Comprehensive Introduction and Patho-Epidemiology
The management of chronic Achilles tendon ruptures represents a formidable challenge in orthopedic surgery, demanding a sophisticated understanding of altered local biology, biomechanics, and soft-tissue envelope constraints. Unlike acute ruptures, where direct end-to-end apposition is typically achievable, chronic ruptures—classically defined as those presenting greater than three months post-injury—are characterized by substantial tendon retraction, massive gap formation, and profound degenerative changes within the musculotendinous unit. The pathological cascade initiated by a neglected or misdiagnosed rupture fundamentally alters the surgical landscape, rendering primary repair impossible and necessitating complex reconstructive strategies such as allograft augmentation or replacement.
The pathophysiology of chronic Achilles rupture is rooted in a failed, disorganized healing response. Following the initial mechanical failure, the unopposed proximal pull of the powerful gastrocnemius-soleus complex causes immediate and progressive retraction of the proximal tendon stump. Concurrently, the intervening hematoma undergoes a process of fibrous organization. However, due to the relative hypovascularity of the region and the absence of mechanical tension to guide cellular alignment, the resulting scar tissue is mechanically inferior. Histological analysis of this pseudotendon reveals extensive fibrovascular proliferation, lymphocytic infiltration, and myxomatous degeneration. The highly organized, longitudinally oriented type I collagen fibers characteristic of a healthy tendon are replaced by a chaotic matrix of type III collagen. This elongated, weak fibrous mass is wholly incapable of withstanding the extreme physiological tensile forces generated during the stance phase of gait.
Epidemiologically, chronic ruptures often occur in patients with pre-existing, silent tendinopathy. The watershed area, located 2 to 6 cm proximal to the calcaneal insertion, is notoriously hypovascular and susceptible to microtraumatic cumulative injury. Risk factors exacerbating this vulnerability include systemic conditions such as diabetes mellitus, gout, and hyperthyroidism, as well as iatrogenic factors like local corticosteroid injections and systemic fluoroquinolone administration. Over time, the triceps surae muscle belly undergoes progressive atrophy and fatty infiltration, further complicating functional recovery even after structural continuity is restored. Allograft reconstruction, utilizing a fresh-frozen Achilles tendon with a calcaneal bone block, has emerged as a robust, biomechanically superior solution for bridging massive defects, restoring the resting length of the musculotendinous unit, and providing a durable construct capable of enduring aggressive postoperative rehabilitation.
Detailed Surgical Anatomy and Biomechanics
Musculotendinous Architecture and the Triceps Surae
The Achilles tendon is the largest and strongest tendon in the human body, formed by the confluence of the aponeuroses of the gastrocnemius (medial and lateral heads) and the soleus muscles. This complex, known collectively as the triceps surae, acts as the primary plantarflexor of the ankle. The gastrocnemius crosses both the knee and the ankle joints, making its tension highly dependent on knee position, whereas the soleus crosses only the ankle. The musculotendinous junction typically lies 6 to 8 cm proximal to its insertion. As the tendon descends, its fibers undergo a complex 90-degree internal rotation, with the gastrocnemius fibers inserting laterally and the soleus fibers inserting medially on the posterior calcaneal tuberosity. This spiraling architecture is critical for the tendon's ability to store and release elastic energy during locomotion.
Vascular Supply and the Paratenon Envelope
Unlike tendons enclosed within a true synovial sheath, the Achilles tendon is surrounded by a paratenon—a bilaminar layer of highly vascularized areolar tissue that functions to reduce friction and provide the primary blood supply to the epitenon. The intrinsic vascular supply is derived from the musculotendinous junction proximally and the osseous insertion distally. The extrinsic supply, traversing the mesotenon anteriorly, is supplied by branches of the posterior tibial and peroneal arteries. Angiographic studies have definitively identified a "watershed" zone of relative hypovascularity 2 to 6 cm proximal to the calcaneal insertion. This precarious blood supply is the primary anatomical substrate for degenerative tendinopathy and subsequent rupture, dictating the necessity for meticulous paratenon preservation during surgical exposure to optimize postoperative healing.
Neurovascular Topography of the Posterior Leg
Surgical navigation of the posterior lower leg requires an intimate knowledge of the regional neurovascular anatomy to prevent devastating iatrogenic complications. The sural nerve is the most critical structure at risk during the posterior approach. Formed by the union of the medial sural cutaneous nerve (from the tibial nerve) and the sural communicating branch (from the common peroneal nerve), it descends superficially between the heads of the gastrocnemius. As it courses distally, it crosses the lateral border of the Achilles tendon approximately 9 to 12 cm proximal to the calcaneal insertion, running alongside the small saphenous vein. Dissection in the subcutaneous plane must be meticulous to identify, mobilize, and protect this nerve; injury results in painful neuromas and lateral foot numbness. Deep to the Achilles tendon, separated by the deep posterior compartment fascia and Kager's fat pad, lie the posterior tibial artery and tibial nerve. While generally protected during superficial tendon work, aggressive anterior dissection or over-penetration during calcaneal trough creation can jeopardize these deep structures.
Biomechanical Implications of Chronic Rupture
The biomechanical consequences of a chronic Achilles rupture are profound. The triceps surae generates forces up to 3 to 4 times body weight during normal walking, and up to 12.5 times body weight during running or jumping. Loss of continuity abolishes the primary plantarflexion power, severely disrupting the "second rocker" of gait—the phase where the tibia advances over the plantigrade foot. Patients exhibit a characteristic calcaneal gait, characterized by a lack of push-off, decreased step length on the contralateral side, and an inability to perform a single-leg heel raise. Prolonged discontinuity leads to adaptive shortening of the proximal muscle belly and elongation of the interposed scar tissue, fundamentally altering the length-tension relationship of the muscle. The primary biomechanical goal of allograft reconstruction is to re-establish this precise length-tension relationship, restoring the resting equinus of the foot and allowing the triceps surae to generate effective force across the ankle joint.
Exhaustive Indications and Contraindications
The decision to proceed with a massive allograft reconstruction is dictated by the morphological characteristics of the defect, the quality of the remaining host tissue, and the functional demands of the patient. Preoperative templating and intraoperative assessment are critical in determining the appropriate reconstructive algorithm.
| Parameter | Indications for Allograft Reconstruction | Relative / Absolute Contraindications |
|---|---|---|
| Defect Size | Massive defects > 5 cm after radical debridement of all fibrotic and degenerative tissue. | Defects < 3 cm (amenable to V-Y advancement or local turndown flaps). |
| Tissue Quality | Severe, diffuse tendinopathy where local host tissue is insufficient to hold suture or withstand load. | Healthy proximal muscle belly capable of excursion (favors primary repair if mobilized). |
| Surgical History | Failed prior primary repairs, failed V-Y advancements, or failed FHL transfers requiring salvage. | Active local or systemic infection (Absolute). |
| Patient Factors | High functional demands requiring restoration of maximal plantarflexion power and structural integrity. | Severe peripheral vascular disease, poorly controlled diabetes mellitus, or profound neuropathy (Relative/Absolute). |
| Soft Tissue Envelope | Adequate posterior skin coverage capable of healing over a bulky allograft construct. | Compromised posterior skin flap, active ulceration, or history of severe radiation therapy to the posterior ankle (Absolute). |
Pre-Operative Planning, Templating, and Patient Positioning
Clinical Evaluation and Imaging Modalities
Meticulous preoperative planning begins with a comprehensive clinical examination. The hallmark of a chronic rupture is a palpable gap, though this may be obscured by organized hematoma or hypertrophic scar tissue. The Thompson-Simmond test will be positive (lack of plantarflexion upon calf squeeze), and the Matles test will demonstrate increased resting dorsiflexion of the affected foot when the patient is prone with knees flexed to 90 degrees. Assessing the resting tension of the contralateral, uninjured limb is critical, as this will serve as the intraoperative template for tensioning the allograft.
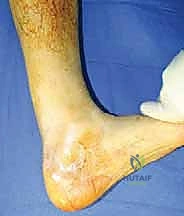
Imaging is indispensable for surgical templating. While ultrasound can provide dynamic assessment, Magnetic Resonance Imaging (MRI) remains the gold standard. MRI accurately delineates the extent of tendon retraction, the length of the true defect (excluding the pseudotendon), and the degree of fatty infiltration within the gastrocnemius and soleus muscle bellies. Severe fatty atrophy (Goutallier stage 3 or 4) may portend a poorer functional outcome regardless of the structural success of the reconstruction. Based on MRI measurements, the appropriate fresh-frozen Achilles tendon allograft with an intact calcaneal bone block is ordered from the tissue bank.
Anesthesia, Positioning, and Hemostasis
The procedure is typically performed under general anesthesia supplemented with a regional popliteal sciatic nerve block. The regional block provides excellent preemptive analgesia, minimizes volatile anesthetic requirements, and facilitates a smoother postoperative transition.
The patient is positioned prone on a radiolucent operating table. Chest rolls extending from the clavicle to the iliac crests are utilized to decompress the abdomen, decreasing venous pressure and minimizing epidural venous bleeding. The knees are slightly flexed to relax the gastrocnemius. A well-padded pneumatic tourniquet is applied to the proximal thigh. It is imperative to avoid placing the tourniquet on the calf, as this will tether the triceps surae, artificially restricting excursion and leading to catastrophic overtensioning or undertensioning of the graft. The entire lower extremity from the toes to the proximal thigh is prepped and draped in a sterile fashion to allow for intraoperative assessment of resting foot posture and full range of motion.
Step-by-Step Surgical Approach and Fixation Technique
Incision, Exposure, and Neurovascular Protection
Following exsanguination and tourniquet inflation to 300-350 mmHg, a longitudinal incision is made slightly medial to the midline of the posterior calf, extending from the musculotendinous junction to the distal aspect of the posterior calcaneal tuberosity. A slightly medial incision mitigates the risk to the sural nerve, which courses posterolaterally. Full-thickness fasciocutaneous flaps are meticulously raised. The subcutaneous tissue must not be separated from the underlying deep fascia, as preserving this vascular plexus is critical for wound healing.
The sural nerve is identified proximally and traced distally, protected with a vessel loop, and gently retracted laterally. The deep fascia is incised longitudinally, revealing the thickened, scarred paratenon. The paratenon is incised and carefully elevated off the underlying pseudotendon, preserving it for later closure over the allograft.
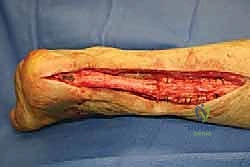
Radical Debridement of the Pathological Tendon
The fundamental principle of this reconstruction is the complete excision of all non-viable, degenerative tissue. The pseudotendon is excised en bloc. Proximally, the tendon is sharply debrided until healthy, pearly-white, longitudinally oriented collagen fibers are encountered. The quality of the proximal stump is tested by placing a heavy non-absorbable locking stitch (e.g., Krackow technique) to ensure it can withstand tension. Distally, the remnant of the Achilles tendon is completely excised from its insertion on the posterior calcaneal tuberosity, exposing the cortical bone in preparation for the osseous trough.
Allograft Preparation and Calcaneal Trough Creation
The fresh-frozen Achilles allograft is thawed in warm saline. The calcaneal bone block attached to the allograft is meticulously contoured using an oscillating saw and rongeurs into a trapezoidal or rectangular prism, typically measuring 2.0 cm wide, 2.5 cm long, and 1.5 cm deep.
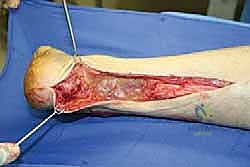
Attention is then turned to the host calcaneus. Using an oscillating saw and sharp osteotomes, a recipient trough is created in the posterior calcaneal tuberosity, perfectly matching the dimensions of the contoured allograft bone block.
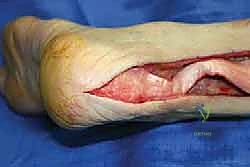
The trough must be entirely within cancellous bone to promote rapid osteointegration, but care must be taken not to breach the subtalar joint or the plantar cortex.
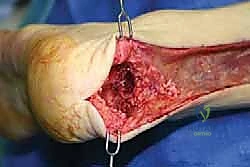
Bone Block Insertion and Rigid Internal Fixation
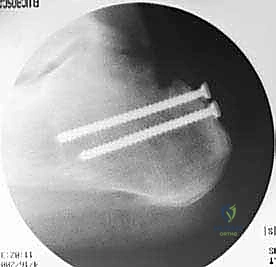
The allograft bone block is impacted into the host calcaneal trough. The fit should be highly stable (press-fit) before any hardware is introduced.
Rigid internal fixation is achieved using two fully threaded 4.0 mm or 4.5 mm titanium or stainless steel cancellous screws, placed with washers. The screws are directed from posterior to anterior, slightly superior to inferior, aiming toward the dense bone of the calcaneal body. Fluoroscopy is utilized to confirm anatomic placement of the bone block, appropriate screw trajectory, and avoidance of articular penetration.
Proximal Anastomosis, Weaving, and Precise Tensioning
With the distal insertion securely fixed, the proximal anastomosis is addressed. The allograft tendon is draped over the proximal host stump to assess the required length. A Pulvertaft weave or a fish-mouth anastomosis is utilized to maximize the surface area for biological healing.
Tensioning is the most critical and technically demanding step of the procedure. The knee is flexed to 90 degrees. The foot is placed in approximately 15 to 20 degrees of plantarflexion, strictly matching the resting equinus of the contralateral, uninjured limb.
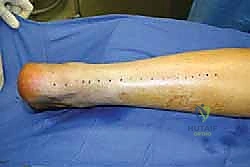
While an assistant holds the foot in this precise position, the allograft is woven through the host tendon and secured under maximal physiological tension using multiple heavy, non-absorbable sutures (e.g., #2 FiberWire or Ethibond) in a figure-of-eight or mattress configuration.
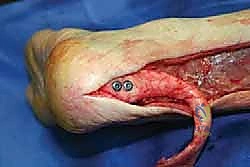
Once the primary anastomosis is secure, the tourniquet is deflated, and meticulous hemostasis is achieved. The paratenon, which was preserved during the initial exposure, is carefully closed over the allograft construct using fine absorbable sutures. This layer is vital for revascularization of the allograft and preventing adhesions to the overlying skin.
The deep subcutaneous tissue and skin are closed in layers. A sterile, non-adherent dressing is applied, and the limb is immobilized in a well-padded short-leg splint with the ankle in 15 to 20 degrees of gravity equinus to protect the reconstruction.
Complications, Incidence Rates, and Salvage Management
Despite meticulous surgical technique, allograft reconstruction of the Achilles tendon carries a distinct complication profile, primarily driven by the tenuous soft-tissue envelope and the avascular nature of the massive graft.
| Complication | Estimated Incidence | Etiology and Prevention | Salvage Management |
|---|---|---|---|
| Wound Dehiscence / Necrosis | 5% - 15% | Caused by excessive skin tension, poor flap handling, or smoking. Prevented by strict adherence to full-thickness flaps and paratenon closure. | Local wound care, negative pressure wound therapy (NPWT). Severe cases require plastic surgery consultation for local rotational flaps (e.g., sural artery flap) or free tissue transfer. |
| Deep Infection | 2% - 5% | Bacterial seeding of the avascular allograft. Prevented by strict sterility, preoperative antibiotics, and minimizing OR time. | Aggressive surgical debridement, removal of allograft if deeply seeded, placement of antibiotic spacer, and prolonged IV antibiotics. Eventual salvage with FHL transfer or free functional muscle transfer. |
| Sural Nerve Injury | 3% - 10% | Iatrogenic transection or traction neuropraxia during posterior exposure. Prevented by early identification and gentle retraction. | Neuromodulators (gabapentin). If a painful neuroma forms, surgical excision and burying of the proximal nerve stump into deep muscle belly is required. |
| Re-rupture / Graft Failure | 2% - 6% | Non-compliance with rehab, premature weight-bearing, or failure of biological incorporation. | Revision surgery. Options become severely limited; requires massive FHL transfer, turndown flaps, or pantalar arthrodesis in extreme, low-demand salvage scenarios. |
| Disease Transmission | < 0.01% | Viral or bacterial transmission from the donor tissue. Prevented by rigorous tissue bank screening and sterilization protocols. | Managed medically based on the specific pathogen transmitted. |
Phased Post-Operative Rehabilitation Protocols
Rehabilitation following massive allograft reconstruction is inherently slower and more conservative than after primary acute repair. The avascular allograft relies entirely on creeping substitution and host tissue ingrowth, a process that takes many months. A premature return to load-bearing will result in catastrophic elongation or rupture of the construct.
Phase I: Maximal Protection (Weeks 0 - 2)
The patient remains strictly non-weight-bearing (NWB) in the initial postoperative splint, immobilized in 15-20 degrees of plantarflexion. Elevation and strict pain control are emphasized. At two weeks, sutures are removed, and the wound is meticulously inspected.
Phase II: Controlled Mobilization (Weeks 2 - 6)
The patient is transitioned to a controlled ankle motion (CAM) boot equipped with a large heel lift (typically three wedges). Weight-bearing is initiated gradually, starting with touch-down weight-bearing and progressing to partial weight-bearing as tolerated. Active range of motion (ROM) out of the boot is permitted, but strictly limited to plantarflexion and dorsiflexion up to the neutral position (0 degrees). Passive dorsiflexion stretching is absolutely contraindicated.
Phase III: Progressive Loading (Weeks 6 - 12)
The heel wedges are sequentially removed (one every 1-2 weeks) to slowly stretch the musculotendinous unit and restore a plantigrade foot. Full weight-bearing in the CAM boot without wedges is typically achieved by week 8. Gentle, closed-kinetic-chain strengthening exercises (e.g., seated heel raises, stationary cycling) are initiated.
Phase IV: Advanced Strengthening (Months 3 - 6)
The patient is transitioned to standard supportive footwear. Open and closed-kinetic-chain strengthening progresses aggressively. Proprioceptive training and eccentric loading protocols are introduced to stimulate collagen realignment and hypertrophy of the host triceps surae.
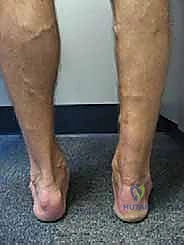
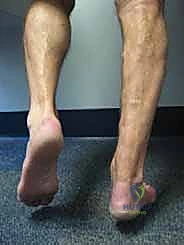
Phase V: Return to Activity (Months 6 - 12+)
Return to high-impact sports or heavy manual labor is delayed until 9 to 12 months postoperatively. Criteria for return to sport include symmetrical ankle ROM, normal gait mechanics, and isokinetic strength testing demonstrating at least 85-90% strength compared to the contralateral limb.
Summary of Landmark Literature and Clinical Guidelines
The evolution of chronic Achilles reconstruction is heavily documented in orthopedic literature. The foundational classification of Achilles defects by Kuwada (1990) established the paradigm that defects greater than 6 cm necessitate complex reconstruction, often involving synthetic grafts, autografts, or allografts.
Maffulli's extensive research into the histopathology of tendinopathy provided the biological rationale for radical debridement, proving that failure to excise myxomatous and fibrovascular tissue leads to high failure rates. Myerson's clinical algorithms further refined surgical decision-making, highlighting that while FHL transfer is excellent for moderate gaps or older, low-demand patients, massive structural allografts are biomechanically superior for younger, high-demand patients requiring restoration of the lever arm and maximal plantarflexion torque.
Recent biomechanical studies have validated the calcaneal bone block technique, demonstrating that bone-to-bone healing at the insertion site provides significantly higher ultimate failure loads and stiffness compared to soft-tissue-only anchors or transosseous sutures. Consequently, Achilles allograft with a calcaneal bone block remains the gold standard for massive, chronic defects in the active patient population, offering a reliable, structurally sound, and functionally restorative solution to one of orthopedics' most challenging pathologies.
