Unlock Optimal Bone Graft Anterior Harvest: Iliac Crest Guide
Introduction & Epidemiology
Autogenous bone graft remains the biological gold standard in orthopedic and reconstructive surgery due to its unique trifecta of osteoconductive matrix, osteoinductive growth factors, and osteogenic cells. The anterior iliac crest (AIC) is the most frequently utilized donor site for autogenous bone graft harvest, attributable to its accessibility, abundant bone stock, and favorable anatomical location relative to numerous recipient sites, particularly in spinal fusion and long bone non-union surgery.
The widespread application of AIC graft spans various subspecialties, including reconstructive orthopedics, spine surgery, craniofacial surgery, and trauma. Its versatility allows for the harvest of cortical, cancellous, or corticocancellous grafts, tailored to specific reconstructive demands. While allograft and synthetic bone graft substitutes offer advantages in terms of limitless supply and avoidance of donor site morbidity, their biological inferiority often necessitates augmentation or selection for lower demand fusion scenarios. The superior osteogenic potential of autograft, particularly from the iliac crest, continues to drive its preference in challenging fusion environments, revision surgeries, and compromised host settings. Understanding the nuanced surgical technique for AIC graft harvest is paramount to optimizing graft yield while minimizing donor site morbidity, which remains a significant concern for patients undergoing this procedure.
Surgical Anatomy & Biomechanics
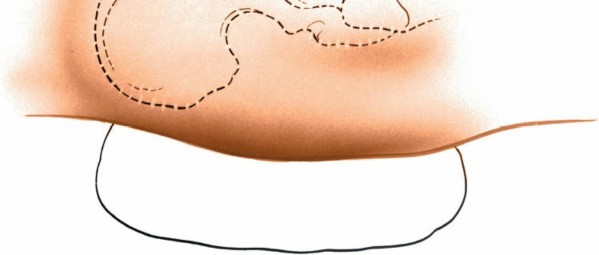
The iliac crest represents the superior border of the ala of the ilium and is a critical anatomical landmark for both palpation and surgical access. The anterior aspect of the iliac crest, specifically the region posterior to the anterior superior iliac spine (ASIS), offers a rich source of cancellous bone, encased by dense cortical bone of the inner and outer tables.
Bony Anatomy
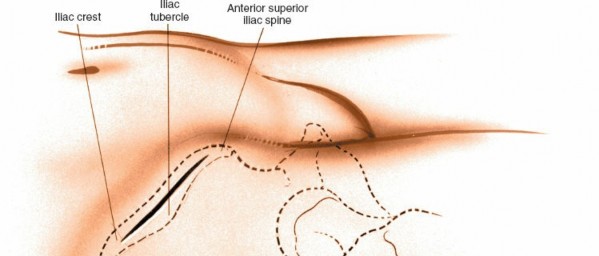
- Anterior Superior Iliac Spine (ASIS) : The most prominent anterior point of the iliac crest, serving as a key surgical landmark. It is the origin for the sartorius muscle and the tensor fasciae latae, and the inguinal ligament attaches here.
- Iliac Tubercle : Located approximately 5-7 cm posterior to the ASIS along the outer lip of the iliac crest, it marks the widest point of the iliac crest and is a useful reference for the origin of the iliotibial band.
- Outer Lip (External Oblique Line) : Provides origin for the tensor fasciae latae anteriorly, and the external and internal oblique muscles.
- Inner Lip (Internal Oblique Line) : Gives origin to the iliacus muscle and the transversus abdominis muscle.
- Intermediate Line : Lies between the outer and inner lips.
Neurovascular Anatomy
The vicinity of the AIC poses risks to several critical neurovascular structures. Meticulous dissection and retraction are essential for their preservation.
- Lateral Femoral Cutaneous Nerve (LFCN) : The most frequently injured nerve during anterior iliac crest harvest. It typically exits the pelvis near the ASIS, passing under or through the inguinal ligament, approximately 1-2 cm medial to the ASIS. Its course is variable, however, making it susceptible to direct injury during incision, retraction, or periosteal elevation. Injury can lead to meralgia paresthetica (pain, numbness, or paresthesia in the anterolateral thigh).
- Superior Cluneal Nerves : Terminal sensory branches of the dorsal rami of the L1-L3 spinal nerves. They cross the iliac crest approximately 7-8 cm posterior to the ASIS to innervate the skin of the upper gluteal region. While more commonly associated with posterior iliac crest harvest, larger or more posterior AIC harvests can place these nerves at risk.
- Deep Circumflex Iliac Artery and Vein : These vessels run along the inner lip of the iliac crest, deep to the iliacus muscle. While typically protected by the iliacus and the inner cortical table, aggressive inner table harvest or inadvertent penetration can lead to significant hemorrhage.
- Femoral Nerve : Originating from L2-L4, it passes under the inguinal ligament medial to the ASIS, providing motor innervation to the quadriceps and sensory innervation to the anterior thigh and medial leg. It is generally well protected during standard AIC harvest but is at risk with excessively medial or deep dissection.
- Ilioinguinal Nerve : Derived from L1, it pierces the transversus abdominis and internal oblique muscles and runs superficial to the spermatic cord. It innervates the skin of the superior medial thigh and parts of the external genitalia. It is at risk with incisions that extend too medially or with aggressive dissection of the abdominal wall musculature.
Muscular Attachments
-
Outer Table
:
- Sartorius : Originates from the ASIS.
- Tensor Fasciae Latae (TFL) : Originates from the outer lip of the iliac crest, posterior to the ASIS.
- External Oblique, Internal Oblique, Transversus Abdominis : Their aponeuroses attach to the outer lip of the iliac crest.
-
Inner Table
:
- Iliacus : Originates from the iliac fossa (inner surface of the ilium) and the inner lip of the iliac crest. This muscle forms part of the iliopsoas group and is a primary hip flexor.
Biomechanics of Graft Healing
The success of bone graft incorporation hinges on the principles of osteoconduction, osteoinduction, and osteogenesis. AIC autograft provides all three:
*
Osteoconduction
: The bony matrix acts as a scaffold for new bone ingrowth.
*
Osteoinduction
: Endogenous growth factors (e.g., bone morphogenetic proteins, BMPs) within the graft stimulate mesenchymal stem cells to differentiate into osteoblasts.
*
Osteogenesis
: Viable osteocytes, osteoblasts, and mesenchymal stem cells within the harvested cancellous bone directly contribute to new bone formation.
The biomechanical integrity of the donor site post-harvest is crucial. Removal of bicortical grafts, especially large ones, can compromise the structural strength of the ilium, leading to potential stress fractures, though this is rare with typical anterior harvest techniques. Careful attention to the remaining bony architecture is paramount.
Indications & Contraindications
The decision to utilize an anterior iliac crest bone graft is predicated on a careful assessment of patient-specific factors, surgical requirements, and the availability of suitable alternatives.
Primary Indications for AIC Autograft
-
Spinal Fusion
:
- Anterior Cervical Discectomy and Fusion (ACDF) : For interbody support and fusion, often as a tricortical strut graft.
- Anterior Lumbar Interbody Fusion (ALIF) : For interbody support and biological fusion.
- Spinal deformity correction : Particularly for anterior column reconstruction or extensive fusions requiring significant graft volume.
- Pseudarthrosis / Revision Spinal Fusion : Where previous fusion attempts have failed and maximum biological augmentation is desired.
- Long Bone Non-Unions : Especially in cases of atrophic or oligotrophic non-unions where biological stimulation and structural support are needed (e.g., tibia, femur, humerus).
-
Arthrodesis
:
- Foot and Ankle Arthrodesis : For fusion of joints (e.g., subtalar, tibiotalar) in patients with severe arthritis, deformity, or avascular necrosis.
- Hand and Wrist Arthrodesis : For carpal fusions or reconstruction following trauma or tumor resection.
- Craniofacial and Oral Maxillofacial Reconstruction : For alveolar ridge augmentation, mandibular reconstruction, or orbital floor repair.
- Oncologic Reconstruction : Following resection of benign or low-grade malignant bone tumors where reconstruction requires viable bone.
- Bone Defects : Filling cavitary defects from trauma, infection, or tumor excision.
- Joint Reconstruction : Augmentation of acetabular or glenoid defects during total hip or shoulder arthroplasty, respectively.
Considerations for Alternative Graft Sources or Contraindications to AIC Graft
| Category | Considerations / Contraindications |
|---|---|
| Local Factors | * Active Infection : Donor site infection or septicemia. |
| * Previous Harvest : Significant previous harvest from the ipsilateral iliac crest, leading to insufficient bone stock or compromised structural integrity. | |
| * Local Skin/Soft Tissue Compromise : Radiation dermatitis, severe scarring, burns, or dermatological conditions over the harvest site. | |
| * Tumor/Metastasis : Presence of primary bone tumor or metastatic disease within the ilium. | |
| * Inadequate Bone Stock : Osteoporosis or other metabolic bone diseases leading to poor bone quality and quantity, increasing risk of fracture. | |
| Systemic Factors | * Severe Coagulopathy : Uncontrolled bleeding disorder. |
| * Anemia : Severe anemia that cannot be corrected preoperatively, increasing transfusion risk. | |
| * Systemic Illnesses : Uncontrolled diabetes, severe malnutrition, or other conditions that impair wound healing or increase infection risk. | |
| Patient Preference | * Patient Refusal : Despite detailed counseling on the benefits and risks, some patients may decline autograft in favor of alternatives to avoid donor site morbidity. |
| Availability of Alternatives | * Allograft : When significant graft volume is required, or when donor site morbidity is a primary concern. Risks include disease transmission (though rare with current processing) and immunogenicity. Osteoconductive and weakly osteoinductive, but not osteogenic. |
| * Synthetic Bone Graft Substitutes : Calcium phosphates, ceramics, polymers. Primarily osteoconductive, some with osteoinductive properties (e.g., recombinant human BMP). Useful for small defects or augmentation, but lack osteogenic cells. | |
| * Demineralized Bone Matrix (DBM) : Osteoconductive and osteoinductive (due to residual BMPs), but variability in osteoinductive potential. | |
| * Mesenchymal Stem Cell (MSC) Augmentation : Concentrated bone marrow aspirate (BMA) or adipose-derived MSCs can be added to allograft or synthetics to enhance osteogenic potential, reducing the need for large autograft volumes. | |
| Surgical Requirements | * Minimal Graft Volume : For small defects, alternatives may suffice without the need for a separate donor site. |
| * Specific Graft Shape/Strength : For complex structural reconstructions where AIC graft may not provide the ideal morphology or biomechanical strength (e.g., vascularized fibula flap for large defects). |
Pre-Operative Planning & Patient Positioning
Thorough pre-operative planning and meticulous patient positioning are crucial steps to optimize access, maximize graft harvest, and minimize complications.
Pre-Operative Planning
- Patient Assessment : Review medical history for comorbidities that might affect wound healing, bleeding risk, or bone quality (e.g., diabetes, coagulopathies, osteoporosis). Assess prior abdominal or pelvic surgeries that might alter local anatomy.
- Radiographic Evaluation : While not always strictly necessary for a straightforward AIC harvest, a pre-operative pelvic radiograph or CT scan can assess iliac crest morphology, bone stock, and identify any previous donor site involvement or pathology. This is particularly relevant for large reconstructive cases.
- Informed Consent : Comprehensive discussion with the patient regarding the indications for autograft, the specific harvest site (anterior vs. posterior), expected graft volume, potential donor site complications (e.g., pain, numbness, hematoma, infection, fracture), and the possibility of utilizing alternative graft sources if harvest is insufficient or contraindicated.
- Site Selection : Determine the ipsilateral or contralateral side based on the primary surgical site, patient positioning, and surgeon preference. Often, the ipsilateral side is chosen for logistical reasons.
Patient Positioning
The patient is typically placed in the supine position on the operating table, allowing for simultaneous access to the primary surgical site (e.g., anterior cervical spine, abdomen) and the iliac crest.
- Arm Positioning : The ipsilateral arm should be abducted on an arm board, allowing adequate space for the surgical team.
-
Torso Elevation
: A small sandbag, bolster, or folded blanket is placed
under the gluteal (cluneal) area of the side from which the graft will be taken
. This maneuver serves two critical purposes:
- Elevates the Iliac Crest : Brings the crest into a more accessible and prominent position.
-
Internally Rotates the Pelvis
: Facilitates access to the outer table of the ilium and helps to flatten the contour of the abdominal wall near the crest, making the dissection less challenging.

- Draping : The iliac crest should be prepared and draped as a separate, sterile surgical field, allowing for adequate exposure while maintaining sterility for the primary procedure. Many surgeons prefer a "double-drape" technique, creating a distinct isolation of the harvest site. This ensures that the primary surgical field is not contaminated by instruments used for graft harvest, which can be particularly true if the harvest is performed by a separate team.
Detailed Surgical Approach / Technique
The anterior approach to the iliac crest for bone graft harvest requires a systematic, layered dissection to optimize graft yield and minimize iatrogenic injury.
Landmarks and Incision
-
Palpation of Landmarks
:
- The anterior superior iliac spine (ASIS) is the most critical and easily palpable landmark. It is vital to accurately identify the ASIS and trace the contour of the iliac crest posteriorly.
-
Continue palpating along the superior border of the iliac crest to determine the desired extent of the harvest.

-
Incision Placement
:
- The incision is typically made 2-3 cm posterior and parallel to the ASIS. This curvilinear incision follows the natural skin lines (Langer's lines) and the contour of the iliac crest.
- The length of the incision is dictated by the amount of graft required. For small cancellous grafts, a 3-5 cm incision may suffice. For larger cortical or corticocancellous grafts, an incision of 5-10 cm or more may be necessary.
- Some surgeons prefer an incision directly over the ASIS, extending posteriorly, especially if a large tricortical graft incorporating the ASIS is needed, though this may increase the risk of LFCN injury and cosmetic deformity.
-
Another variant is a transverse incision just inferior to the ASIS, or an oblique incision that starts just posterior to the ASIS and extends superiorly and posteriorly. This can be beneficial for nerve protection.

Dissection to the Iliac Crest
- Skin and Subcutaneous Tissue : Incise the skin and subcutaneous tissue down to the superficial fascia. Careful hemostasis should be maintained.
-
Superficial Fascia and Nerve Protection
:
- Cautious dissection through the superficial fascia is crucial. This is the layer where the terminal branches of the lateral femoral cutaneous nerve (LFCN) may be encountered.
- Use blunt dissection with small retractors or scissors to identify and gently retract any visible nerve branches. The LFCN typically runs approximately 1-2 cm medial to the ASIS, but its course is highly variable. If identified, it should be protected and retracted carefully, preferably superiorly and medially, away from the operative field.
- Pearl : A common technique to minimize LFCN injury is to place the incision slightly posterior to the ASIS and perform subperiosteal dissection immediately, minimizing superficial soft tissue stripping anteriorly where the nerve is most vulnerable.
- Deep Fascia : Incise the deep fascia (fascia lata and oblique aponeurosis) overlying the iliac crest.
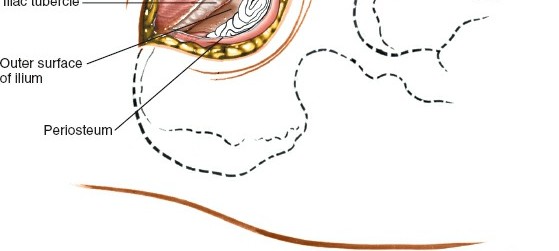
Subperiosteal Exposure of the Iliac Crest
- Identification of Muscle Origins : At the ASIS, identify the origins of the sartorius muscle (anterior-most aspect) and the tensor fasciae latae (posterior to sartorius on the outer lip).
- Periosteal Incision : Make a longitudinal incision through the periosteum directly along the superior aspect of the iliac crest. This incision should extend for the desired length of the graft.
-
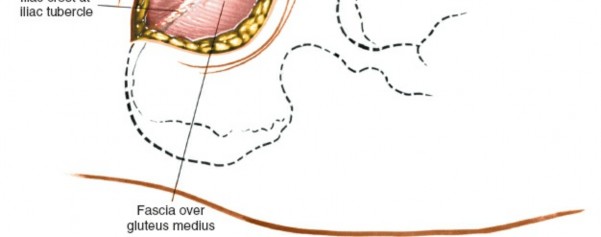
Subperiosteal Elevation (Outer Table)
:
- Using a Cobb elevator or similar periosteal elevator, carefully strip the muscle origins from the outer lip of the iliac crest. This includes the sartorius, tensor fasciae latae, and the abdominal wall musculature (external oblique, internal oblique, transversus abdominis).
- Elevate these muscles subperiosteally in an inferior and lateral direction. Ensure that the elevation is kept tight to the bone to protect the overlying soft tissues and nerves.
-
Caution
: Avoid excessive retraction or forceful elevation anteriorly, which can stretch or directly injure the LFCN.
-
Subperiosteal Elevation (Inner Table)
:
- Direct the periosteal elevator medially to elevate the iliacus muscle from the inner lip of the iliac crest and the iliac fossa. This exposes the inner cortical table.
- The iliacus muscle should be gently retracted inferiorly and medially.
-
Caution
: Be mindful of the deep circumflex iliac vessels running along the inner table, deep to the iliacus. Aggressive medial stripping or uncontrolled instrument insertion can injure these vessels.
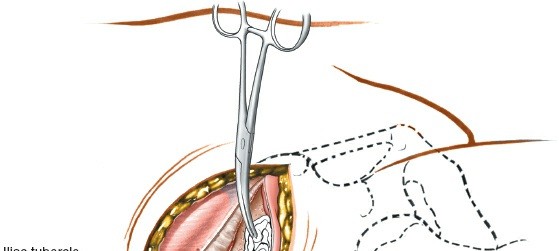
Graft Harvest
The type of graft harvested (cancellous, cortical, or corticocancellous) depends on the requirements of the recipient site.
-
Cancellous Graft Harvest
:
- Most commonly harvested from the medullary cavity between the inner and outer tables of the iliac wing, posterior to the ASIS.
- Using an osteotome or oscillating saw, create a cortical window (typically unicortical, from the outer table) to access the cancellous bone. The dimensions of the window should be sufficient for graft retrieval but kept as small as possible to preserve donor site integrity.
- Once the cortical window is made, curettes, gouges, or specialized bone graft harvesters are used to scoop out the cancellous bone.
- Pearl : The "wing" of the ilium, posterior to the ASIS, offers the richest source of cancellous bone.
-
Monocortical Graft Harvest
:
- Involves removing a segment of either the outer or inner cortical table along with the underlying cancellous bone.
- Typically performed using osteotomes or an oscillating saw to create the desired size and shape.
- Used for non-structural applications or for augmenting fusion sites.
-
Corticocancellous (Bicortical/Tricortical) Graft Harvest
:
- Requires removal of both the outer and inner cortical tables, along with the intervening cancellous bone. This provides a structural graft with osteoconductive, osteoinductive, and osteogenic properties.
- Often used for spinal interbody fusion (e.g., ACDF, ALIF) or reconstructing significant bone defects.
- The size and shape are carefully templated based on recipient site requirements.
- Use an oscillating saw or osteotomes to make precise cuts.
-
Technique for Bicortical Graft
:
- Make parallel cuts through the outer cortex with an oscillating saw, defining the length and width of the desired graft.
- Then, make a perpendicular cut at one end to allow a lever point.
- Carefully use a straight osteotome or gouge to separate the inner cortex, ensuring controlled force to avoid unintended fracture of the remaining ilium.
- Caution : When harvesting bicortical grafts, be aware of the depth of the cuts to avoid entering the pelvic cavity prematurely or injuring intrapelvic structures. The ilium is thicker anteriorly and then thins out posteriorly.
-
Tricortical Graft (e.g., for ACDF)
: Often harvested from the ASIS itself, providing a trapezoidal or cuboid block with three cortical surfaces. This is a robust structural graft.

Hemostasis and Wound Closure
-
Hemostasis of the Donor Site
:
- Once the graft is harvested, meticulous hemostasis of the donor site is critical to prevent hematoma formation.
- Bone wax is applied to the cut bony surfaces to seal bleeding cancellous bone.
- Electrocautery for soft tissue bleeding.
- Placement of absorbable hemostatic agents (e.g., Gelfoam, Surgicel) soaked in thrombin can be beneficial within the defect.
- Some surgeons advocate for packing the defect with synthetic bone graft substitute or allograft chips to prevent collapse and facilitate healing, though this is debated.
-
Donor Site Closure
:
- Periosteum/Muscle Repair : The elevated periosteum and muscle origins (sartorius, TFL, abdominal obliques, iliacus) should be meticulously reapproximated to the iliac crest using strong, absorbable sutures. This helps to restore the anatomical barrier, reduce dead space, minimize potential for hernia, and decrease post-operative pain.
- Fascial Closure : The deep fascia should be closed.
- Subcutaneous Closure : Close the subcutaneous tissue in layers to obliterate dead space.
- Skin Closure : Skin is closed with sutures or staples.
-
Drainage
: A suction drain (e.g., Jackson-Pratt) is often placed in the donor site, especially after larger harvests, to prevent hematoma and seroma formation. The drain is typically removed when output is minimal (<30-50 mL/24h).

Complications & Management
Donor site morbidity is the most significant drawback of autogenous bone graft harvest. A thorough understanding of potential complications and their management is essential for effective patient care.
| Complication | Incidence (Approximate) | Salvage Strategies / Management |
|
Pain at Harvest Site
| 60-80% initially, diminishing over 6-12 months. Chronic pain >6 months 5-15% | *
Conservative
: NSAIDs, analgesics (opioids for severe acute pain), ice/heat, rest, physical therapy.
Interventional
: Local anesthetic injections, steroid injections, radiofrequency ablation for persistent neuropathic pain.
Surgical
: Excision of painful neuroma if intractable. |
|
Donor Site Pain (Chronic)
| 5-15% (can be higher with posterior approaches) | *
Conservative
: Analgesics, physical therapy, nerve blocks, capsaicin/lidocaine patches.
Interventional
: Steroid injections, radiofrequency ablation, peripheral nerve stimulation.
Surgical
: Excision of painful neuroma or scar tissue. Revision repair of fascial defect if indicated. |
|
Neurovascular Injury (e.g., LFCN)
| LFCN injury incidence 0.5-20% (symptomatic 0.5-5%) | *
Conservative
: Analgesics, NSAIDs, neuropathic pain medications (gabapentin, pregabalin), local anesthetic nerve blocks, topical lidocaine/capsaicin.
Surgical
: Neurolysis or neurectomy for persistent, debilitating symptoms after conservative failure (rare). |
|
Neuroma (Chronic Nerve Pain)
| 0.5-5% (higher likelihood in specific nerve pathways) | *
Conservative
: As above for chronic pain.
Interventional
: Targeted nerve blocks with local anesthetic/steroid, pulsed radiofrequency.
Surgical
: Excision of the neuroma and burying the nerve end in muscle or bone to prevent recurrence. Nerve repair/grafting if significant motor deficit or large sensory area affected (rare for LFCN). |
|
Infection
| 0.5-5% (can be superficial or deep) | *
Conservative
: Local wound care, topical/oral antibiotics for superficial infections.
Surgical
: Incision and drainage, debridement of necrotic tissue, removal of foreign material, IV antibiotics based on culture sensitivities for deep infections. Wound VAC therapy may be necessary. |
|
Hematoma / Seroma
| 2-10% (Hematoma higher with inadequate hemostasis, seroma with poorly closed dead space) | *
Conservative
: Observation for small, stable collections. Compression, ice.
Interventional
: Aspiration for symptomatic seromas or expanding hematomas. Consider drain reinsertion if collection recurs.
Surgical
: Evacuation and washout for large, symptomatic, or infected hematomas. Meticulous hemostasis and dead space closure. |
|
Hernia (less common in AIC)
| < 1% (anterior approach has a low rate due to muscular/fascial strength) | *
Surgical
: Hernia repair with direct approximation of fascial layers. Mesh augmentation rarely required for primary repair but considered for recurrent defects or large fascial deficiencies. |
|
Iliac Crest Fracture
| Rare, but possible with aggressive or deep harvest, particularly with bicortical/tricortical grafts. | *
Conservative
: Non-weight-bearing (if severe) or protected weight-bearing, pain management. Most heal with conservative care due to inherent stability of pelvic ring. |
|
Cosmetic Deformity
| Subjective, 1-5% (depressed scars, contour deformities) | *
Surgical
: Scar revision, fat grafting, dermal fillers, or allograft bone chips to augment contour. Surgical correction of contour deformities. |
|
Bleeding / Hematoma
| 2-10% | *
Conservative
: Pressure dressing, ice, close monitoring. |
|
Deep Vein Thrombosis (DVT) / Pulmonary Embolism (PE)
| Uncommon (<1%) but potentially fatal. Increases with prolonged immobility or hypercoagulable states. | *
Prevention
: Early mobilization, prophylactic anticoagulation, mechanical thromboprophylaxis (e.g., sequential compression devices).
Management
: Anticoagulation (heparin, LMWH, DOACs) for acute DVT/PE. IVC filter for contraindications to anticoagulation or recurrent PE. |
|
Deep Venous Thrombosis (DVT) and Pulmonary Embolism (PE)
| Overall rate <1%, but potentially lethal. Increased risk with prolonged immobility, hypercoagulable states. | *
Prevention
: Early ambulation, pneumatic compression devices, prophylactic anticoagulation (LMWH or fondaparinux) for high-risk patients.
Management
: Full therapeutic anticoagulation for DVT/PE. IVC filter for contraindications to anticoagulation or recurrent PE. |
|
Deep Venous Thrombosis (DVT) / Pulmonary Embolism (PE)
| Overall rate <1%, but potentially lethal. Increases with prolonged immobility, hypercoagulable states. | *
Prevention
: Early ambulation, pneumatic compression devices, prophylactic anticoagulation (LMWH or fondaparinux) for high-risk patients.
Management
: Full therapeutic anticoagulation for DVT/PE. IVC filter for contraindications to anticoagulation or recurrent PE. |
|
Deep Venous Thrombosis (DVT) / Pulmonary Embolism (PE)
| Overall rate <1%, but potentially lethal. Increases with prolonged immobility, hypercoagulable states. | *
Prevention
: Early ambulation, pneumatic compression devices, prophylactic anticoagulation (LMWH or fondaparinux) for high-risk patients.
Management
: Full therapeutic anticoagulation for DVT/PE. IVC filter for contraindications to anticoagulation or recurrent PE. |
|
Swelling and Bruising
| Near universal (90-100%), typically resolves within weeks. | *
Conservative
: R.I.C.E. (Rest, Ice, Compression, Elevation). Analgesics. Early ambulation to promote lymphatic drainage. |
|
Gait Disturbance
| Less than 1%, possible if harvest extends close to hip joint capsule or involves significant muscle injury | *
Conservative
: Physical therapy, crutches or walker for support, gradual resumption of ambulation. |
|
Post-operative pain
| Expected and managed; up to 80% experience moderate-severe pain post-op | *
Pharmacological
: Multimodal analgesia including NSAIDs, acetaminophen, local anesthetic infiltration, regional blocks, and opioids as needed.
Physical
: Ice, rest, controlled activity. Early mobilization. |
|
Gait disturbance or Limp
| Less than 1% | *
Conservative
: Physical therapy to strengthen hip abductors, core stability, gait training. Crutches or walker for temporary support. |
|
Sexual Dysfunction (rare, usually transient) | Rare, possibly associated with nerve injury or psychological factors. | *
Conservative
: Counseling, medication adjustments if applicable. |
|
Chronic Pain
| 5-15% of patients after AIC harvest. Leads to functional limitations and reduced quality of life. | *
Multimodal approach
: Pharmacotherapy (NSAIDs, neuropathic agents like gabapentin/pregabalin, sometimes low-dose antidepressants), physical therapy, cognitive behavioral therapy, and interventional pain management (nerve blocks, radiofrequency ablation, spinal cord stimulation in rare intractable cases).
Surgical
: Targeted neurectomy for specific painful neuromas, or revision of painful nonunion of the iliac crest fragment (extremely rare). |
|
Damage to the inguinal ligament
| Rare, depends on the incision location and depth of dissection. | *
Surgical
: Direct repair of the ligament. Physical therapy for core and abdominal strengthening to compensate. |
|
Inadequate Graft Volume
| Potential limitation if requirements exceed harvestable quantity or quality. | *
Consider
: Bone graft substitutes (allograft, synthetic, DBM, cells). Consider repeat procedure on contralateral side if needed, but increased morbidity. |
|
Inadequate Graft Volume
| Potential limitation if requirements exceed harvestable quantity or quality. | *
Consider
: Bone graft substitutes (allograft, synthetic, DBM, cells). Consider repeat procedure on contralateral side if needed, but increased morbidity. |
|
Anesthetic Complications
| All risks inherent to general anesthesia, including allergic reactions, cardiac events, pulmonary events, and neurologic deficits. | *
Preventative
: Thorough pre-anesthetic evaluation, optimal patient preparation, meticulous technique during induction and maintenance, continuous monitoring of vital signs, advanced airway management strategies.
Management
: Immediate intervention following advanced cardiac life support (ACLS) and advanced trauma life support (ATLS) guidelines. Reversal agents for specific medications. Consultation with anesthesiology critical care. |
|
Deep Venous Thrombosis (DVT) and Pulmonary Embolism (PE)
| Overall rate <1%, but potentially lethal. Increased risk with prolonged immobility, hypercoagulable states. | *
Prevention
: Early ambulation, pneumatic compression devices, prophylactic anticoagulation (LMWH or fondaparinux) for high-risk patients.
Management
: Full therapeutic anticoagulation for DVT/PE. IVC filter for contraindications to anticoagulation or recurrent PE. |
|
Contralateral Leg Pain
| 1-3% | *
Conservative
: NSAIDs, physical therapy, rest. |
Clinical & Radiographic Imaging