AMPUTATIONS IN CHILDREN: ACQUIRED ETIOLOGIES AND SURGICAL MANAGEMENT
Amputations in the pediatric population represent a unique and highly specialized domain within orthopedic surgery. Unlike adult amputations, which are predominantly driven by dysvascular disease or severe diabetic neuropathy, pediatric amputations are fundamentally influenced by the dynamic processes of skeletal growth, physeal development, and continuous motor maturation.
Pediatric limb deficiencies are broadly classified into two distinct categories: congenital and acquired. Epidemiological surveys from specialized pediatric amputee centers indicate that approximately 60% of childhood amputations are secondary to congenital limb deficiencies, whereas 40% are secondary to acquired conditions. This chapter focuses exclusively on the surgical management, biomechanical considerations, and longitudinal care of acquired amputations in children.
Etiology of Acquired Pediatric Amputations
Acquired amputations in children are most frequently necessitated by severe trauma, followed by neoplastic disease and fulminant infections.
- Trauma: The mechanisms of injury vary significantly by age cohort. In older children and adolescents, high-energy trauma such as motor vehicle accidents (MVAs), gunshot wounds, and injuries from heavy power tools are the leading causes of limb loss. Conversely, in toddlers and young children, household accidents—most notably involving rotary lawnmowers—are the predominant cause of traumatic amputation.
- Neoplasm: Primary bone sarcomas (e.g., osteosarcoma, Ewing sarcoma) may necessitate amputation when limb-salvage surgery is contraindicated due to neurovascular involvement or massive soft-tissue extension.
- Infection: Severe systemic infections, such as meningococcemia leading to purpura fulminans, can result in peripheral ischemia and gangrene, necessitating multiple symmetric amputations.
- Dysvascular Conditions: Unlike the adult population, dysvascular amputations in children are exceedingly rare. When they do occur, they are typically secondary to acute thrombotic or embolic events associated with underlying systemic anomalies, such as Kawasaki disease, severe coagulopathies, or iatrogenic vascular complications.
FUNDAMENTAL PRINCIPLES OF PEDIATRIC AMPUTATION SURGERY
While many fundamental surgical techniques utilized in adult amputations are applicable to children, the overriding factors of general somatic growth and specific stump growth dictate a paradigm shift in surgical decision-making. Krajbich established six foundational principles for childhood amputation surgery that remain the gold standard in pediatric orthopedics:
- Preserve Length at All Costs: Length preservation is the most critical factor in pediatric amputation. Because children are actively growing, a stump that appears adequate at age two may become functionally useless by skeletal maturity. For example, 70% to 75% of the longitudinal growth of the femur occurs at the distal femoral physis. Consequently, a transfemoral amputation performed in a toddler will result in an extremely short, mechanically disadvantaged stump in adulthood.
- Preserve Important Growth Plates: The retention of physes ensures proportional growth of the residual limb. Even a very short transtibial stump in a young child can develop into a highly functional stump by adulthood, provided the proximal tibial physis remains intact and functional.
- Perform Disarticulation Rather Than Transosseous Amputation: Whenever anatomically feasible, disarticulation (e.g., Syme, knee, or elbow disarticulation) is vastly superior to transosseous amputation in the pediatric patient.
- Preserve the Knee Joint: The energy expenditure required for ambulation increases exponentially with the loss of the knee joint. Preserving the anatomical knee joint is paramount for optimizing the child's functional mobility and reducing the metabolic cost of walking.
- Stabilize and Normalize the Proximal Portion of the Limb: Ensure that proximal joints (e.g., the hip in a lower extremity amputation) are stable, congruent, and free of contractures to maximize the biomechanical efficiency of the residual limb.
- Manage Comorbidities: The surgeon must be prepared to address concurrent clinical issues, such as traumatic brain injury, polytrauma, or psychological distress, which frequently accompany traumatic limb loss.
Clinical Pearl: The metabolic cost of ambulation for a child with a transtibial amputation is approximately 10-15% higher than normal, whereas a transfemoral amputation increases energy expenditure by up to 65%. This underscores the absolute necessity of preserving the knee joint and the proximal tibial physis whenever oncologically and traumatologically safe.
DISARTICULATION VS. TRANSOSSEOUS AMPUTATION
In pediatric orthopedics, disarticulation is the procedure of choice. It provides the child with a well-balanced, sturdy residual limb capable of direct end-weight bearing.
Biomechanical and Prosthetic Advantages of Disarticulation
- Prevention of Terminal Overgrowth: Disarticulation completely eliminates the risk of terminal bone overgrowth, the most common and troublesome complication of pediatric transosseous amputations.
- Physeal Preservation: By disarticulating through the joint, the adjacent physes are preserved, ensuring continued longitudinal growth of the residual limb segment.
- Enhanced Prosthetic Suspension: Disarticulation preserves the natural metaphyseal flares (e.g., the femoral condyles or the malleoli). These bulbous bony contours provide excellent anatomical anchors for prosthetic suspension, eliminating the need for cumbersome belts, harnesses, or silicone suction lanyards. This is particularly critical given the high mechanical demands and rigorous activity levels of pediatric patients.
TERMINAL BONE OVERGROWTH: PATHOPHYSIOLOGY AND MANAGEMENT
Terminal bone overgrowth is a highly significant, uniquely pediatric complication that occurs almost exclusively following transosseous amputations. It is characterized by the formation of a sharp, pencil-shaped bony spike at the distal end of the transected bone.
Pathophysiology
It is a common misconception that terminal overgrowth is driven by the proximal physis pushing the bone distally. In reality, overgrowth is caused by appositional new bone formation at the distal transected end of the medullary canal. The periosteum and endosteum remain highly osteogenic in children, and without the regulatory presence of an articular surface or a closed physis, bone continues to form longitudinally.
Clinical Presentation and Incidence
The resulting elongated, pencil-shaped bone spike acts as an internal spear. It causes progressive soft-tissue tension, leading to:
* Localized edema and erythema.
* Intractable pain with prosthetic wear.
* Adventitious bursa formation over the bone spike.
* In severe, neglected cases, frank penetration of the bone through the distal skin flap.
Overgrowth is most frequently observed following traumatic amputations. It is highly age-dependent, occurring much more frequently in younger children (under age 10) than in adolescents. The anatomical incidence, in decreasing order of frequency, is: Humerus > Fibula > Tibia > Femur > Radius > Ulna.
Surgical Warning: Epiphysiodesis (surgical arrest of the proximal growth plate) is strictly contraindicated for the treatment of terminal overgrowth. It does not halt the distal appositional bone formation and only serves to unnecessarily shorten the residual limb, compounding the patient's functional deficit.
Treatment Modalities
Historically, capping the transected bone with synthetic devices (e.g., silicone, polyethylene, or titanium caps) was attempted. However, these have demonstrated unacceptably high failure rates due to foreign body reactions, deep infections, bursa formation, and mechanical fracture of either the implant or the host bone.
Today, terminal overgrowth is treated effectively with surgical resection of the excess bone, ideally combined with an autologous biological capping procedure.
SURGICAL TECHNIQUE: THE STUMP CAPPING PROCEDURE
To prevent recurrent terminal overgrowth, the gold standard technique involves capping the transected medullary canal with an autologous osteochondral or epiphyseal graft. This biological cap acts as a physical barrier to appositional bone formation and integrates seamlessly with the host bone.
If the capping is performed during the index procedure (the initial amputation), the graft is typically harvested from the amputated limb (e.g., the distal fibula, a metatarsal head, or a phalangeal physis). If performed as a revision procedure for established overgrowth, a tricortical graft from the iliac crest is utilized.
Step-by-Step Epiphyseal Grafting (Index Procedure)
Step 1: Exposure and Preparation of the Host Bone
The distal aspect of the residual limb is exposed. In cases of revision, the bursa and the pencil-shaped overgrowth are carefully excised. The medullary canal of the host bone (e.g., the tibia) is identified and reamed slightly to accept the graft.
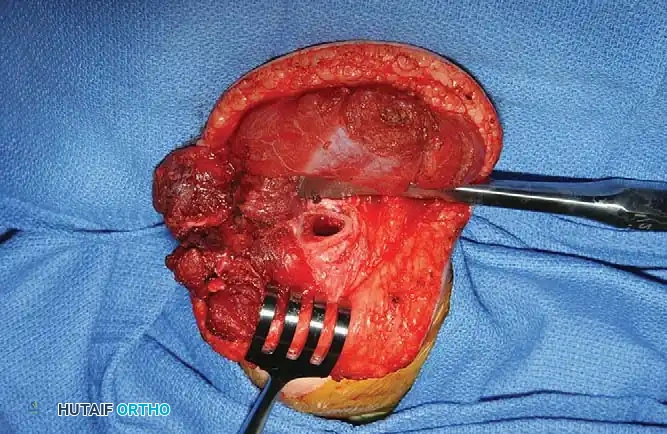
Fig. 9-6A: Initial exposure of a transtibial amputation in a 2-year-old child, demonstrating the transected distal tibia requiring biological capping to prevent future overgrowth.
Step 2: Graft Harvest and Contouring
The surgeon harvests an appropriate epiphyseal segment from the amputated, non-viable portion of the limb. The distal fibula is an excellent donor site for a transtibial amputation. The graft is meticulously fashioned using a rongeur or a fine burr. The goal is to create a "mushroom" shape: a narrow peg of diaphyseal bone designed to fit into the host medullary canal, topped by a wider cap of epiphyseal cartilage.
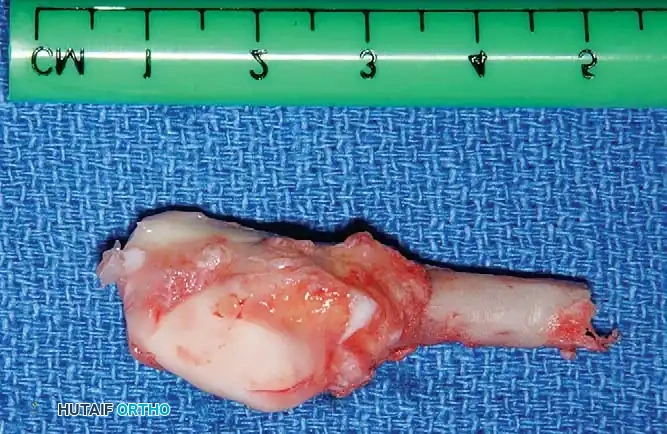
Fig. 9-6B: The distal fibula, harvested from the amputated leg, is carefully fashioned and contoured to fit precisely into the cut end of the host tibia.
Step 3: Implantation and Fixation
The fashioned fibular graft is inverted and press-fit into the prepared medullary canal of the tibia. The cartilaginous cap must completely cover the exposed cortical edges of the host bone to effectively arrest appositional osteogenesis. If the press-fit is not inherently stable, it may be secured with heavy non-absorbable transosseous sutures or temporary smooth Kirschner wires.
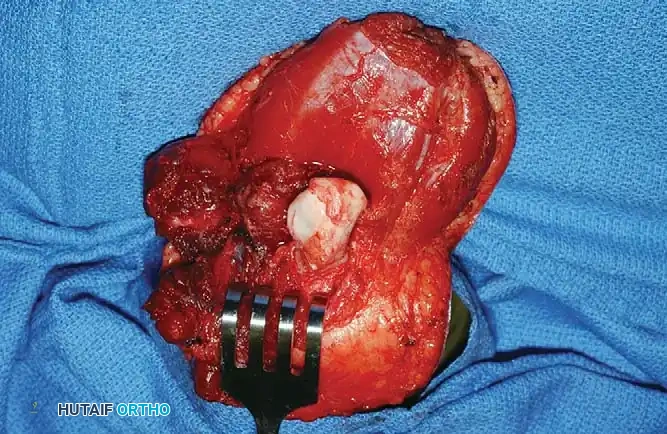
Fig. 9-6C: The contoured fibular graft is securely press-fit into the medullary canal of the tibia, providing a biological cartilaginous cap that will prevent terminal bone overgrowth.
Step 4: Soft Tissue Coverage
The myocutaneous flaps are closed over the capped bone. Because of the robust vascularity in children, flaps can be closed under moderate tension if necessary, though a tension-free closure remains the ideal standard.
PEDIATRIC TISSUE TOLERANCE AND COMPLICATION PROFILES
Children possess a remarkable physiological resilience that significantly alters postoperative management and complication profiles compared to adults. Due to rapid cellular turnover, increased basal metabolic rates, and pristine vascular networks, pediatric patients can tolerate interventions that would routinely fail in older populations.
Soft Tissue Management
- Skin Traction: Children can tolerate forceful skin traction to stretch soft tissues over a bone end, a technique that would almost certainly cause ischemic skin necrosis in an adult dysvascular patient.
- Skin Grafting: Extensive split-thickness or full-thickness skin grafts take readily on pediatric amputation stumps and are highly durable, even in weight-bearing areas, provided they are properly managed with modern silicone prosthetic liners.
- Tension Closure: The closure of skin flaps under moderate tension is generally well-tolerated and rarely leads to wound dehiscence.
Neurological and Pain Complications
- Phantom Limb Pain: While adults frequently suffer from debilitating phantom limb pain, this phenomenon is exceedingly rare in children, particularly those amputated at a very young age. Phantom sensations may be present but are almost never painful.
- Neuromas: Terminal neuromas inevitably form at the transected ends of major nerves. However, in children, these neuromas rarely become symptomatic or troublesome enough to require surgical excision. Standard traction neurectomy (pulling the nerve distally, transecting it sharply, and allowing it to retract deep into the muscle belly) is highly effective.
Bony Spurs
It is important to differentiate terminal overgrowth from bony spurs. One or more osteophytic spurs frequently develop at the margins of the transected bone, typically at the site of periosteal stripping or muscle attachment. Unlike terminal overgrowth, these spurs grow laterally or medially (traction exostoses) rather than longitudinally. They almost never penetrate the skin, rarely cause significant prosthetic fitting issues, and almost never require surgical resection.
PROSTHETIC MANAGEMENT AND LONGITUDINAL CARE
The rehabilitation and prosthetic fitting of a child amputee must be viewed as a dynamic, longitudinal process that parallels normal motor and cognitive development. The multidisciplinary team—comprising the orthopedic surgeon, prosthetist, physical therapist, and pediatric psychologist—must continuously adapt the treatment plan as the child grows.
Developmental Prosthetic Progression
- Infants and Toddlers (6 to 15 months): Prosthetic fitting should commence when the child begins to pull to a stand, matching the normal developmental milestone for ambulation. Initial prostheses should be simple, highly durable, and lightweight. For transfemoral amputees of this age, the prosthesis is typically fabricated without an articulated knee joint. A locked, straight leg provides the necessary stability for early walking and prevents the child from collapsing under their own weight.
- Preschool and Early School Age (3 to 7 years): As the child's balance and gait mechanics mature, modifications are introduced. Articulated knee joints are added to transfemoral prostheses. For upper extremity amputees, mobile elbow joints or simple mechanical terminal devices (e.g., a voluntary-opening hook) are integrated to assist with bimanual play and self-feeding.
- Adolescence (12 to 18 years): Teenagers place immense mechanical and social demands on their prostheses. They can take full advantage of sophisticated technology, including dynamic-response carbon fiber feet, microprocessor-controlled knees, and myoelectric upper limb prostheses. Specialized "activity-specific" prostheses (e.g., running blades, swimming attachments, or weightlifting hooks) should be prescribed to encourage athletic participation and social integration.
Psychological Considerations
Psychological distress following an amputation is surprisingly rare in young children, who generally adapt to their limb difference with remarkable resilience. However, as children reach adolescence, body image, peer acceptance, and self-esteem become paramount. The psychological impact of the amputation may suddenly become severe, manifesting as depression, social withdrawal, or rejection of the prosthesis. Proactive psychological screening and peer-support groups (e.g., amputee camps) are essential components of adolescent care.
The Importance of Close Observation
Because of their high activity levels and continuous skeletal growth, children require meticulous, ongoing observation. A well-fitting socket can become unacceptably tight within a matter of months due to somatic growth. Surgeons and prosthetists must schedule frequent follow-ups to monitor for:
* Skin breakdown or excessive pressure over bony prominences.
* The need for socket modifications or complete prosthetic replacements.
* Early signs of terminal bone overgrowth requiring radiographic evaluation.
* Joint contractures, which must be aggressively managed with physical therapy and dynamic splinting.
