species.
- Bacteremia/Septicemia:
- Treatment of bloodstream infections caused by susceptible organisms, often originating from other primary infection sites.
- Endocarditis Prophylaxis:
- For specific high-risk patients undergoing certain dental or surgical procedures.
3.2. Dosage Guidelines
Dosage of Cefazolin must be individualized based on the severity of the infection, the susceptibility of the causative organism, the patient's renal function, and body weight.
Typical Adult Dosing:
| Indication | Dosage | Frequency |
|---|---|---|
| Surgical Prophylaxis | 1 g IV | 30-60 minutes prior to incision, then 0.5-1 g every 6-8 hours for 24 hours post-op |
| Mild to Moderate Infections | 250 mg to 1 g IV or IM | Every 6 to 8 hours |
| Moderate to Severe Infections | 500 mg to 1 g IV or IM | Every 6 to 8 hours |
| Severe, Life-Threatening Infections | 1 g to 1.5 g IV or IM | Every 6 hours |
| Acute Uncomplicated Urinary Tract Infections | 1 g IV or IM | Every 12 hours |
Pediatric Dosing:
- General Infections: 25-100 mg/kg/day, divided into doses administered every 6 to 8 hours. The total daily dose should not exceed the maximum adult dose.
Dosage Adjustment in Renal Impairment:
For patients with impaired renal function, the dosing interval or dose must be adjusted to prevent accumulation and potential toxicity.
| Creatinine Clearance (CrCl) mL/min | Recommended Cefazolin Dosage | Frequency |
|---|---|---|
| > 55 | Normal dose | Every 8 hours |
| 35-54 | Normal dose | Every 12 hours |
| 11-34 | Half of normal dose | Every 12 hours |
| < 10 | Half of normal dose | Every 18-24 hours |
For patients on hemodialysis, a supplemental dose may be required after dialysis.
Administration:
- Intravenous (IV): Cefazolin can be administered by slow IV push over 3-5 minutes or by intermittent IV infusion over 30-60 minutes. It should be reconstituted with sterile water for injection, 0.9% sodium chloride, or other compatible IV fluids.
- Intramuscular (IM): Reconstitute with sterile water for injection. IM injections should be given deep into a large muscle mass (e.g., gluteal region) to minimize pain.
4. Risks, Side Effects, or Contraindications
While generally well-tolerated, Cefazolin, like all medications, carries potential risks and side effects.
4.1. Contraindications
- Hypersensitivity: Cefazolin is strictly contraindicated in patients with a known severe hypersensitivity (e.g., anaphylaxis, severe skin reaction) to cefazolin or any other cephalosporin antibiotic.
- Penicillin Allergy: Due to potential cross-reactivity, Cefazolin should be used with extreme caution in patients with a history of severe immediate hypersensitivity reactions (e.g., anaphylaxis, angioedema, urticaria) to penicillin. The risk of cross-reactivity between penicillins and first-generation cephalosporins like Cefazolin is low but not negligible (estimated 1-10%).
4.2. Common Side Effects
- Gastrointestinal: Nausea, vomiting, diarrhea, abdominal cramps, anorexia. These are usually mild and transient.
- Local Reactions:
- IM Injection: Pain, tenderness, induration at the injection site.
- IV Infusion: Phlebitis (inflammation of the vein) or thrombophlebitis (clot formation with inflammation) at the injection site.
- Allergic Reactions: Rash, pruritus (itching), urticaria (hives).
4.3. Serious Side Effects (Rare but Significant)
- Anaphylaxis: A severe, life-threatening allergic reaction characterized by widespread hives, swelling (angioedema), difficulty breathing, and a drop in blood pressure. Requires immediate medical attention.
- Clostridioides difficile-associated Diarrhea (CDAD): Can range from mild diarrhea to severe, life-threatening pseudomembranous colitis. This can occur during or after antibiotic treatment.
- Seizures: Particularly in patients with renal impairment who receive inappropriately high doses.
- Nephrotoxicity: While rare with Cefazolin alone, the risk may increase when co-administered with other nephrotoxic drugs (e.g., aminoglycosides).
- Hematologic Effects: Eosinophilia, leukopenia, neutropenia, thrombocytopenia, positive Coombs' test, and very rarely, hemolytic anemia.
- Hepatotoxicity: Transient elevations in liver enzymes (AST, ALT, alkaline phosphatase).
4.4. Warnings and Precautions
- History of Penicillin Allergy: Carefully assess the nature of previous reactions. If severe, Cefazolin should generally be avoided.
- Renal Impairment: Dose adjustment is critical to prevent accumulation and adverse effects.
- Prolonged Use: May lead to superinfection (overgrowth of non-susceptible organisms, including fungi) or C. difficile infection.
- Coagulation Abnormalities: Very rarely, Cefazolin can affect vitamin K metabolism, leading to hypoprothrombinemia and bleeding. Monitor INR in at-risk patients, especially those with renal or hepatic impairment, poor nutritional status, or on long-term therapy.
4.5. Drug Interactions
- Probenecid: Co-administration with probenecid (a uricosuric agent) decreases the renal tubular secretion of Cefazolin, leading to increased and prolonged plasma concentrations. This interaction can be used therapeutically to enhance Cefazolin levels, but also increases the risk of side effects.
- Anticoagulants (e.g., Warfarin): Cefazolin may rarely potentiate the effects of oral anticoagulants, increasing the risk of bleeding. Close monitoring of INR (International Normalized Ratio) is recommended.
- Nephrotoxic Drugs: Concurrent use with other drugs known to cause kidney damage (e.g., aminoglycosides, loop diuretics like furosemide) may increase the risk of nephrotoxicity.
- Live Bacterial Vaccines: Cefazolin, as an antibiotic, may reduce the therapeutic effect of live bacterial vaccines (e.g., BCG vaccine, typhoid vaccine).
- Alcohol: While less common than with some other cephalosporins (e.g., cefotetan, cefoperazone), a disulfiram-like reaction (flushing, headache, nausea, vomiting, chest pain) has been reported with Cefazolin and alcohol in rare instances. Patients should be advised to avoid alcohol during treatment.
4.6. Pregnancy and Lactation Warnings
- Pregnancy (FDA Pregnancy Category B):
- Animal reproduction studies have not demonstrated a risk to the fetus.
- There are no adequate and well-controlled studies in pregnant women.
- Cefazolin should be used during pregnancy only if clearly needed and the potential benefits outweigh the potential risks to the fetus.
- It is generally considered one of the safer antibiotics for use during pregnancy when indicated.
- Lactation (Breastfeeding):
- Cefazolin is excreted in human breast milk in low concentrations.
- While generally considered safe, caution should be exercised.
- Monitor breastfed infants for potential adverse effects such as diarrhea, candidiasis (thrush), or allergic reactions (rash).
- The benefits of breastfeeding should be weighed against the potential risks to the infant and the mother's clinical need for Cefazolin.
4.7. Overdose Management
In the event of an overdose with Cefazolin, symptoms are generally an extension of its known side effects and may include:
- Nausea, vomiting, diarrhea, epigastric distress.
- In severe cases, particularly in patients with renal insufficiency, CNS effects such as seizures, tremors, and myoclonic jerks can occur.
Management of Overdose:
- Symptomatic and Supportive Care: There is no specific antidote for Cefazolin overdose. Treatment should focus on managing symptoms and providing supportive care.
- Maintain Vital Functions: Ensure adequate airway, breathing, and circulation.
- Monitor: Closely monitor renal function, electrolytes, and neurological status.
- Hemodialysis: Cefazolin can be removed from the bloodstream by hemodialysis or peritoneal dialysis. In cases of severe overdose, especially with significant renal impairment, dialysis may be considered to accelerate drug elimination.
5. Massive FAQ Section
Here are some frequently asked questions about Cefazolin 500 mg, providing quick answers to common concerns.
Q1: What is Cefazolin 500 mg used for?
A1: Cefazolin 500 mg is an antibiotic primarily used to treat a variety of bacterial infections, including skin and soft tissue infections, bone and joint infections (like osteomyelitis and septic arthritis), urinary tract infections, and respiratory tract infections. It is also very commonly used for surgical prophylaxis to prevent infections before and during surgery, especially in orthopedic procedures.
Q2: How is Cefazolin administered?
A2: Cefazolin is typically administered intravenously (IV) as an injection or infusion, or intramuscularly (IM) as an injection into a large muscle. The 500 mg strength refers to the amount of medication in each dose, which is then reconstituted and prepared for injection by a healthcare professional.
Q3: Is Cefazolin safe if I have a penicillin allergy?
A3: If you have a history of a severe, immediate allergic reaction to penicillin (e.g., anaphylaxis, angioedema), Cefazolin should generally be avoided due to a small but present risk of cross-reactivity. For milder penicillin allergies (e.g., mild rash), Cefazolin may be used with caution and close monitoring. Always inform your doctor about all your allergies.
Q4: Can Cefazolin cause Clostridioides difficile infection?
A4: Yes, like many broad-spectrum antibiotics, Cefazolin can disrupt the normal balance of bacteria in the gut, which can lead to an overgrowth of Clostridioides difficile (formerly Clostridium difficile). This can cause C. difficile-associated diarrhea (CDAD), ranging from mild to severe, potentially life-threatening colitis. Seek medical attention if you develop severe or persistent diarrhea during or after Cefazolin treatment.
Q5: How quickly does Cefazolin start working?
A5: When administered intravenously, Cefazolin begins to work almost immediately, reaching therapeutic concentrations in the bloodstream and tissues rapidly. For infections, you might start to feel better within 24-48 hours, but it's crucial to complete the full course of treatment as prescribed. For surgical prophylaxis, it's given just before surgery to ensure protection from the start.
Q6: What should I do if I miss a dose of Cefazolin?
A6: Since Cefazolin is typically administered by a healthcare professional in a clinical setting, missed doses are uncommon. However, if you are receiving it on an outpatient basis and miss an appointment for your dose, contact your doctor or clinic immediately to reschedule. Do not double up on doses.
Q7: Can Cefazolin be used during pregnancy or breastfeeding?
A7: Cefazolin is classified as FDA Pregnancy Category B, meaning animal studies have not shown harm, but human studies are limited. It is generally considered one of the safer antibiotics for use in pregnancy when clearly needed. It is excreted in low concentrations in breast milk. While generally considered safe, infants should be monitored for potential side effects like diarrhea or rash. Always discuss the risks and benefits with your doctor.
Q8: Are there any foods or drinks I should avoid while on Cefazolin?
A8: Generally, there are no specific food interactions with Cefazolin. However, it's advisable to avoid alcohol, as rare reports of disulfiram-like reactions (nausea, flushing, headache) have been noted with Cefazolin, though it's more common with other cephalosporins. Always follow your doctor's specific dietary advice.
Q9: What are the most common side effects of Cefazolin?
A9: The most common side effects include gastrointestinal issues like nausea, vomiting, and diarrhea. Local reactions at the injection site, such as pain, tenderness, or phlebitis (vein inflammation), are also common. Mild allergic reactions like skin rash or itching can occur.
Q10: How long will I need to take Cefazolin?
A10: The duration of Cefazolin treatment varies depending on the type and severity of the infection. For surgical prophylaxis, it's often given for only 24 hours. For active infections, treatment can range from a few days to several weeks, especially for severe bone or joint infections. Always complete the full course of treatment prescribed by your doctor, even if you start feeling better, to prevent the infection from returning and to reduce the risk of antibiotic resistance.
Q11: Is Cefazolin effective against MRSA?
A11: No, Cefazolin is generally not effective against Methicillin-Resistant Staphylococcus aureus (MRSA). Cefazolin is a first-generation cephalosporin and primarily targets methicillin-susceptible S. aureus (MSSA). If MRSA is suspected or confirmed, different antibiotics are required.
Q12: Why is Cefazolin often used for surgery?
A12: Cefazolin is a preferred antibiotic for surgical prophylaxis because it has several ideal characteristics:
* Effective Spectrum: Covers common bacteria that cause surgical site infections, particularly Staphylococcus aureus and Streptococcus species.
* Good Tissue Penetration: Achieves high concentrations in surgical tissues, including bone and joint fluid.
* Favorable Pharmacokinetics: Has a relatively long half-life, allowing for convenient dosing schedules around surgery.
* Low Toxicity: Generally well-tolerated with a good safety profile.
* Cost-Effective: Often a more economical choice compared to newer antibiotics with similar efficacy for prophylaxis.