The Baclofen Pump Refill: A Comprehensive Medical SEO Guide to Intrathecal Spasticity Management
For individuals living with severe, chronic spasticity, an intrathecal baclofen (ITB) pump can be a life-changing therapy. This advanced medical device delivers baclofen directly to the spinal cord, offering superior spasticity control with fewer systemic side effects compared to oral medications. A critical component of this therapy's success is the regular and precise "Baclofen Pump Refill." This exhaustive guide, curated by an expert Medical SEO Copywriter and Orthopedic Specialist, delves into every facet of the Baclofen pump refill process, providing authoritative insights for patients, caregivers, and healthcare professionals.
Understanding the Intrathecal Baclofen Pump System
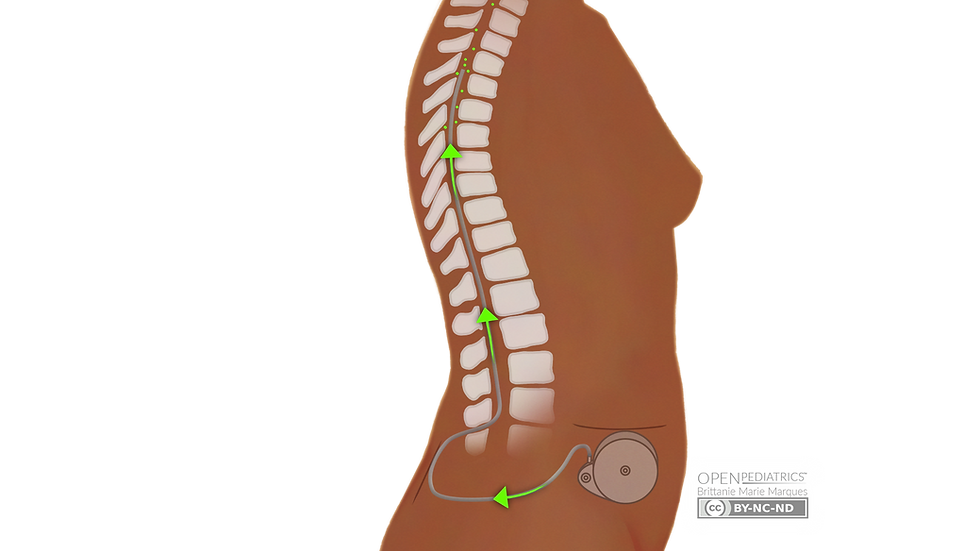
The intrathecal baclofen pump system consists of an implanted, programmable pump (typically placed in the abdominal wall) and a catheter that delivers baclofen directly into the intrathecal space surrounding the spinal cord. This direct delivery bypasses the blood-brain barrier and allows for significantly lower doses of baclofen to achieve therapeutic effects, minimizing systemic side effects.
The pump contains a reservoir that holds liquid baclofen. As this reservoir depletes, it requires periodic refilling – the "Baclofen Pump Refill" – to ensure continuous medication delivery and sustained spasticity control.
Deep Dive into Baclofen's Mechanism of Action and Pharmacokinetics
Baclofen, a gamma-aminobutyric acid (GABA) derivative, is a centrally acting skeletal muscle relaxant. Its efficacy in managing spasticity stems from its unique mechanism of action and the optimized pharmacokinetics achieved through intrathecal delivery.
Mechanism of Action
Baclofen acts as an agonist at the GABA-B receptors located on the presynaptic terminals of afferent nerve fibers in the spinal cord. By activating these receptors, baclofen inhibits the release of excitatory neurotransmitters, such as glutamate and aspartate. This leads to:
- Hyperpolarization of primary afferent terminals: This reduces the excitability of spinal motor neurons.
- Reduced monosynaptic and polysynaptic reflex transmission: This directly decreases the frequency and severity of muscle spasms and improves muscle tone.
In essence, baclofen calms the overactive nerve signals in the spinal cord that cause spasticity, allowing for greater muscle relaxation and improved functional mobility.
Pharmacokinetics of Intrathecal Baclofen
The intrathecal route of administration dramatically alters baclofen's pharmacokinetic profile compared to oral delivery.
- Direct Delivery: Baclofen is delivered directly into the cerebrospinal fluid (CSF), allowing it to reach GABA-B receptors in the spinal cord rapidly and efficiently.
- High Local Concentration: This method achieves therapeutic concentrations at the target site (spinal cord) with much lower overall doses, often 1/100th to 1/1000th of oral doses.
- Minimal Systemic Absorption: Only a small fraction of intrathecally delivered baclofen reaches the systemic circulation, significantly reducing systemic side effects like profound sedation or generalized weakness often associated with high oral doses.
- Rapid Onset: Effects are typically observed within hours of initiating infusion or dose adjustments, though full therapeutic effect may take days.
- Sustained Effect: Continuous infusion maintains stable concentrations at the spinal cord, providing consistent spasticity control throughout the day.
- Metabolism and Excretion: Once absorbed into the systemic circulation, baclofen is primarily excreted unchanged by the kidneys. A small percentage is metabolized in the liver.
Extensive Clinical Indications and Usage of Intrathecal Baclofen Therapy
Intrathecal baclofen therapy, and consequently its regular refill, is indicated for patients with severe chronic spasticity who have not responded adequately to other treatments, such as oral antispasticity medications, or who experience intolerable side effects at effective oral doses. The primary goal is to reduce spasticity, improve function, and alleviate pain.
Detailed Indications
| Indication Category | Specific Conditions | Key Characteristics |
|---|---|---|
| Cerebral Origin | Cerebral Palsy (CP) | Severe, generalized spasticity, often impacting gait, posture, and self-care. |
| Stroke | Persistent, debilitating spasticity post-stroke, often unilateral. | |
| Traumatic Brain Injury (TBI) | Chronic spasticity resulting from brain injury. | |
| Anoxia | Spasticity due to oxygen deprivation to the brain. | |
| Spinal Origin | Multiple Sclerosis (MS) | Progressive spasticity, often accompanied by spasms and pain. |
| Spinal Cord Injury (SCI) | Severe spasticity below the level of injury, interfering with mobility and activities of daily living. | |
| Transverse Myelitis | Spasticity resulting from inflammation of the spinal cord. | |
| Other Myelopathies | Various conditions causing spinal cord damage and spasticity. |
Patient Selection Criteria
Patients considered for ITB therapy typically undergo a rigorous screening process, often involving a test dose of intrathecal baclofen to assess responsiveness. Key criteria include:
- Severe, chronic spasticity: Significantly impairing function, causing pain, or hindering care.
- Inadequate response to oral antispasticity agents: Or unacceptable side effects at therapeutic doses.
- Successful screening test: Demonstrating a significant reduction in spasticity with a temporary intrathecal dose.
- Absence of contraindications: (discussed below).
- Reliable support system: For ongoing care and refill appointments.
- Realistic expectations: Understanding the goals and limitations of therapy.
The Baclofen Pump Refill Process
The refill is a sterile, outpatient procedure performed by a trained healthcare professional (e.g., neurosurgeon, neurologist, pain specialist, or nurse practitioner).
- Preparation: The skin over the pump site is meticulously cleaned and sterilized.
- Access: A special non-coring needle is used to access the pump's refill port, which is designed to prevent coring of the septum.
- Aspiration (Optional but Recommended): A small amount of residual baclofen may be aspirated to confirm needle placement and measure remaining volume, ensuring accurate refill calculations.
- Refill: The appropriate concentration and volume of baclofen are slowly injected into the pump reservoir.
- Programming (Crucial): The pump is then interrogated and reprogrammed using a handheld programmer. This allows for:
- Verification of current dose and remaining volume.
- Adjustment of dosage or infusion rate based on the patient's current spasticity levels and clinical response.
- Setting of refill reminders and alarms.
- Documentation: All details of the refill, including volume, concentration, dose adjustments, and next refill date, are thoroughly documented.
Dosage Guidelines and Management
ITB therapy is highly individualized. There are no fixed dosage guidelines; rather, dosage is titrated to achieve optimal spasticity control with minimal side effects.
- Initial Dosing: Following successful screening, the initial dose is typically very low and gradually increased over days to weeks in a hospital setting.
- Maintenance Dosing: Once a stable therapeutic dose is achieved, it is maintained through continuous infusion. Doses can range widely, from tens of micrograms to over 1000 micrograms per day.
- Programmable Pumps: Modern pumps allow for complex dosing schedules, including:
- Continuous infusion: A steady dose throughout the day.
- Bolus dosing: Higher doses at specific times (e.g., before physical therapy or at night).
- Adjustments: Doses are adjusted based on patient response, activity levels, and potential side effects.
- Refill Frequency: The frequency of refills depends on the pump's reservoir size, the patient's daily dose, and the concentration of baclofen used. It typically ranges from every 1 to 3 months. Missing a refill can lead to severe baclofen withdrawal.
Risks, Side Effects, and Contraindications Associated with Baclofen Pump Refill
While ITB therapy is highly effective, it is not without risks. Understanding these is crucial for patient safety and optimal management.
Contraindications
- Hypersensitivity: Known allergy to baclofen.
- Acute Infection: Especially at the surgical site or systemic infection, which increases the risk of meningitis.
- Unsuccessful Screening: Failure to respond to an intrathecal baclofen test dose.
- Pump Malfunction/Complications: Unresolved issues with the implanted device.
- Body Size/Weight: Insufficient body size to accommodate the pump.
Potential Risks and Side Effects Related to the Pump System
| Category | Specific Risks | Description |
|---|---|---|
| Surgical Risks | Infection (meningitis, wound infection) | Can be severe and life-threatening, requiring explantation and antibiotic treatment. |
| CSF Leak | May cause headaches, nausea, or require surgical repair. | |
| Catheter Malposition/Kinking/Breakage | Can lead to underdosing, loss of efficacy, or baclofen withdrawal. Requires revision surgery. | |
| Hematoma/Seroma | Collection of blood or fluid at the surgical site. | |
| Device Risks | Pump Malfunction/Failure | Can lead to sudden cessation of baclofen delivery and severe withdrawal. Requires urgent intervention. |
| Reservoir Depletion | If refill is missed or delayed, leading to withdrawal. | |
| Catheter Disconnection | Leads to loss of drug delivery. | |
| Refill-Specific | Overdose during refill | Due to incorrect concentration, volume, or programming error. |
| Needle misplacement (into subcutaneous tissue) | Leading to systemic absorption and potential overdose, or failure to deliver baclofen intrathecally. |
Baclofen-Related Side Effects (Often Dose-Dependent)
- Hypotonia (Flaccidity): Excessive muscle weakness.
- Sedation/Drowsiness: Can impair cognitive function.
- Nausea/Vomiting: Gastrointestinal discomfort.
- Headache: Common, especially if CSF pressure changes.
- Dizziness: Vertigo or lightheadedness.
- Hypotension: Low blood pressure.
- Urinary Retention/Incontinence: Bladder dysfunction.
- Seizures: Rare, but can occur, especially with rapid dose changes or withdrawal.
Baclofen Withdrawal Syndrome (A Medical Emergency)
This is a critical risk if a refill is missed, delayed, or if the pump malfunctions. Symptoms can develop rapidly and include:
- Increased spasticity, hypertonia, and rigidity
- Pruritus (itching)
- Dysautonomia (fever, altered mental status, tachycardia, labile blood pressure)
- Rhabdomyolysis
- Seizures
- Hallucinations
- Coma
- Death
Immediate medical attention is required for suspected baclofen withdrawal.
Drug Interactions
While intrathecal delivery minimizes systemic interactions, caution is still warranted, particularly with medications that affect the central nervous system.
- CNS Depressants: Co-administration with other CNS depressants (e.g., opioids, benzodiazepines, sedatives, alcohol, general anesthetics) can lead to additive effects, increasing sedation, respiratory depression, and hypotension. Close monitoring is essential.
- Antihypertensives: Baclofen may cause a further decrease in blood pressure when used concurrently with antihypertensive agents.
- Tricyclic Antidepressants (TCAs): There have been reports of increased hypotonia with concomitant use of TCAs and oral baclofen; this potential interaction should be considered with ITB.
- Other Antispasticity Agents: While ITB often replaces oral antispasticity drugs, if used concurrently, their effects may be additive.
Pregnancy and Lactation Warnings
Pregnancy (FDA Pregnancy Category C)
- There are no adequate and well-controlled studies of intrathecal baclofen in pregnant women.
- Animal studies have shown adverse effects on development at doses significantly higher than typical human doses.
- Baclofen should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus.
- If a woman becomes pregnant while on ITB therapy, the risks and benefits should be carefully re-evaluated by her healthcare team. Sudden discontinuation can be dangerous.
Lactation
- Baclofen is excreted in human milk after oral administration. It is unknown whether intrathecally administered baclofen is excreted in human milk, but systemic absorption is minimal.
- Caution should be exercised when baclofen is administered to a nursing mother. The decision to continue or discontinue nursing or baclofen therapy should take into account the importance of the drug to the mother and the potential risks to the infant.
Overdose Management
Baclofen overdose is a serious medical emergency, particularly if it occurs intrathecally. Prompt recognition and intervention are critical.
Symptoms of Overdose
- Profound Hypotonia: Severe muscle weakness, flaccidity.
- Somnolence/Drowsiness: Progressing to stupor and coma.
- Respiratory Depression: Shallow, slow breathing, potentially leading to respiratory arrest.
- Bradycardia: Slow heart rate.
- Hypothermia: Low body temperature.
- Seizures: Can occur, particularly in severe cases.
- Loss of Consciousness: Unresponsiveness.
- Pinpoint Pupils: Miosis.
Management of Overdose
- Immediate Medical Attention: Call emergency services immediately.
- Discontinue Baclofen Delivery: If a programmable pump is involved, the infusion rate should be immediately decreased to the lowest possible setting or temporarily stopped by a trained professional. For non-programmable pumps, surgical intervention may be required to empty the reservoir or remove the pump.
- Supportive Care:
- Respiratory Support: Maintain a patent airway. Endotracheal intubation and mechanical ventilation may be necessary, especially for respiratory depression.
- Cardiovascular Support: Monitor vital signs closely. Administer intravenous fluids and vasopressors as needed to maintain blood pressure.
- Temperature Management: Address hypothermia.
- Seizure Management: Administer anticonvulsants if seizures occur.
- Antidote Consideration (Physostigmine): While not a direct antidote for baclofen, physostigmine has been used to reverse some of the CNS depressant effects in severe oral baclofen overdose. Its role in intrathecal overdose is less established and should only be considered by specialists with extreme caution, given its potential for adverse cardiac effects.
- CSF Drainage: In severe intrathecal overdose, withdrawing cerebrospinal fluid (CSF) via lumbar puncture can help reduce baclofen concentration in the intrathecal space.
- Monitoring: Continuous monitoring of neurological status, respiratory function, and vital signs is essential until the patient stabilizes.
Prevention is key: Strict adherence to refill protocols, accurate dosage calculations, proper needle placement during refill, and careful programming are paramount to preventing overdose.
Massive FAQ Section: Your Questions About Baclofen Pump Refills Answered
Q1: What is a Baclofen pump refill and why is it necessary?
A1: A Baclofen pump refill is a routine medical procedure where the medication reservoir of an implanted intrathecal baclofen pump is replenished. It's necessary because the pump continuously delivers baclofen to your spinal cord, and the reservoir will eventually run out. Regular refills ensure uninterrupted therapy and consistent spasticity control.
Q2: How often do I need a Baclofen pump refill?
A2: The frequency of refills varies depending on your daily baclofen dose, the concentration of the medication, and the size of your pump's reservoir. Most patients require a refill every 1 to 3 months. Your healthcare provider will schedule your refills based on your individual needs.
Q3: What happens if I miss a Baclofen pump refill appointment?
A3: Missing a refill appointment is a serious medical concern. It can lead to baclofen withdrawal syndrome, which is a life-threatening condition characterized by increased spasticity, fever, altered mental status, seizures, and even death. If you miss or anticipate missing an appointment, contact your healthcare provider immediately.
Q4: What should I expect during a Baclofen pump refill procedure?
A4: The procedure is typically done in an outpatient clinic. You'll lie down, and the skin over your pump site (usually in your abdomen) will be cleaned and sterilized. Your doctor or nurse will use a special needle to access the pump's refill port, withdraw any remaining medication (optional), and then inject the new baclofen solution. The pump will then be reprogrammed to ensure correct dosing. The process is usually quick, lasting about 15-30 minutes.
Q5: Is the Baclofen pump refill painful?
A5: Most patients experience minimal discomfort. The refill port is located just under the skin. A local anesthetic may be used to numb the area before the needle is inserted. You might feel a slight pressure or sting.
Q6: Can my baclofen dose be adjusted during a refill?
A6: Yes, absolutely. The refill appointment is an opportune time for your healthcare provider to assess your spasticity control, discuss any side effects, and adjust your baclofen dose if needed. The pump is programmable, allowing for precise dose changes.
Q7: What are the potential risks associated with the refill procedure itself?
A7: Risks are generally low but include infection at the refill site, needle misplacement (leading to subcutaneous delivery of baclofen or a "pocket fill"), or, rarely, an accidental overdose if incorrect volume or concentration is administered. These risks are minimized by performing the procedure in a sterile environment by trained professionals.
Q8: How can I prepare for my Baclofen pump refill appointment?
A8: Follow any specific instructions from your clinic, such as showering with an antiseptic soap. Be prepared to discuss your current spasticity levels, any changes in your symptoms, and any side effects you might be experiencing. Bring a list of all your current medications.
Q9: What are the signs of baclofen overdose or withdrawal that I should watch for after a refill?
A9:
* Overdose: Excessive muscle weakness (floppiness), severe drowsiness, very slow or shallow breathing, dizziness, confusion, or loss of consciousness.
* Withdrawal: Sudden increase in spasticity, itching, fever, confusion, rapid heart rate, or seizures.
Report any of these symptoms to your healthcare provider immediately.
Q10: Can I travel with a Baclofen pump?
A10: Yes, you can travel with an intrathecal baclofen pump. It's advisable to carry your pump identification card. Inform airport security about your implanted device. The pump is designed to withstand normal atmospheric pressure changes during air travel. Always ensure you have enough medication for your trip and know how to contact medical assistance if needed.
Q11: Are there different concentrations of baclofen used for refills?
A11: Yes, baclofen for intrathecal delivery comes in various concentrations (e.g., 50 mcg/mL, 500 mcg/mL, 2000 mcg/mL). The concentration chosen depends on your daily dose requirements and the desired refill frequency. Higher concentrations allow for longer intervals between refills for patients on higher daily doses.
Q12: How long does the baclofen pump itself last?
A12: The battery life of an intrathecal baclofen pump typically ranges from 5 to 7 years, though some newer models may last longer. When the battery nears depletion, the pump will need to be surgically replaced in a procedure similar to the initial implantation.
Q13: Does the Baclofen pump cure spasticity?
A13: No, the Baclofen pump does not cure spasticity. It is a highly effective management tool that helps to reduce the severity of spasticity and associated symptoms, improving comfort, function, and quality of life. It requires ongoing therapy and regular refills.
Q14: What if the pump makes an alarm sound?
A14: If your pump emits an alarm sound, it indicates a critical issue such as a low battery, an empty reservoir, or a pump malfunction. Contact your healthcare provider immediately. Do not wait, as this could lead to baclofen withdrawal or other serious complications.
This comprehensive guide underscores the critical nature of the Baclofen pump refill in maintaining effective spasticity management. Adherence to refill schedules, vigilant monitoring, and close collaboration with your healthcare team are paramount for the safety and success of intrathecal baclofen therapy.